ABSTRACT
This study investigates, using random effect modeling, the relevant risk factors associated with perinatal mortality at the various stages of perinatal mortality. We found that there is general correlation pattern between the stages of perinatal mortality. The conventional analysis results of the data showed, the relevant maternal risk factors in the various stages are different and the circumstances of individuals change substantially from stage to stage. It is found that allowing for the correlation between stages changes the magnitude of the covariate estimates but the extent of the change is not the same for all. Such that some relevant variables that are not significant in the conventional analysis become significant in random effect analysis. Therefore this correlation should be considered in the analysis of data to avoid misleading results.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/aje.2008.53.63
URL: https://scialert.net/abstract/?doi=aje.2008.53.63
INTRODUCTION
Perinatal mortality is a fundamental area in epidemiological studies and related fields (Congdon, 1998; McClure et al., 2007), because perinatal mortality has been used for international comparisons as an indicator of national health and social development (Rankin et al., 2005; Glinianaia et al., 2005; Sameshima and Ikenoue, 2007; Uddian and Hossain, 2008). The rate of infant mortality decrease in the world, because the United Nations and other organizations have been actively involved to reducing infant mortality in the world (Nault, 1997; Uddian and Hossain, 2008). However perinatal mortality has not followed the same pattern, especially in developing countries and continues to present a huge burden (Kusiako et al., 2000). So that 98% of perinatal mortality occurring in developing countries (Kusiako et al., 2000; Paul and Singh, 2004). Mother`s age, complication during labour and pregnancy, low birthweight, short gestational age and supervision during delivery are the common perinatal mortality risk factors in developing countries (Shah et al., 2000; Kusiako et al., 2000; Broek et al., 2003; Paul and Singh, 2004; Uddian and Hossain, 2008).
The interval between delivery and week 4 after birth is divided into three subintervals, allowing for the possibility of death during delivery (fresh stillbirth), in the first week after birth (early neonatal deaths), or between the first and fourth weeks after birth (late neonatal deaths). It is called these stages 1, 2 and 3 respectively. Stillbirth and early neonatal deaths taken together form a group called perinatal death (Macfarlane and Mugford, 1984). Some of relative perinatal mortality risk factors have been investigated in the last two decades e.g. birthweight, baby`s gender, pregnancy problems, mother`s age, maternal height and weight, gestation, delivery induction and supervision during delivery.
Both the birthweight and gestational age indicate the progress of pregnancy (Hack and Fanaroff, 2000). In fact, birthweight is generally considered one of the best indicators of a newborn`s chances of survival (Nault, 1997) and major neonatal morbidity increases with decreasing gestational age and birthwight (Hack and Fanaroff, 2000; Glinianaia et al., 2005). So that low birthwight babies (birthwight less than 2,500 grams) are more prone to dying than other babies (Meis et al., 1997; Burt and Pai, 2001; Shah et al., 2000; Frcog et al., 2001). Conley et al. (2006) and Shah et al. (2000) reported that small babies are exposed to the risk of neonatal death. Gestation is another fundamental measure of the progress of pregnancy. Bacak et al. (2005) reported that small gestational age increase the risk of neonatal mortality. Some studies identified that the risk profile of gestation is J-shaped: pre-term deliveries being at greatest risk, term deliveries at least risk and post-term deliveries at a slightly elevated risk (Frcog et al., 2001; MacDorman et al., 2007). Moreover, some authors suggest to use the ratio of birthweight to gestation (BGA) in order to show the effect of the combination of the two above risk factors, birthweight and gestation, in perinatal mortality (Magowan et al., 1998).
One of the possible risk factors in perinatal mortality is the gender of the baby. Some authors have shown that the male mortality rate is observed to be higher (Macfarlane and Mugford, 1984; Nault, 1997; Bacak et al., 2005).
Maternal age also is a risk factor associated with perinatal mortality (Bacak et al., 2005; Glinianaia et al., 2005; Dodds et al., 2006; Reddy et al., 2006; Smith and Fretts, 2007). Luke and Brown (2007) reported that older maternal age is associated with increasing risks for many pregnancy complication and infant mortality. However, Haldre et al. (2007) reported that despite major socio-economic changes resulting in improvements in obstetric care and growth in income, teenagers remained a higher risk group. But, some studies identified that these is J-shaped relationship between maternal age and perinatal mortality (Macfarlane et al., 1995; Shah et al., 2000; Bateman and Simpson, 2006; MacDorman et al., 2007; Uddian and Hossain, 2008).
Several studies showed that abnormality during pregnancy is a feature of perinatal mortality (Kusiako et al., 2000; Sheiner et al., 2004; Khong, 2006; Sameshima and Ikenoue, 2007; Smith and Fretts, 2007). The reason behind this may be that the mothers whose had some problems during the current pregnancy have babies that may have a reduced chance of survival.
Mother`s weight is an objective indicator of her level of heath. Kusiako et al. (2000) reported that thinness of the mother increase the risk of perinatal mortality. Many researchers investigated the relationship between mother`s weight and birthweight (Skjaerven et al., 1997; Johansson et al., 2007; Smith and Fretts, 2007). Skjaerven et al. (1997) reported that BMW (birthweight/mother`s weight) is a risk factor negatively associated with neonatal mortality.
If the health of the mother or baby would be endangered by allowing the pregnancy to continue, the labour will be induced by using artificial means (Kaplan et al., 1995; Almstrom et al., 1995; Ohel et al., 1996; Rand et al., 2000; James et al., 2001; Chanrachakul and Herabutya, 2002). Ramos et al. (2003) reported that women whose labour was induced experienced a lower perinatal mortality rate. However, this difference was not statistically significant. However, Alexander et al. (2000) reported that labor complications, including oxytocin induction, increase significantly from 40 to 42 weeks.
The risk factor foetal distress is defined as physical stress experienced by a foetus during labour as a result of not receiving enough oxygen. The most stressful period of labour for a baby is during a contraction, when the uterus tightens and thus reduces the baby`s supply of oxygen from the placenta. Fetal distress also increases the risk of cesarean section (Ramos et al., 2003).
Inhalational analgesia is one of possible risk factor for early neonatal deaths. Assessment of the effect of drugs on perinatal mortality is very difficult; patients with known intrauterine deaths or abnormal infants are usually given more potent analgesics; patients in long or abnormal labour will also be given the stronger agents. Butler and Bonham (1963) reported that the parts of U.K. with the lowest perinatal mortality, the Eastern and Southern countries (National Child Development Survey (NCDS)), have the greatest utilisation of volatile analgesics.
Delivery supervision is another risk factory for perinatal mortality. It is expected that trained supervision persons at delivery reduce the chance of perinatal mortality (Andersson et al., 2000; Broek et al., 2003).
Although magnitude and independence of some of these risk factors have been challenged, but it is considered the most of these risk factors in our analysis.
MATERIALS AND METHODS
Longitudinal analysis of perinatal mortality raises the issues: non-stationarity, duration dependency and unobserved explanatory variables which introduce correlation over time within the sequence of observed measurements on individuals in the survey (Xue and Brookmeyer, 1997; Scheike and Jensen, 1997; Zadkarami, 2000).
In general, individuals have propensities for life which commonly vary from stage to stage, because of individual characteristics and unobserved genetic factors lying behind the life process. Unobserved factors (random effect or frailty in the biomedical literature and residual heterogeneity in the sociological literature.) that have not been explicitly included as independent variables in the model create inter-stage correlation which affects the covariate parameter estimates in the various stages. In biostatistics, where the subjects of concern are often humans or animals, there is often important biological variation between subjects. It is important to include this heterogeneity in the statistical model to avoid biases (Song et al., 2004; Ananth et al., 2005). Taking account of heterogeneity or frailty between the individuals in population-based mortality studies is an important issue and a variety of models for heterogeneity have been proposed Xue and Brookmeyer (1997), Congdon (1998), Gao (2004) and Ananth et al. (2005) but the original use of the term frailty in a mortality context is due to Vaupel et al. (1979). The main problem with their models is that they did not allow for time-variant heterogeneity. If the possibility of time-variation is accepted, treating heterogeneity as time-invariant makes for misleading inference. In order to consider the changing circumstances of the babies in the various stages of perinatal mortality, The time-variant random effect model is used to determine the inter-stage correlation. However, in perinatal mortality contexts, heterogeneity between individuals has usually been ignored (Thomas et al., 1991; Middle and Macfarlane, 1995; Faden et al., 1997; Meis et al., 1997; Magowan et al., 1998; Frcog et al., 2001; Sameshinima and Ikenoue, 2007; Johansson et al., 2007; Uddian and Hossain, 2008).
The 1958 British national cohort study (National Child Development Survey, NCDS) was collected on babies born in one week (3-9 march 1958) in England, Wales and Scotland. Data were collected through birth records and face-to face interviews with parents at birth. The data was analyzed in various aspects (Wildschut et al., 1997; Spencer, 2006). It is managed to use 14,222 individuals, whom delivery naturally, by handling the missing values on 40 variables associated with perinatal mortality (Zadkarami, 2000).
Statistical Analysis: Analysis of data is carried out in two ways: Conventional analysis and Random effect analysis.
In conventional analysis, the individual observations are assumed independent over time and the data analyzed cross-sectionally. By using the GLIM package (Aitkin et al., 2005) a series of models are fitted to find an optimal set of maternal risk factors in the various stages. The results are obtained by using the logistic regression. The logistic regression is popular in the analysis of perinatal mortality (Meis et al., 1997; Congdon, 1998; Kusiako et al., 2000; Shah et al., 2000; Sheiner et al., 2004; Richter et al., 2007; Uddian and Hossain, 2008).
The analysis of the data can be carried out in several steps. At each stage, consider the relevant maternal risk factors that are significant on their own. In general, the variables are divided into two groups, continuous and categorical. Various models are fitted to find the best polynomial sequence for each continuous explanatory variable. The variable is fitted not only linearly, but also with quadratic, cubic and quartic terms. The levels of the categorical variables are collapsed for which these levels have unduly low frequencies (less than 10) to reduce the problem of IMLE (infinite maximum likelihood estimate) (Diamond et al., 1986) for categorical variables at a significance level (α = 0.05). The final set of the relevant risk factors is found by the forward stepwise procedure with the logistic regression (α = 0.01).
| Table 1: | Deviance of the model fitting to stages 1, 2 and 3 |
 | |
| **: p < 0.01 | |
This level was selected rather than the traditional level (α = 0.05) because the stepwise procedure selects the categorical variables for which most of the categorical levels are significant at level 0.05 (Zadkarami, 2000).
The findings indicated that the relevant maternal risk factors in the various perinatal mortality stages are different and the circumstances of individuals change substantially from stage to stage. For this reason, the logistic-normal random effect is used to investigate the general pattern of correlation between stages to find out the true correlation structure between successive stages. Therefore the random effect has multivariate normal with mean zero and positive definite covariance matrix. The logistic-normal random effect is popular for analyzing the correlated binary responses (Shih and Albert, 1999; Congdon, 1998; Sashegyi et al., 2001).
Including the random effects in the model improves the deviance by 12.313 for a gain of 1 degrees of freedom (p < 0.001) as reported in Table 1.
The routine E04UCF have been used to maximize the log likelihood function and estimate the covariate parameters. This is a library routine that was designed to minimize (maximize) a function subject to constraints using a sequential quadratic programming method and modified Newton-Raphson procedure as described in the NAG (1993) Library, mark 16. There is no need to supply derivatives, because derivatives are approximated by finite differences.
RESULTS AND DISCUSSION
The result of conventional and random effect models are presented respectively. However the results of random effect model are considered only in the discussion.
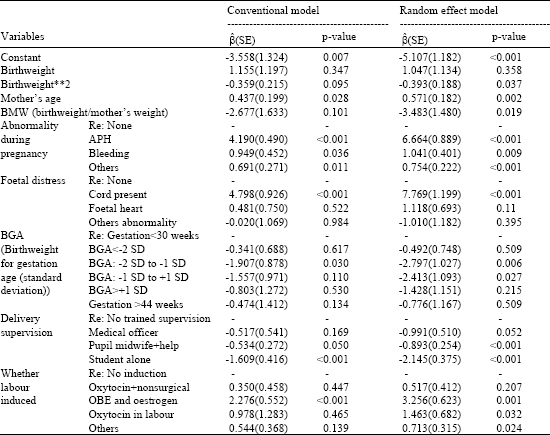
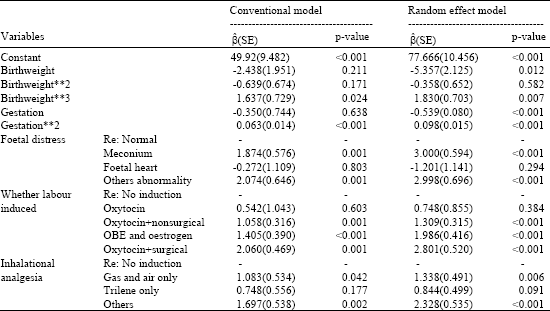
Conventional Analysis: Bleeding during pregnancy increases the risk of stillbirth 2.58 times. However stillbirth was found the highest (p < 0.001) among the babies whose mothers were involved in APH (accidental antepartum haemorrhage) during pregnancy (p < 0.001). The results also showed that the foetal distress (cord present) associated positively with the risk of fresh stillbirth (p < 0.001). Moreover, foetal distress (meconium and other abnormality) increase the risk of early neonatal deaths, 6.51 and 7.96 times, respectively (Table 2).
The mother`s age is another risk factor which is associated positively with fresh stillbirth so that older mothers are at risk of losing their babies during delivery (p < 0.05).
Labour induction by OBE (oxytocin but essential) and oestrogen was found 9.74 times increases the risk of fresh stillbirth as compared to mothers who delivered without induction. However, Labour induction by oxytocin+nonsurgical, OBE andoestrogen and oxytocin+surgical increase the risk of early neonatal deaths 2.88, 4.08 and 7.85 times, respectively.
The results in Table 2 and 3 showed that birthweight is not associated with fresh stillbirth. However birthweight is associated statistically with early neonatal deaths non-linearly (polynomial models). The results identify that BMW is not associated with fresh stillbirth. The findings indicated that babies with BGA (the ratio of birthweight to gestation) near average have more change (6.73 times) to survival at stage one as compared to babies with gestation less than 30 weeks.
| Table 2: | Result of fitting model at stage 1, stillbirth |
 | |
| APH: Accidental antepartum haemorrhage; OBE: Oxytocin but essential | |
The results also showed that trained supervision persons at delivery reduce the chance of fresh stillbirth (5 times).
The results showed that all levels of inhalational analgesia are associated positively with risk of early neonatal deaths. The variable gestation is another risk factor that is associated with early neonatal deaths non-linearly (polynomial).
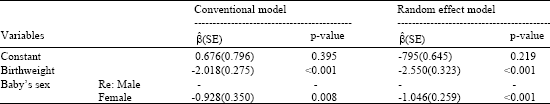
The results indicated that small babies are exposed to the risk of neonatal death statistically (p < 0.001). Furthermore, female babies have a greater chance of survival (2.85 times) at stage 3, late neonatal deaths, compared with male babies.
Random Effect Model: Allowing for the random effect in model changes the magnitude of the covariate estimates but the extent of the change is not the same for all, e.g., the estimate of birthweight ranges from 1.155 to 1.047, from -2.438 to -5.357 and -2.018 to -2.55 in stages one and two and three, respectively whereas that for (birthweight) ranges from -0.359 to -0.393 and from 0.639 to -0.358 in stages one and two, respectively. However, the estimate for foetal distress (foetal heart) changes from 0.481 to 1.118 and from -0.272 to -1.201 in stages one and two, respectively. The estimate for mother`s age ranges from 0.437 to 0.571 whereas that for BMW ranges from -2.677 to -3.483 in stage one as reported in Table 2-4.
Additionally, taking into consideration the correlation in the model may affect the significance levels of some covariate estimates. The results indicated that variables (birthweight)2, BMW, whether labour induced (oxytocin in labour and others) and (BGA: -1 SD to +1 SD) in stage one and variable birthweight in stage two are significant (p < 0.05) in the random effect model but there are not significant in the conventional model.
| Table 3: | Results of fitting model at stage 2, early neonatal deaths |
 | |
| Table 4: | Result of fitting model at stage 3, late neonatal deaths |
 | |
Furthermore, variables mother`s age, (BGA: -2 SD to -1 SD) in stage one and variables (birthweight)3 and inhalational analgesia (gas and air only) are significant at level, p < 0.01, in the random effect model, but those variables are significant at level, p < 0.05, in the conventional model. In general, the magnitude of association of the relative risk factors in stages 1, 2 and 3 are different in the two models, random effect and conventional. Ignoring the correlation across various stages of perinatal mortality (cross-sectional or conventional analysis) resulted in misleading statistical inference. Therefore this correlation should be considered in the analysis of data to avoid misleading results.
In the random effect model, variables BMW, BGA and delivery supervision are negatively associated with risk of fresh stillbirth. However, variables mother`s age, foetal distress, abnormality during pregnancy and whether labour induced are positively associated with risk of fresh stillbirth. But birthweight is associated with risk of fresh stillbirth non-linearly (quadratic).
The results indicated that variables foetal distress, inhalational analgesia and whether labour induced are positively associated with risk of early neonatal deaths. However, variables birthweight and gestation are associated with risk of early neonatal death non-linearly (quadratic and cubic respectively) in the random effect model. But birthweight and baby`s sex are the only variables that are negatively associated with late neonatal death. More investigations of the perinatal mortality risk factors are presented in discussion section.
The object of this study was, firstly, to investigate the relevant risk factors associated with various stages of perinatal mortality, secondly, to identify the general correlation pattern between various stages of perinatal mortality and finally, to verify the effect of this correlation on the relevant risk factors association. Random effect is one of popular methods to handling correlation in the correlated binary responses (Xue and Brookmeyer, 1997; Gao, 2004; Farrell and Sutradhar, 2006). The results identify that there is different correlation pattern between perninatal mortality stages and allowing for the random effect changes the magnitude of the covariate estimates. Now, the discussion is focused on the investigation of the risk factors association with perinatal mortality in the random effect model. The present study has been showed that the variables birthweight and gestation are associated with fresh stillbirth and early neonatal deaths non-linearly (polynomial models) but only birthweight is associated negatively with late neonatal deaths (p < 0.001). The importance of birthweight and gestation for the foetal outcome has been known for many years (Hack and Fanaroff, 2000; Glinianaia et al., 2005; Conley et al., 2006) and some studies have addressed different relationship between birthweight and gestation, with perinatal mortality (Nault, 1997; Frcog et al., 2001). Some studies have identified that small gestational age increase the risk of neonatal mortality statistically (Bacak et al., 2005) and the low birthwight babies group is one of high perinatal mortality risk group of babies (Meis et al., 1997; Burt and Pai, 2001; Frcog et al., 2001). The finding results also showed that babies with BGA near average have more change to survival at stage one, fresh stillbirth (p < 0.05). The association between smallness for gestational age (SGA) and perinatal morbidity were investigated and SGA babies is one of the high risk of perinatal mortality group (Kady and Gardosi, 2004; Figueras et al., 2007).
Many researchers have been investigated the relationship between mother`s weight and birthweight (Skjaerven et al., 1997; Johansson et al., 2007). Skjaerven et al. (1997) reported that BMW is a risk factor negatively associated with neonatal mortality. However, finding of this study indicate that the variable BMW is negatively associated with fresh stillbirth (p < 0.05).
The result in Table 4 indicated that female babies have a greater (2.85 times) chance of survival compared to male babies at stage 3, late neonatal. Some studies have shown that the male neonatal mortality rate is observed to be higher (Nault, 1997; Bacak et al., 2005).
The positive significant linear relationship between stillbirth and maternal age was found (p < 0.01). Some studies identified similar results (Bacak et al., 2005; Glinianaia et al., 2005; Dodds et al., 2006; Reddy et al., 2006; Luke and Brown, 2007). However, some authors have shown J-shaped relationship between maternal age and perinatal mortality (Macfarlane et al., 1995; Uddian and Hossain, 2008).
The findings indicated that the abnormality during pregnancy increased the risk of fresh stillbirth (p < 0.01). Many studies identified the positive association between the abnormality during pregnancy and perinatal mortality (Sheiner et al., 2004; Khong, 2006; Sameshima and Ikenoue, 2007).
The results in Table 2 and 3 confirmed that the variable foetal distress (cord present and meconium) also increases the risk of fresh stillbirth and early neonatal deaths, respectively. Ramos et al. (2003) reported that fetal distress also increases the risk of cesarean section.
Systematic reviews have been indicated that the labour will be induced by using artificial means if the health of the mother or baby would be endangered by allowing the pregnancy to continue (Kaplan et al., 1995; Almstrom et al., 1995; Ohel et al., 1996; James et al., 2001; Chanrachakul and Herabutya, 2002). The finding of the study also showed that labour is induced by OBE and oestrogen and oxytocin+surgical increases the risk of fresh stillbirth at least 4.32 times. However, the risk of early neonatal deaths increases 3.7, 7.29 and 16.46 times for babies whose mothers induced by oxytocin+nonsurgical, OBE and oestrogen and oxytocin+surgical, respectively. Ramos et al. (2003) reported that women whose labour was induced experienced a lower perinatal mortality rate. However, this difference was not statistically significant.
It is observed that the variable inhalational analgesia is associated positively with risk of early neonatal deaths. Butler and Bonham (1963) reported that some areas in UK with the lowest perinatal mortality, the Eastern and Southern countries (in NCDS data set), have the greatest utilisation of volatile analgesics.
The results of this study identified that trained supervision persons at delivery reduce the chance of fresh stillbirth. Similar results are reported by Andersson et al. (2000).
CONCLUSIONS
The results of study identified that there are different correlation patterns between the perinatal mortality stages, as displayed in Table 1 and allowing for the random effect changes the magnitude of the covariate estimates but the extent of the change is not the same for all. Such that some relevant variables that are not significant in the conventional model become significant in random effect model and versa as. Wrong correlation assumption would have led to misleading results.
The results indicated that the variables BMW, BGA and delivery supervision are negatively associated with risk of fresh stillbirth. However, variables mother`s age, foetal distress, abnormality during pregnancy and whether labour induced are positively associated with risk of fresh stillbirth. But variable birthweight is associated with risk of fresh stillbirth non-linearly (quadratic).
The results in Table 2-4 confirmed that variables foetal distress, inhalational analgesia and whether labour induced are positively associated with risk of early neonatal deaths. However, variables birthweight and gestation are associated with risk of early neonatal death non-linearly, quadratic and cubic respectively. But birthweight and baby`s sex, female babies are the only variables that are negatively associated with late neonatal death.
ACKNOWLEDGMENT
The author would like to thanks the referees for their very helpful comments.
REFERENCES
- Alexander, J.M., D.D. Mcintire and K.J. Leveno, 2000. Forty weeks and beyond: pregnancy outcomes by week of gestation. Obstet. Gynecol., 96: 291-294.
PubMedDirect Link - Almstrom, H., L. Granstrom and G. Ekman, 1995. Serial antenatal monitoring compared with labor induction in post-term pregnancies. Acta Obstet. Gynecol Scand., 74: 599-603.
PubMedDirect Link - Ananth, C.A., R.W. Platt and D.A. Savitz, 2005. Regression models for clustered binary responses: Implications of ignoring the intracluster correlation in an analysis of perinatal mortality in twin gestations. Ann. Epid., 15: 293-301.
Direct Link - Andersson, T., U. Hogberg and S. Bergstrom, 2000. Community-based prevention of perinatal deaths: Lessons from nineteenth-century Sweden. Int. J. Epid., 29: 542-548.
Direct Link - Bacak, S.J., K. Baptiste-Robrty, E. Amon, B. Ireland and T. Leet, 2005. Risk factors for neonatal mortality among extremely low birth-weight infants. Am. J. Obstet. Gynecol., 192: 862-867.
CrossRefDirect Link - Bateman, B.T. and L.L. Simpson, 2006. Higher rate of stillbirth at the extremes of reproductive age: A large nationwide sample of deliveries in the United States. Am. J. Obstet. Gynecol., 194: 840-845.
Direct Link - Van Der Broek, N.R., S.A. White, C. Ntonya, M. Ngwale, T.R. Cullinan, M.E. Molyneux and J.P. Neilson, 2003. Reproductive health in rural Malawi: A population-based survey. Br. J. Obstet. Gynecol., 110: 902-908.
CrossRefPubMedDirect Link - Burt, B.A. and S. Pai, 2001. Does low birthweight increase the risk of caries? A systematic review. J. Den. Edu., 65: 1024-1027.
Direct Link - Chanrachakul, B. and Y. Herabutya, 2002. Postterm with favorable cervix: Is induction necessary? Eur. J. Obstet. Gynecol. Reprod., 106: 154-157.
Direct Link - Congdon, P., 1998. A multilevel model for infant health outcomes: Maternal risk factors and geographic variation. J. Royal Stat. Soc.: Series D, 47: 159-182.
CrossRefDirect Link - Conley, D., K.W. Strully and N.B. Bennett, 2006. Twin differences in birth weight: The effects of genotype and prenatal environment on neonatal and post neonatal mortality. Econ. Hum. Biol., 4: 151-183.
Direct Link - Diamond, I., J.W. McDonald and I.H. Shah, 1986. Proportional hazards models for current status data: Application to the study of differentials in age at weaning in Pakistan. Demo, 23: 607-620.
PubMedDirect Link - Dodds, L., N.D. King, D.B. Fell, B.A. Armson, A. Allen and C. Nimrod, 2006. Stillbirth risk factors according to timing of exposure. Ann. Epid., 16: 607-613.
CrossRefPubMedDirect Link - Faden, V.B., B.I. Graubard and M. Dufour, 1997. The relationship of drinking and birth outcome in a US national sample of expectant mothers. Paediat. Perinat. Epid., 11: 167-180.
CrossRefPubMedDirect Link - Figueras, F., J. Figueras, E. Meler, E. Eixarch, O. Coll, E. Gratacos, J. Gardosi and X. Carbonell, 2007. Customised birthweight standards accurately predict perinatal mortality. Arc. Dis. Child.-Fetal Neon. Edit., 92: 277-280.
Direct Link - Frcog, T.C., G.P. Frcog and J.C. Mfphm, 2001. The risk of neonatal death in relation to birth weight and maternal hypertensive disease in infants born at 24-32 weeks. Eur. J. Obstet. Rep. Biol., 95: 114-118.
Direct Link - Gao, S., 2004. A shared random effect parameter approach for longitudinal dementia data with non-ignorable missing data. Stat. Med., 23: 211-219.
PubMedDirect Link - Glinianaia, S.V., J. Rankin, R. Bell, M.S. Pearce and L. Parker, 2005. Temporal changes in the distribution of population risk factors attenuate the reduction in perinatal mortality. J. Clin. Epid., 58: 1299-1307.
CrossRefPubMedDirect Link - Hack, M. and A.A. Fanaroff, 2000. Outcomes of children of extremely low birthweight and gestational age in the 1990s. Sem. Neonat., 5: 89-106.
Direct Link - Haldre, K., K. Rahu, H. Karro and M. Rahu, 2007. Is a poor pregnancy outcome related to young maternal age? A study of teenagers in Estonia during the period of major socio-economic changes (from 1992 to 2002). Eur. J. Obstet. Gynecol. Reprod. Biol., 131: 45-51.
Direct Link - James, C., S.S. George, N. Gaunekar and L. Seahadri, 2001. Management of prolonged pregnancy: A randomized trial of induction of labour and antepartum foetal monitoring. Natl. Med. J. India, 14: 270-273.
Direct Link - Johansson, K., Y. Linne, S. Rossner and M. Neovius, 2007. Maternal predictors of birthweight: The importance of weight gain during pregnancy. Obstet. Res. Clin. Prac., 1: 243-252.
CrossRefDirect Link - Kady, S.M. and J. Gardosi, 2004. Perinatal mortality and fetal growth restriction. Best Pract. Res. Clin. Obstet. Gynecol., 18: 397-410.
CrossRefDirect Link - Kaplan, B., G. Goldman, Y. Peled, R. Hecht-Resnick, A. Neri and J. Ovadia, 1995. The outcome of post-term pregnancy. A comparative study. J. Perinat. Med., 23: 183-189.
CrossRefPubMedDirect Link - Khong, T.Y., 2006. The placenta in stillbirth. Curr. Diagn. Pathol., 12: 161-172.
CrossRefDirect Link - Kusiako, T., C. Ronsmans and L.V.D. Paal, 2000. Perinatal morality attributable to complications of childbirth in Matlab, Bangladesh. Bul. Wor. Heal. Organ., 78: 621-627.
Direct Link - Luke, B. and M.B. Brown, 2007. Contemporary risk of maternal morbidity and adverse outcomes with increasing maternal age and plurality. Fert. Ster., 88: 283-293.
Direct Link - Magowan, B.A., M. Bain, E. Juszczak and K. McInneny, 1998. Neonatal mortality amongst Scottish pre-term singleton births (1985-1994). Br. J. Obstet. Gynecol., 105: 1005-1010.
PubMedDirect Link - McClure, E.M., R.L. Goldenberg and C.M. Bann, 2007. Maternal mortality, stillbirth and measures of obstetric care in developing and developed countries. Int. J. Gynecol. Obstet., 96: 139-146.
Direct Link - Meis, P.J., R. Michielutte, T.J. Peters, H.B. Wells, R.E. Sands, E.C. Coles and K.A. John, 1997. Factors associated with term low birthweight in Cardiff Wales. Paediat. Perinat. Epid., 11: 287-297.
CrossRefPubMedDirect Link - Middle, C. and A. Macfarlane, 1995. Labour and delivery of normal primiparous women: Analysis of routinely collected data. Br. J. Obstet. Gynecol., 102: 970-977.
CrossRefPubMedDirect Link - Paul, V.K. and M. Singh, 2004. Regionalized perinatal care in developing countries. Semi. Neonat., 9: 117-124.
CrossRefPubMedDirect Link - Ramos, L.S., F. Olivier, I. Delke and A.M. Kaunitz, 2003. Labor induction versus expectant management for postterm pregnancies: A systematic review with meat-analysis. Obstet. Gynecol., 101: 1312-1318.
Direct Link - Rand, L., J.N. Robinson K.E. Economy and E.R. Norwitz, 2000. Post-term induction of labor revisited. Obstet. Gynecol., 96: 779-783.
CrossRefPubMedDirect Link - Rankin, J., M.S. Pearce, R. Bell, S.V. Glinianaia and L. Parker, 2005. Perinatal mortality rates: Adjusting for risk factor profile is essential. Paedia. Peri. Epid., 19: 56-58.
Direct Link - Reddy, U.M., C.W. Ko and M. Willinger, 2006. Maternal age and the risk of stillbirth throughout pregnancy in the United States. Am. J. Obstet. Gynecol., 195: 764-770.
Direct Link - Sameshima, H. and T. Ikenoue, 2007. Risk factors for perinatal deaths in Southern Japan: Population-based analysis from 1998 to 2005. Early Hum. Devel., 83: 319-323.
CrossRef - Sashegyi, A.I., K.S. Brown and P.J. Farrell, 2001. On the correspondence between population-averaged models and a class of cluster-specific models for correlated binary data. Stat. Prob. Lett., 52: 135-144.
Direct Link - Scheike, T.H. and T.K. Jensen, 1997. A discrete survival model with random effects: An application to time to pregnancy. Biometerics, 53: 318-329.
PubMedDirect Link - Shah, N.M., M.A. Shah, A.A. Khalaf, M.M. Mustafa and A. Al-Sayed, 2000. Searching for socioeconomic risk factors in perinatal mortality in Kuwait: A case control study. Soc. Sci. Med., 51: 539-550.
Direct Link - Sheiner, E., A. Levy and M. Mazor, 2004. Precipitate labor: Higher rates of maternal complications. Eur. J. Obstet. Gynecol. Repod. Biol., 116: 43-47.
CrossRefDirect Link - Shih, J.H. and P.S. Albert, 1999. Latent model for correlated binary data with diagnostic error. Biometerics, 55: 1232-1235.
CrossRefPubMedDirect Link - Skjaerven, R., A.J. Wilcox, N. Qyen and P. Magnus, 1997. Mothers' birthweight and survival of their offspring: Population based study. Br. Med. J., 314: 1376-1380.
PubMedDirect Link - Song, J.X., J.T. Wassell and A. Kapadia, 2004. Relative mortality for correlated lifetime data. Comput. Stat. Data Anal., 45: 849-864.
Direct Link - Thomas, P., J. Golding and T.J. Peters, 1991. Delayed antenatal care: Does it effect pregnancy outcome? Soc. Sci. Med., 32: 715-723.
CrossRefPubMedDirect Link - Uddian, M.J. and M.Z. Hossain, 2008. Predictors of infant mortality in a developing country. Asian J. Epidemiol., 1: 1-16.
CrossRefDirect Link - Vaupel, J.W., K.G. Manton and E. Stallard, 1979. The impact of heterogeneity in individual frailty on the dynamics of mortality. Demography, 16: 439-454.
CrossRefPubMedDirect Link - Xue, X. and R. Brookmeyer, 1997. Regression analysis of discrete time survival data under heterogeneity. Stat. Med., 16: 1983-1993.
PubMedDirect Link








