ABSTRACT
Research studies were conducted in the green house to investigate the nature of pathogenicity of Alternaria solani, 5 isolates on two tomato species Lycopersicon esculentum and L. esculentum x L. hirsutum F1 hybrids and to describe the procedures by which it would be possible to determine the 5 isolates of Alternaria pathogenic action and progressive disease development. The results showed that the five isolates of Alternaria solani were non-pathogenic against the two tomato species tested in the experiment.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2002.703.704
URL: https://scialert.net/abstract/?doi=ajps.2002.703.704
INTRODUCTION
Early blight of tomato is a fungal disease caused by Alternaria solani (Ellis & Martin) Jones and grout (Maiero et al., 1989). It is a three-phase disease, which can produce leaf spots, stem canker and fruit rot. But the foliar phase is the most common and destructive part of the disease. The disease is characterized by dark colored leaf spots that are necrotic in the center and have concentric rings pattern seemingly associated with the periodic development of the fungus. When lesions expand and become more numerous, leaves are blighted and plants are prematurely defoliated.
Hence a number of cultivars with this moderate but useful degree of resistance have been released (Barksdale and Stoner, 1977; Gardner, 1988; Nash and Gardner, 1988a; 1988b). In developing a screening method for tomato early blight, considerable difficulty has been experienced in getting Alternaria solani to sporulate profusely in culture. In the past, several researchers used mycelial fragments as inoculation in their intensive screening and breeding methods. A few others relied on natural infection (Bashi and Rotem, 1974; Douglas, 1972). On the other hand, some researchers have been able to obtain sporulation in the lab (Barksdale, 1969; Maiero et al., 1989; Nash, 1988; Nash and Gardner, 1988). Maiero et al. (1989), grown twelve isolates on Lima bean agar for 6 days at 22 oC under normal diurnal light conditions. Aerial mycelium was scraped and the cultures were uncovered inverted and placed in diurnal light at ambient room temperature for 24 hour to induce sporulation-spores from all twelve isolates were mixed with distilled water to produce a spore suspension of 5000 spares/ml. McCallan and Chan (Barksdale, 1969) induced sporulation by growing cultures on PDA, scraping them and then putting them in a moist chamber with the lid on a jar and then placing them in a window sunlight. Barksdale (1969) induced sporulation by growing cultures on Lima bean agar (LBA) at about 23 oC in plastic petri dishes. Nash (1988) induced sporulation on LBA at about 23 oC. The aerial mycelia were flattened with a spatula and uncovered plates were inverted and exposed to eight hours of light at 23 oC, then twelve hours of dark at 19 oC. Conidia were harvested after twenty-eight hours by flooding cultures with d H20 and rubbing the surface lightly with a spatula.
Maiero et al. (1989), grown twelve isolates on Lima bean agar for 6 days at 22 oC under normal diurnal light conditions. Aerial mycelium was scraped and the cultures were uncovered inverted and placed in diurnal light at ambient room temperature for 24 hours to induce sporulation-spores from all twelve isolates were mixed with distilled water to produce a spore suspension of 5000 spores/ml.
The objectives of the study were to produce fungal spore inoculum for each five isolate in the laboratory and to study those conditions that would favor rapid systematic disease development on tomato seedlings in vitro.
MATERIALS AND METHODS
Plant material: Two tomato species, L. esculentum and L. esculentum x L. hirustum F1 hybrid sixty seedlings were produced in individual containers in the greenhouse. After thirty days the disease-free seedlings were transferred into 6 inches pots. The plants were watered as required. No fungicide was applied in any stage.
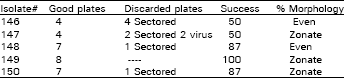
Inoculum preparation and inoculation: Five isolations of Alternaria solani were collected from tomatoes blighted early in the field in the Lexington area. These isolates were grown on reconstituted lima bean agar (LBA) at about 23 oC in 9 cm disposable plastic petri dishes. This procedure was similar to Nash and Gardner (1988) as mentioned previously, but with some modifications-one week old cultures were handled in the following pattern (Table 1). The isolate# written on back of plate lid was discarded and mycelium flattened with flamed spatula, plates inoculated were inverted on tray for ten hours of light at 23 oC then fourteen hours of dark at 19 oC. The culture dish lids were removed but one plate of each isolate was not uncovered and then aerial mycelium was flattened with a spatula the cultures were inverted on racks one centimeter above a tray surface for slow drying in an incubator for ten hours at 23 oC, then fourteen hours of dark at 19 oC. The inverted plates were then covered with plastic and uncovered plates were also covered. It was observed that uncovered plates became dried under the above diurnal temperature.
| Table 1: | Uncovering of culture |
 | |
After thirty hours, the flattened cultures were flooded with d H20 and the surface rubbed lightly with a spatula. The spore suspension for each isolate from different plates was mixed together. The conidia suspension was then filtered through four layers of cheesecloth. Spore concentration was estimated with the aid of a haematocytometer to be 10,000 spores per milliliter of distilled water. This spore suspension contained about equal numbers of spores from each of the five isolates mentioned above. Inoculation in the greenhouse was accomplished with a hand sprayer, by atomizing the spore suspension on leaves up to the level that suspension water ran off the leaves. Two plants of each tomato variety in every block were sprayed only by clean water as a control. All plants were then covered with plastic bags and bind with rubber bands to provide a moist chamber. The plastic covers were removed after fifteen hours.
Greenhouse test for pathogenicity: Thirty plants of each accession were selected and grown as mentioned previously. The plants were placed in the greenhouse in five blocks, six plants of each variety in one block. When plants became six weeks old, then inoculation was accomplished by spraying three hundred milliliters of spore suspension (diluted) of five isolates per treatment or thirty milliliters per plant on ten plants. Two plants in each block were sprayed only with water as check. Following inoculation, plants were covered with plastic bags and closed with rubber bands for fifteen hours. The uncovered plants were then sprayed by water daily in the evening to encourage lesion development. The disease data on the percentage of necrotic leaf area and tolerance to defoliation were recorded with the following: disease scale parameters; No, very slight, moderate, severe, extreme and complete defoliation.
RESULTS AND DISCUSSION
All the plants leaves were observed routinely for Alternaria disease development. We found that Alternaria disease spots were present only on Lycopersion esculentum leaves but were not significant and therefore, no defoliation of plants occurred (Table 2). Water spray L. esculentum (check) plants have no spots on the leaves and L. esculentum x L. hirsutum F1 plant leaves also have no spots.
| Table 2: | Fungal inoculum and Lycopersicon species reaction |
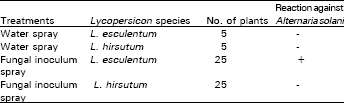 | |
| - : No spots | +: Spots |
Symptoms of early blight are often difficult to obtain on young seedlings. It was reported by Marisa et al. (1989) that early blight is associated with physiological maturity of the plant; older, senescing leaves are more susceptible then young immature leaves. It was found that an air temperature of 16 oC for two weeks following inoculation was more favorable for symptoms development (Pound, 1951). Moreover, as the susceptibility of tomato plants to infection by A. solani is determined by the age of the host (Rotem, 1994). It was reported by Barksdale (1969) that after one week of inoculation, the plants which received repeated moist periods, the lesions were about twice as large as those which received one initial incubation. It was found that the development of early blight symptoms on tomato plants is affected by inoculum concentration, leaf wetness duration, plant age and host susceptibility (Vloutoglou and Kalogerakis, 2000). In similar experiments Coffey and Marshall (1975), showed that early blight severity on young tomato plants increased from 5x103 to 8x104 conidia ml-1. A positive relationship between inoculum concentration and symptom development has also been demonstrated for other Alternaria species (Vloutoglou, 1994). Vloutoglou and Kalogerakis (2000) showed that tomato cultivars become increasingly susceptible to A. solani infection as plants aged. It was also found in earlier experiments by Moore (1942) that susceptibility of tomato plants to A. solani infection was age dependent the pathogen caused color rot in seedlings and early blight in mature plants, whereas middle aged plants were relatively tolerant. Moreover Voutolou and Kalogerakis (2000) found that percentages of leaf area affected by the pathogen and defoliation increased with increasing leaf wetness duration up to 24 hours. As it was observed from the experimental results that no significant disease spots were present on the leaves of the two tomato species and as a result no defoliation of inoculated plants occurred and these findings will partially conform the Coffey and Marshall (1975) and Vloutoglou and Kalogerkis (2000), who showed that increased conidial concentration increased early blight severity on young tomato plants. Thus we assume that it is either tolerance of the two tomato species, inoculum concentration ml-1 and/or the unattended chance factors might affected the controlled conditions in the greenhouse.
The results of the study showed that Alternaria disease screening for Lycopersicon species can be carried out in vitro by careful handling of the fungal isolates and hence forth inoculation of the plants for the early blight symptom development. Therefore, similar studies should be undertaken to develop best disease screening procedures and methods for different plants species in vitro.
REFERENCES
- Bashi, E. and J. Rotem, 1974. Adaptation of four pathogens to semi arid habitats as conditioned by penetration rate and germinating spore survival. Phytopathology, 64: 1035-1039.
CrossRefDirect Link - Coffey, M.D. and W.C. Marshall, 1975. The effect of early blight disease caused by Alternaria solani on shoot growth of young tomato plants. Ann. Applied Biol., 80: 17-26.
CrossRefDirect Link - Douglas, D.R. and J.J. Pavek, 1972. Screening potatoes for field resistance to early blight. Am. J. Potato Res., 49: 1-6.
CrossRefDirect Link - Gardner, R.G., 1989. Combining ability estimates for early blight resistance in tomato. J. Am. Soc. Hort. Sci., 114: 118-121.
Direct Link - Maiero, M., T.J. Ng and T.H. Barksdale, 1989. Combining ability estimates for early blight resistance in tomato. J. Am. Soc. Hort. Sci., 114: 118-121.
Direct Link - Maiero, M., T.J. Ng and T.H. Barksdale, 1990. Genetic resistance to early blight in tomato breeding lines. Hort Sci., 25: 344-346.
Direct Link - Rotem, J., 1994. The Genus Alternaria, Biology, Epidemiology and Pathogenicity. APS Press, USA., Pages: 326.
Direct Link - Vloutoglou, I., 1999. Evaluation of tomato cultivars and hybrids for resistance to Alternaria solani infection. Ann. Applied Biol., 134: 48-49.
Direct Link - Vloutoglou, I. and S.N. Kalogerakis, 2000. Effects of inoculum concentration, wetness duration and plant age on development of early blight (Alternaria solani) and on shedding of leaves in tomato plants. J. Plant Pathol., 49: 339-345.
CrossRefDirect Link








