Research Article
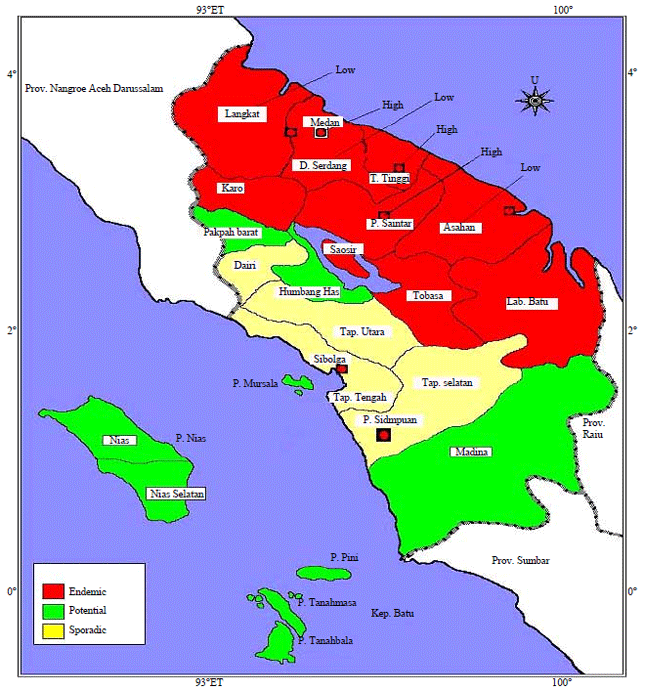
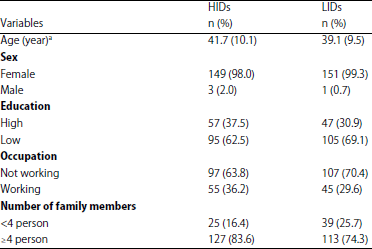
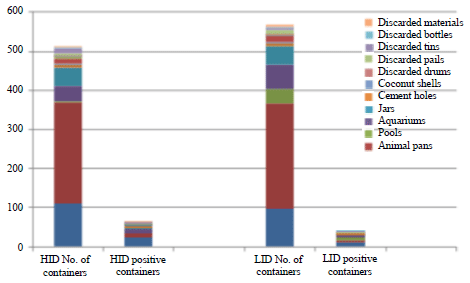
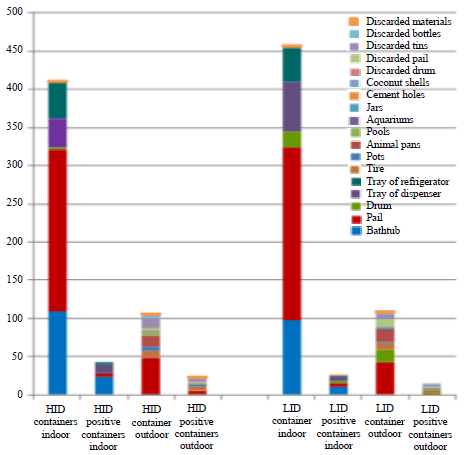
Key Breeding Place for Dengue Vectors and the Impact of Larvae Density on Dengue Transmission in North Sumatera Province, Indonesia
Faculty of Public Health, University of Sumatera Utara, 20155 Medan, North Sumatera, Indonesia
LiveDNA: 62.12515
Tri Makmur
Faculty of Medicine, Islamic University of Sumatera Utara, Medan, North Sumatera, Indonesia
LiveDNA: 62.12519
Nurul Huda
Food Technology Program, School of Industrial Technology, Universiti Sains Malaysia, 11800 Penang, Malaysia
LiveDNA: 62.12518