Research Article
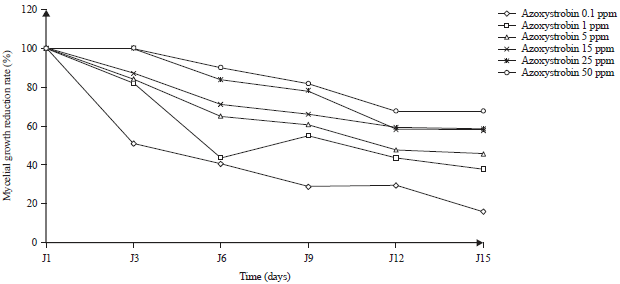
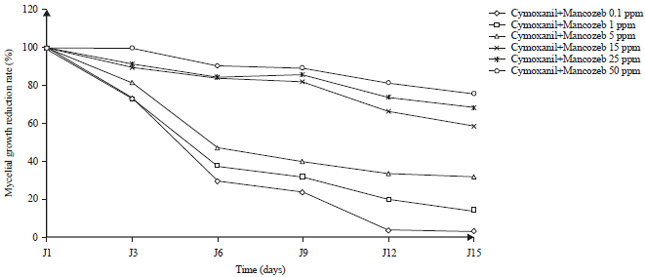
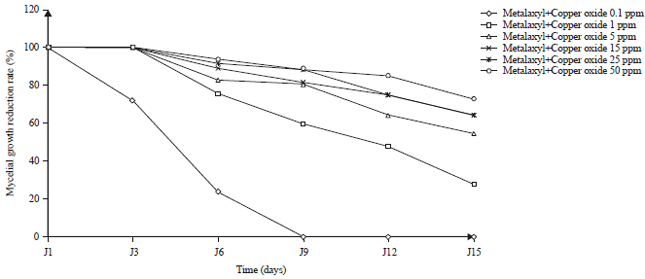
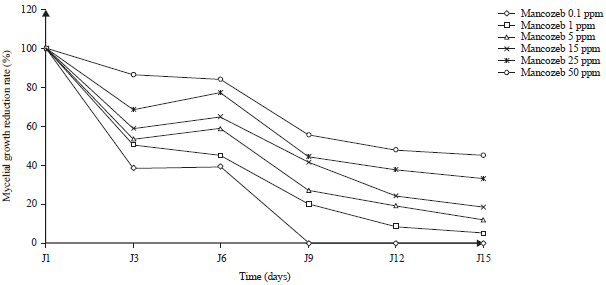
Effect of Some Synthetic Fungicides on the in vitro Growth of Colletotrichum gloeosporioides, Causative Agent of Cashew Tree Anthracnose in Côte d’ivoire
Laboratoire de Physiologie Vegetale, UFR Biosciences, Universite Felix Houphouet-Boigny, 22 BP 582 Abidjan 22, Cote d�Ivoire
LiveDNA: 225.19956
Abo Kouabenan
Laboratoire de Phytopathologie, Institut National Polytechnique Felix Houphouet-Boigny de Yamoussoukro, BP 1313 Yamoussoukro, Cote d`Ivoire
Camara Brahima
Laboratoire de Physiologie Vegetale, UFR Biosciences, Universite Felix Houphouet-Boigny, 22 BP 582 Abidjan 22, Cote d�Ivoire
Soro Sibirina
3Laboratoire de Physiologie Vegetale, UFR Agroforesterie, Universite Jean Lorougnon Guede de Daloa, BP 150 Daloa, Cote d`Ivoire
LiveDNA: 225.18303
Ouattara Gnire Mariam
Universite Alassane Ouattara, BP V 18 Bouake 01, Cote d`Ivoire
Tuo Seydou
Laboratoire de Physiologie Vegetale, UFR Biosciences, Universite Felix Houphouet-Boigny, 22 BP 582 Abidjan 22, Cote d�Ivoire
LiveDNA: 225.12582
Kone Mongomake
Laboratoire de Biologie et Amelioration des Productions Vegetales, UFR des Sciences de la Nature, Universite Nangui Abrogoua, 02 BP 801 Abidjan 02, Cote d`Ivoire
Kone Daouda
Laboratoire de Physiologie Vegetale, UFR Biosciences, Universite Felix Houphouet-Boigny, 22 BP 582 Abidjan 22, Cote d�Ivoire
LiveDNA: 225.16680