Research Article
Inhibition of Membrane Effects of General Anesthetic Propofol by Benzodiazepine Inverse Agonist Tetrahydro-β-carboline
Department of Dental Basic Education, Asahi University School of Dentistry, Mizuho, Gifu, Japan
Since its discovery in 1980 (James and Glen, 1980), propofol (2,6-diisopropylphenol) has been widely used as an intravenous anesthetic with the advantages in rapid onset and offset of action and easy maintenance of general anesthesia but relatively low toxicity. In respect to an anesthetic mechanism for propofol, attention has been primarily focused on its binding to GABAA receptors (Trapani et al., 2000). Propofol potentiates GABA-evoked chloride currents and directly activates chloride currents through GABAA receptors (Mohammadi et al., 2001).
Different drugs potentially interact with propofol and alter its anesthetic potency (Fidler and Kern, 2006). In the interaction on GABAA receptors, benzodiazepines such as midazolam and diazepam show synergism with propofol to potentiate GABA-activated chloride currents (Luo and Sugiyama, 2000). In contrast, benzodiazepine inverse agonist tetrahydro-β-carbolines (Koleva et al., 2012) have the effects opposite to benzodiazepines (Emmanouil and Quock, 1990). One of representative tetrahydro-β-carbolines, 1-methyl-1,2,3,4-tetrahydro-β-carboline (MTBC) is formed from tryptamine by condensing with acetaldehyde and pyruvic acid (Fig. 1). MTBC and its related β-carbolines are present in the human body because of being exogenously supplied through the intake of certain beverages and foods and being endogenously produced through the possible in vivo condensation. Their increasing concentrations in blood, brain and other tissues have been extensively investigated in association with alcohol consumption (Myers, 1989; Pfau and Skog, 2004). Tetrahydro-β-carbolines and β-carbolines have affinities to the benzodiazepine binding site of GABAA receptors, but exert inverse agonist effects (Ferretti et al., 2004; Venault and Chapouthier, 2007). Propofol activates rat locus coeruleus GABAA receptors to decrease the firing rate of neurons (Chen et al., 1999), whereas β-carbolines oppositely stimulate the locus coeruleus neurons (Ruiz-Durántez et al., 2001). The antagonistic interaction is also known between tetrahydro-β-carboline and anesthetic nitrous oxide acting on GABAA receptors (Emmanouil and Quock, 1989).
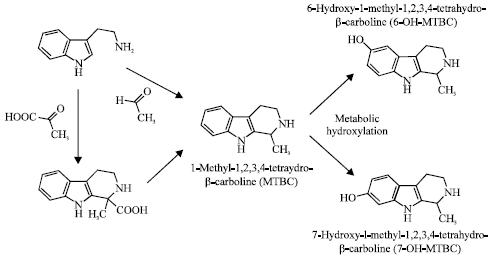 | |
| Fig. 1: | Chemical structures of tetrahydro-β-carbolines |
Aside from acting on the GABAA receptor-channel complex, propofol acts on the lipid bilayer portions of nerve cell membranes to modify their biophysical properties like membrane fluidity (Tsuchiya, 2001; Bahri et al., 2007). Such membrane effects modulate the activities of membrane-embedded receptors (Søgaard et al., 2006). Interestingly, several β-carboline compounds similarly act on lipid bilayers to cause membrane fluidity changes, which is responsible for their pharmacological effects (Peura et al., 1982; Tsuchiya and Ohmoto, 2010).
Both propofol and tetrahydro-β-carbolines possess the ability to act on GABAA receptors and membrane lipids. Previous studies showed their inhibitory interaction at a receptor level. However, little information is available about the interaction at a membrane level when propofol and tetrahydro-β-carbolines are used in combination. The purpose of this study was to verify the hypothetical inhibitory relation between propofol and tetrahydro-β-carbolines (Fig. 1) on their induced membrane fluidity changes by addressing whether MTBC and its metabolites 6-hydroxy-1-methyl-1,2,3,4-tetrahydro-β-carboline (6-OH-MTBC) and 7-hydroxy-1-methyl-1,2,3,4-tetrahydro-β-carboline (7-OH-MTBC) influence the membrane effects of propofol.
Propofol was previously reported to induce membrane fluidization (Tsuchiya, 2001; Bahri et al., 2007). However, a problem against its clinical significance has been indicated, that is, the concentrations for propofol to change membrane fluidity are much higher than those in blood and brain tissues during anesthesia (Franks, 2006). Although tetrahydro-β-carbolines of μmol L-1 or nmol g-1 levels are contained in certain beverages, foods and seasonings (Pfau and Skog, 2004), MTBC in blood is presumed to show low nanomolar or sub-nanomolar concentrations (Kari et al., 1979; Louis et al., 2005). Therefore, using model membranes, the interactions between propofol and tetrahydro-β-carbolines (MTBC, 6-OH-MTBC and 7-OH-MTBC) were investigated with special attention to their clinically and physiologically relevant concentrations.
Materials: Propofol was purchased from Sigma-Aldrich (St. Louis, MO, USA). MTBC, 6-OH-MTBC and 7-OH-MTBC were synthesized as reported previously (Tsuchiya et al., 1995). 1,2-Dipalmitoyl-phosphatidylcholine (DPPC) and 1-palmitoyl-2-oleoylphosphatidylcholine (POPC) were obtained from Avanti Polar Lipids (Alabaster, AL, USA), cholesterol from Wako Pure Chemicals (Osaka, Japan) and 1,6-diphenyl-1,3,5-hexatriene (DPH) and N-phenyl-1-naphthylamine (PNA) from Molecular Probes (Eugene, OR, USA). Dimethyl sulfoxide (DMSO) of spectroscopic grade (Kishida, Osaka, Japan) was used for preparing reagent solutions. All other chemicals were of the highest grade available commercially.
Preparation of membranes: Lipid bilayer membranes were prepared to be liposomes (total lipids of 0.14 mM) suspended in 10 mM tris-HCl buffer (pH 7.4, containing 125 mM NaCl, 5 mM KCl and 0.1 mM EDTA) as reported previously (Peura et al., 1982; Tsuchiya, 2001). Their lipid compositions were 100 mol% DPPC for DPPC membranes which have been most frequently used in membrane interaction studies (Bahri et al., 2007) and 60 mol% POPC and 40 mol% cholesterol for biomimetic membranes (Tsuchiya, 2001).
Determination of membrane effects: DPPC membranes were reacted at 37°C for 30 min with tetrahydro-β-carbolines by adding their solutions in DMSO to liposome suspensions to give final concentrations of 0.01-500 μM for MTBC, 0.01-0.5 μM for 6-OH-MTBC and 0.01-0.5 μM for 7-OH-MTBC. The concentration of DMSO was less than 0.5% (v/v) of the total volume so as not to affect the membrane fluidity. DMSO vehicle of the corresponding volume was added to control. After the reaction, the membranes were labeled with a fluorescent probe DPH or PNA, followed by measuring fluorescence polarization using an RF-540 spectrofluorometer (Shimadzu, Kyoto, Japan) equipped with a polarizer as reported previously (Tsuchiya, 2001). Polarization values were calculated by the formula (IVV-GIVH)/(IVV+GIVH) according to the method of Ushijima et al. (2005), in which I is the fluorescence intensity and the subscripts V and H refer to the vertical and horizontal orientation of the excitation and emission polarizer, respectively. The grating correction factor (G = IHV/IHH) is the ratio of the detection system sensitivity for vertically and horizontally polarized light, which was used to correct the polarizing effects of the monochromator (Brittes et al., 2010). Compared with control, a decrease and an increase of polarization values mean an increase (membrane fluidization) and a decrease of membrane fluidity (membrane rigidification), respectively. Since the polarization values of control membranes differed by varying fluorescence probes and membrane lipid compositions, the polarization changes (%) relative to control values were used for comparing membrane effects. Percent changes are effectively usable for the comparison regardless of the different polarization values of control membranes (Alfahel et al., 1996).
Influence of MTBC on propofol membrane effects: Biomimetic membranes were pretreated with 0.01-5 nM MTBC at 37°C for 30 min. MTBC-pretreated and not-treated membranes were reacted with 0.0625-1 μM propofol at 37°C for 30 min, followed by measuring DPH fluorescence polarization as described above. MTBC and propofol solutions in DMSO were added to liposome suspensions and the concentration of DMSO was adjusted to be less than 0.5% (v/v) of the total volume so as not to affect the membrane fluidity. Control experiments were conducted with the addition of an equivalent volume of DMSO vehicle. The inhibition (%) of propofol membrane effects was obtained by comparing polarization decreases induced by propofol with those by MTBC pretreatment.
Data analysis: Results are expressed as Mean±SEM (n = 8). Data were statistically analyzed by a one-way analysis of variance (ANOVA) followed by a post hoc Fisher’s protected least significant difference (PLSD) test using StatView 5.0 (SAS Institute, Cary, NC, USA). p<0.05 were considered to be statistically significant.
Membrane effects of tetrahydro-β-carbolines: In the pilot experiments using DPPC membranes, MTBC biphasically changed membrane fluidity in a concentration-dependent manner (Fig. 2). As shown by DPH and PNA polarization decreases and increases, MTBC fluidized DPPC membranes at relatively high concentrations but rigidified at relatively low concentrations.
In the concentration range of 0.01-0.5 μM, MTBC, 6-OH-MTBC and 7-OH-MTBC differently acted on DPPC membranes (Fig. 3). MTBC decreased membrane fluidity with the potency depending on concentrations. However, neither 6-OH-MTBC nor 7-OH-MTBC showed any membrane effects.
Inhibition of propofol membrane effects by MTBC: Propofol increased the fluidity of biomimetic membranes at sub-micromolar concentrations as shown by DPH polarization decreases (Fig. 4). Propofol was effective in fluidizing biomimetic membranes at 0.125-1 μM.
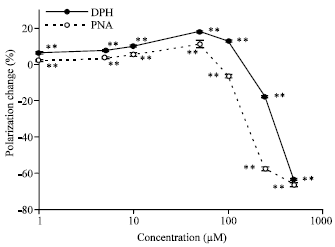 | |
| Fig. 2: | Membrane effects of MTBC. DPPC membranes were reacted with MTBC of the indicated concentrations, followed by determining DPH and PNA fluorescence polarization changes from control. Values are expressed as Mean±SEM (n = 8). **p<0.01, compared with control |
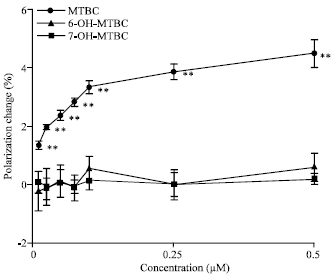 | |
| Fig. 3: | Comparative effects of tetrahydro-β-carbolines on DPPC membranes. DPPC membranes were reacted with MTBC, 6-OH-MTBC and 7-OH-MTBC of the indicated concentrations, followed by determining DPH fluorescence polarization changes from control. Values are expressed as Mean±SEM (n = 8). **p<0.01, compared with control |
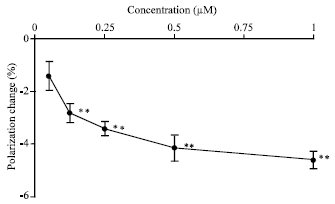 | |
| Fig. 4: | Effects of propofol on biomimetic membranes at sub-micromolar concentrations. Membranes consisting of 60 mol% POPC and 40 mol% cholesterol were reacted with propofol of the indicated concentrations, followed by determining DPH fluorescence polarization changes from control. Values are expressed as Mean±SEM (n = 8) **p<0.01, compared with control |
The fluidizing effects of 0.125 and 0.25 μM propofol on biomimetic membranes were inhibited by MTBC of low nanomolar and sub-nanomolar concentrations (Fig. 5).
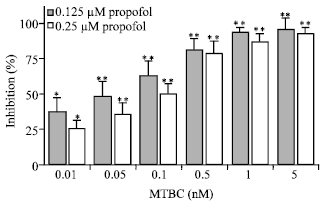 | |
| Fig. 5: | Inhibition by MTBC of propofol effects on biomimetic membranes. Membranes consisting of 60 mol% POPC and 40 mol% cholesterol were pretreated with 0.01-5 nM MTBC and then reacted with 0.125 and 0.25 μM propofol. The inhibition (%) of propofol membrane effects by MTBC was obtained by comparing DPH fluorescence polarization decreases induced by propofol with those by MTBC pretreatment. Values are expressed as Mean±SEM (n = 8). *p<0.05 and **p<0.01, compared with control |
Main findings of this study are as follows: (1) MTBC is able to decrease the fluidity of DPPC membranes at sub-micromolar concentrations, although it biphasically increases membrane fluidity at higher concentrations, (2) its hydroxyl metabolites are not effective in changing membrane fluidity, (3) propofol fluidizes biomimetic membranes consisting of POPC and cholesterol at 0.125-1 μM and (4) such a membrane-fluidizing effect of propofol is inhibited by 0.01-5 nM MTBC.
The blood concentrations of propofol to induce anesthesia were reported to range from 17 to 35 μM (Chen et al., 1999) but propofol in blood was found to be highly bound to proteins with the binding ratio of 97-99% (Servin et al., 1988). The brain/blood concentration ratio of propofol was about three (Shyr et al., 1995). Based on these studies, free propofol in blood is considered to show sub-micromolar concentrations during anesthesia. The membrane lipids of human brains primarily consist of phospholipids with 1-saturated and 2-unsaturated acyl chains and cholesterol (Svennerholm et al., 1994). Therefore, biomimetic membranes prepared with POPC and cholesterol were subjected to the reaction with ~1 μM propofol. Consequently, propofol has been revealed to fluidize biomimetic membranes at clinically relevant concentrations. Propofol would modify the property and structure of biomembranes to influence the transmission function of nerve cells through membrane fluidization (Tsuchiya, 2001; Bahri et al., 2007). The activity and sensitivity of drug receptors including GABAA receptor are also modulated by changes in membrane biophysical property (Gimpl et al., 1997; Sooksawate and Simmonds, 2001).
The in vivo contents of MTBC and its related β-carbolines are increased by drinking alcoholic beverages and several diseases (Myers, 1989; Tsuchiya et al., 1996). Even so, MTBC is presumed to be present at low nanomolar or sub-nanomolar concentrations in blood (Kari et al., 1979; Louis et al., 2005). Therefore, biomimetic membranes were pretreated with ~5 nM MTBC and then reacted with ~0.25 μM propofol. Consequently, it has been proved that the membrane-fluidizing effects of 0.125 and 0.25 μM propofol are significantly inhibited by 0.01-5 nM MTBC. Propofol decreased DPH and PNA fluorescence polarization of DPPC membranes. Since fluorescence probe DPH and PNA are localized in the hydrocarbon core and the hydrophobic upper region of lipid bilayers, respectively, propofol penetrates into lipid bilayer membranes and act on both membrane regions to increase the fluidity (Tsuchiya, 2001; Bahri et al., 2007). Propofol is considered to perturb the alignment of phospholipid acyl chains, resulting in an increase of membrane fluidity. Although, the detailed membrane-interacting mechanism of MTBC is unclear, this study indicates that MTBC acts on the hydrocarbon core of lipid bilayers similarly to propofol, but oppositely decreases membrane fluidity at relatively low concentrations as shown by DPH polarization increases. Several drugs were also reported to influence membrane biophysical properties in a biphasic manner as well as MTBC (Meddings et al., 1991; Gallová et al., 1995). Propofol fluidizes biomimetic membranes at clinically relevant concentrations, whereas MTBC rigidifies at physiologically presumable concentrations.
The combination of different drugs frequently produces synergistic, additive or antagonistic effects. The synergistic interactions of anesthetic drugs have been clinically attracting much attention (Rosow, 1997), while diazepam and midazolam antagonistically interact with morphine, fentanyl and butorphanol (Kissin et al., 1990; Dershwitz et al., 1991). In addition to their interaction on GABAA receptors, propofol and MTBC are likely to show the inhibitory relation at a membrane level that they antagonistically change membrane fluidity. The inhibitory interactions were previously reported between membrane-acting drugs in respect to membrane biophysical modification. Tamoxifen is a selective modulator of estrogen receptors but its membrane action is also responsible for various pharmacological activities. The membrane-fluidizing effect of tamoxifen was inhibited by a receptor antagonist (Tsuda and Nishio, 2005). General anesthetics commonly have membrane-fluidizing effects, some of which were regulated by membrane-rigidifying cholesterol (Burger et al., 2000; Parat, 2006). Cholesterol reduced the potency of GABA at GABAA receptors by causing a decrease of plasma membrane fluidity (Sooksawate and Simmonds, 2001), which is opposite to a propofol-inducing increase of membrane fluidity.
While the clinical implication of propofol membrane effects inhibited by MTBC may be beyond the scope of this study, the speculative discussion would be helpful for the following studies. It has been believed that alcoholic patients acquire the tolerance to anesthesia, thereby requiring larger doses of general anesthetics (Servin et al., 2003). The induction dose of propofol was found to increase in chronic alcoholism (Fassoulaki et al., 1993). The in vivo concentrations of MTBC and its related β-carbolines were remarkably increased by drinking alcoholic beverages and in chronic alcoholics (Myers, 1989; Tsuchiya et al., 1996). Taken together with these previous studies, the present results suggest the hypothesis that the interaction of propofol with MTBC may be, at least in part, responsible for the anesthetic tolerance associated with alcohol consumption. The membrane effects opposite to propofol are confined to MTBC, not found in 6-OH-MTBC and 7-OH-MTBC. Tetrahydro-β-carbolines are metabolized in liver and brain by cytochrome P450 enzymes. MTBC is converted to 6-hydroxyl and 7-hydroxyl metabolites (Tsuchiya et al., 1995), both of which are not able to act on membrane lipid bilayers due to their elevated hydrophilicity. Beta-carbolines show high affinities to CYP2D6 and CYP2E1 belonging to the super family of cytochrome P450, which participate in the metabolism of β-carbolines (Herraiz et al., 2008; Yu, 2008). CYP2D6 and CYP2E1 are induced in liver and brain by alcohol exposure and in chronic alcoholics (Miksys et al., 2002; Lu and Cederbaum, 2008), thereby leading to a decrease of the membrane-rigidifying effect of MTBC, resulting in a reduction of its potency to inhibit propofol membrane effects. The induction of such enzymes for converting MTBC to membrane-inactive hydroxyl metabolites may account for a phenomenon that anesthetic tolerance does not necessarily occur in chronic alcohol consumers.
General anesthetic propofol mechanistically increases the fluidity of biomembranes at clinically relevant concentrations. In contrast, MTBC oppositely decreases membrane fluidity at physiologically presumable concentrations. In addition to their antagonistic interaction on GABAA receptors, propofol and MTBC show the inhibitory interaction on membrane effects.
This study was supported by grants-in-aid for scientific research 20592381 and 23593005 from the Japan Society for the Promotion of Science.