Research Article
Influence of Nitrogen Sources on Growth, Hydrocarbon and Fatty Acid Production by Botryococcus braunii
Not Available
R. Sarada
Not Available
T.R. Shamala
Not Available
G.A. Ravishankar
Not Available
INTRODUCTION
Bioenergy resources have been receiving increasing attention due to their less polluting nature and are renewable as against the conventional fossil fuels, which are also leading to potential depletion. Botryococcus braunii is a green colonial, hydrocarbon rich microalga and is recognized as one of the renewable resource for the production of liquid hydrocarbons (Maxwell et al., 1968; Brown et al., 1969; Anirban et al., 2002). B. braunii is grouped into three different chemical races A, B and L depending on the type of hydrocarbons they synthesize. The race-A produces C23 to C33 odd numbered n-alkadienes, mono-, tri-, tetra-and pentaenes and they are derived from fatty acids and these linear olefins can constitute up to 61% of the dry cell mass of the green active state colonies. Race-L strains produces a single tetraterpene hydrocarbon called lycopadiene (C40H78) and it can constitute 2-8% of the dry biomass. Whereas B race produces polymethylated triterpenoid hydrocarbons called botryococcenes of C30-C37 of general formula CnH2n-10 as major hydrocarbons and small amounts of methyl-branched squalenes (Metzger et al., 1985, 1990; Metzger and Largeau, 1999, 2005; Achitouv et al., 2004). Hydrocarbon oils extracted from B. braunii produces a distillate comprising of 67% gasoline fraction, 15% aviation turbine fuel, 15% diesel fuel and 3% residual fuel after their hydrocracking (Hillen et al., 1982). The fuel fractions so obtained are free from oxides of sulphur and nitrogen (SOX and NOX) after their combustion. It being a photosynthetic organism, it can reduce CO2 emissions by 1.5x105 tons/year (Sawayama et al., 1999) and is also been reported (Sawayama et al., 1992) that B. braunii can grow in Secondary Treated Sewage (STS) by utilizing the inorganic salts like nitrogen and phosphorous sources of the STS. The uptake of some toxic metals like chrome, cadmium and arsenic is also been reported (Sawayama et al., 1995) and there by the alga Botryococcus braunii offers an eco friendly process and product.
Yang et al. (2004a, b) have studied the effects of sulfite and bisulfite on the growth and hydrocarbon production of B. braunii and similarly the effects of nitrate and nitrites. It is necessary to understand the effects of different cultural conditions on B. braunii for its biotechnological exploitation for hydrocarbon production. Therefore the present study focused on the influence of different cultivation parameters like, pH, different nitrogen sources and culture age on the growth and hydrocarbon production of B. braunii.
Algal strain: B. braunii (SAG 30.81) was obtained from Sammlung von AlgenKulturen, pflanzenphysiologisches Institut, Universitat Gottingen, Germany and B. braunii (LB 572) from University of Texas, USA. The stock cultures were maintained on modified Chu13 medium (Largeau et al., 1980) both in liquid and solid media at 25±1°C with 1.7647 erg m-2 s-1 light intensity and 16:8 h light dark cycle.
Influence of culture age: Experiment was conducted to study the effect of culture age on B. braunii growth and hydrocarbon production. Two weeks old cultures of B. braunii (LB 572 and SAG 30.81) were used as inoculum at 20% (v/v) and added to the Erlenmeyer flasks (150 mL) containing 40 mL of Chu 13 modified medium and incubated at 25±1°C with 1.7647 erg m-2 s-1 light intensity and 16:8 h light dark cycle. The culture flasks were harvested at regular intervals of 7 days (week) and biomass and hydrocarbon yields were analyzed. All the experiments were carried out in triplicates
Effect of pH: The experiment was carried out in Erlenmeyer flasks (150 mL) containing 40 mL of Chu 13 modified medium and the pH of the medium was adjusted to 6.0, 6.5, 7.0, 7.5, 8.0 and 8.5 before autoclaving. All the flasks were inoculated with uniform volume (25%V/V) of B. braunii (LB 572) and B. braunii (SAG 30.81) cultures which were grown for 2 weeks. The culture flasks were incubated for four weeks at 25±1°C with 1.7647 erg m-2 s-1 light intensity and 16:8 h light dark cycle and experiment was carried out in triplicates.
Effect of different nitrogen sources: The experiment was carried out in Erlenmeyer flasks (250 mL) containing 80 mL of Chu 13 modified medium (Largeau et al., 1980) with different nitrogen sources such as sodium nitrate, potassium nitrate, ammonium nitrate, calcium nitrate and urea in terms of nitrogen equivalent to potassium nitrate, which is a nitrogen source in modified Chu 13 medium, to study their effect on B. braunii (SAG 30.81 and LB 572) growth and hydrocarbon production. The culture flasks were incubated for 4 weeks at 25±1°C with 1.7647 erg m-2 s-1 light intensity and 16:8 h light dark cycle.
Biomass estimation: The cultures were harvested and the cells were washed with distilled water after centrifugation at 5000 rpm for 10 min. Then the pellet was freeze dried and weight of algal biomass was determined gravimetrically.
Hydrocarbon extraction: Hydrocarbon was extracted by homogenizing the dry biomass with n-hexane repeatedly and supernatants recovered after centrifugation (5000 rpm) were pooled and evaporated to dryness under nitrogen. Hydrocarbon content was measured gravimetrically and expressed as percentage dry weight (Sawayama et al., 1992).
Hydrocarbon analysis: Crude extracts were purified by column chromatography on silica gel with n-hexane as eluent. GC analysis was done on BP-5 capillary column as described by Dayananda et al. (2005). Hydrocarbons were grouped into three categories as less than C20, higher than C30 and in between C20 and C30 with reference to their elution with that of the retention times of the internal standards (triacontane and eicosane).
Fatty acid analysis: Lipids were extracted with chloroform- methanol (2: 1) and quantified gravimetrically. The fatty acid methyl esters (FAME) of the fatty acids were prepared (Christie, 1982). FAME were analyzed by GC-MS equipped with FID using SPB-1 capillary column with a temperature programming from 130 to 280°C at a rate of 2°C min-1 (Dayananda et al., 2006). The FAME were identified by comparing their fragmentation pattern with authentic standards (C12-C28) (Sigma) and also with NIST library.
Influence of culture age: A time course study was carried out on growth and hydrocarbon content in both the strains. The result shows a continuous but slow increase in growth of the organism during the eight-week study (Fig. 1). The growth of LB 572 was higher than SAG 30.81 since the biomass yields were found to be 1.2 and 0.56 g L-1, respectively. Hydrocarbon content in the range of 23-28 and 30-35% (w/w) was observed through out the experimental period for LB 572 and SAG 30.81, respectively. The culture pH were recorded in the range of 7.5 to 8.6 and 7.5 to 9.2 for LB 572 and SAG 30.81, respectively. Kojima and Zhang (1999) reported that maximum hydrocarbon productivity of the alga during exponential and early linear phases of growth. Frenz et al. (1989) reported 18 to 21% (w/w) of hydrocarbon production in B. braunii. It has been reported that in all the three chemical races of B. braunii, the hydrocarbon productivity was optimal during the exponential growth phase of the alga (Metzger et al., 1985, 1990; Largeau et al., 1980). Thus, the production of hydrocarbons appears to be a normal feature of B. braunii. In the present study, the results suggested that the organisms could be harvested when they attained their maximum growth since there is no significant difference in its hydrocarbon productivity during the experimental period.
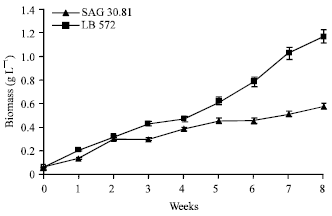 | |
| Fig. 1: | Growth curve of B. braunii (LB 572 and SAG 30.81) cultures grown over an eight weeks culture period |
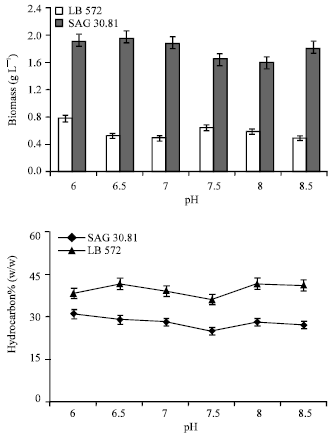 | |
| Fig. 2: | Biomass and hydrocarbon yield of B. braunii (LB 572 and SAG 30) cultures grown under the influence of different pH |
Effect of pH: The biomass yield of B. braunii (SAG 30.81 and LB 572) indicates (Fig. 2) that the organism can grow in the pH range of 6.0-8.5, although higher biomass, 0.77 and 1.9 g L-1 obtained, respectively for SAG 30.81 and LB 572 at pH 6.0. Irrespective of the initial pH of medium, the culture pH after its growth was found to be in the range of 8.0 to 8.8, this indicates the depletion of CO2 or HCO3 through photosynthesis.
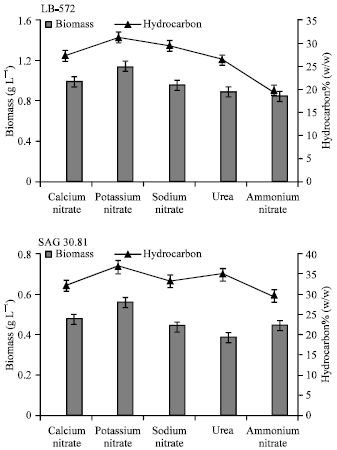 | |
| Fig. 3: | Biomass and hydrocarbon yields of B. braunii (LB 572 and SAG 30.81) grown in different nitrogen sources |
The hydrocarbon content in the organism was not much affected by the pH (Fig. 2), since hydrocarbon yields were in the range of 25-31% (w/w) and 38-41% (w/w), respectively for SAG 30.81 and LB-572. The present results showed that the organism grew better in acidic to neutral pH than the alkaline. Maximum biomass was reported in the cultures of green alga grown at neutral pH whereas lowest in alkaline pH 9.0 (Anirban et al., 2002). However B. braunii has shown its tolerance to pH range of 6 to 9 without much effect on growth and hydrocarbon yields, which shows its adaptability for out door cultivation.
Effect of different nitrogen sources: Data obtained for autotrophic media with different nitrogen sources on growth and hydrocarbon are given in Fig. 3. As shown in Fig. 3, both the strains grew better in the presence of potassium, calcium and sodium nitrates compared to urea and ammonium nitrate. Correlation of hydrocarbon productivity with the biomass yields reveals that the hydrocarbon production is growth associated. In the absence of nitrates the growth was poor and the cells were bleached or turned brown.
| Table 1: | GC analysis of hydrocarbons of B. braunii |
 | |
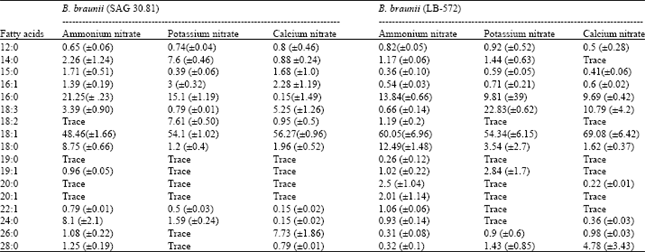
| Table 2: | Fatty acid profile of B. braunii |
 | |
Among the nitrates tested, potassium nitrate appeared to be more effective compared to calcium and sodium nitrates. The yield of biomass was 1.2 and 0.6 g L-1 with 32 and 38% (w/w) of hydrocarbon production respectively in B. braunii LB 572 and SAG 30.81 in potassium nitrate.
The hydrocarbon profile (as analysed by GC) in B. braunii cultures grown in different nitrogen sources is presented in Table 1. Both the strains showed higher proportion of hydrocarbons of chain length greater than C30 in potassium nitrate followed by calcium nitrate while urea showed relatively higher proportion of hydrocarbons of chain length less than C20. Compared to the strain LB 572, SAG 30.81 produced higher percentage of hydrocarbons of more than C30. In all the nitrogen sources, hydrocarbons in the range of C20 to C30 represent the major proportion of the total (60-73%).
The fatty acid content and profile of B. braunii grown in ammonium, potassium and calcium nitrates were studied as a representative of different nitrogen sources and the data is shown in Table 2 and Fig. 4. Oleic acid was found to be the major fatty acid in both the strains as well as in all the nitrogen sources studied. The next dominating fatty acid was palmitic acid, which was relatively in high percentage in the strain SAG 30.81 compared to LB 572. The two strains exhibited variations in their fatty acid profiles under the influence of different nitrogen sources, especially in C18:0, C16:0, C18:2, C18: 3, C24:0 C26:0 and C28:0. Odd chain fatty acids were also present in trace amounts in both the strains (Table 2).
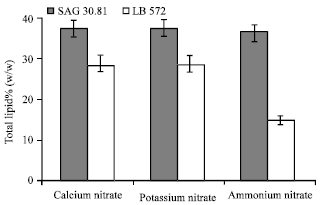 | |
| Fig. 4: | Total lipid contents of B. braunii (LB 572 and SAG) cultures grown in different nitrogen sources |
Anirban et al. (2002) reported that oleic acid was the precursor for the n-alkadienes and the algal strains used in the present study were reported to be as they belong to race A (Dayananda et al., 2005).
As shown in Fig. 4, there is no significant effect of ammonium, calcium and potassium nitrates on total lipid content of SAG 30.81, since the fat content of the alga was found in the range of 36-38% (w/w). In LB 572 calcium and potassium nitrate supplementation resulted in production of 28% of fat whereas in ammonium nitrate it was 15% (Fig. 4).
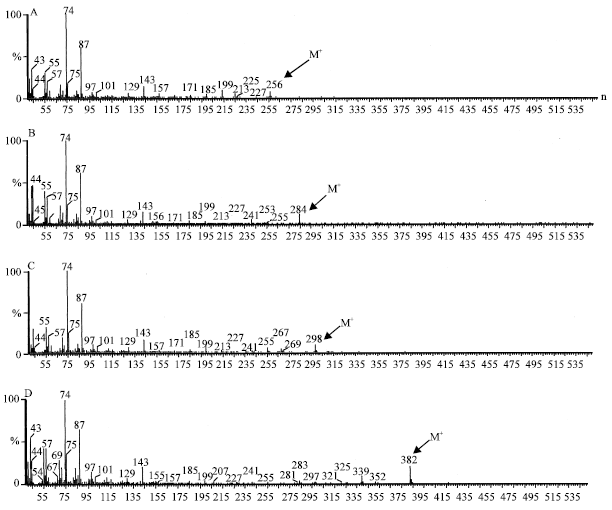 | |
| Fig. 5: | Mass spectra of 12-methyl hexadecanoic acid (A), 14-methyl tetradecanoic acid (B) ,16-Methyl heptadecanoic acid (C) and 25-methyl heptacosanoic acid (D) |
The B. braunii strains, SAG 30.81 and LB 572 were found to produce methyl branched fatty acids viz; 16-methyl heptadecanoic (6 mg g-1), 12-methyl tetradecanoic acid (2 mg g-1), 14-methyl hexadecanoicacid (0.4 mg g-1) and 25-methyl heptacosanoic acids (0.4 mg g-1) in trace amounts as identified by GCMS by comparing their fragmentation pattern with NIST library spectra and were shown in Fig. 5. Recently Dayananda et al. (2006) reported the presence of alkyl substituted fatty acids, 16-methyl heptadecanoic acid and 5, 9, 15-trimethyl tetradecanoic acid in botryococcene producing strain of B. braunii. Although, the significance of these fatty acids is not known, Krishnan (2003), Krishnan and Collin (2003) filed a patent that alkyl substituted fatty acids such as 16-methyl heptadecanoic acids capable of inhibiting endothelial cell and leukocyte proliferation.
The present results describe the influence of culture conditions on B. braunii for maximizing the productivity of biomass and biocrude. These promising results have shown that the organism can grow in the wider range of pH and in different nitrogen sources and can also provide the basis for further development of it’s out door cultivation methodologies.
Authors thank Department of Biotechnology, Government of India, New Delhi for their financial support and Dr. V. Prakash, Director, CFTRI for his support to carry out this work.