ABSTRACT
The aim of this study was to evaluate the effects of arbuscular mycorrhizal fungus on the mineral nutrition and yield of Trifolium alexandrinum plants under different salinity levels (2.2, 5 and 10 dS m-1). A pot experiment was conducted under glasshouse conditions in 2007. The results showed positive effects of inoculation with mycorrhizal fungi on total dry weight, root length and nutrient uptake of the T. alexandrinum at high and low salinity levels. Mycorrhizal colonization increased Total Dry Weight (TDW) of mycorrhizal plants 5.29 times more than control plants. In shoot system of non-AM plants, Na+ concentration was increased while the concentrations of K+, Mg2+ and Ca2+ were decreased with raising salinity stress. The Na+ level in shoots of AM plants showed slight increase with raising salinity levels. This experiment showed that phosphorus levels in the plants were reduced with increasing salinity but the AM plants showed higher values of phosphorus at all salinity levels. Thus, it could be concluded that AM fungi increased phosphorus uptake and saline stress in plants was thereby alleviated.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ja.2009.79.83
URL: https://scialert.net/abstract/?doi=ja.2009.79.83
INTRODUCTION
Salt stress has become an ever-increasing threat to food production, irrigation being a major problem of agricultural fields due to gradual salinization. Salt stress has three fold effects: it reduces water potential, causes ion imbalance or disturbance in ion homeostasis and toxicity (Rabie and Almadini, 2005). This altered water status leads to initial growth reduction and limitation of plant productivity. Since, salt stress involves both osmotic and ionic stress (Hayashi and Murata, 1998; Munns, 2002; Benlloch-Gonzales et al., 2005). Growth suppression is directly related to total concentration of soluble salts or osmotic potential of soil water (Flowers, 2004; Pascal et al., 2005; Katerji et al., 2005). The detrimental effect, as death of plants or decrease in productivity is observed at the whole-plant level. Suppression of growth occurs in all plants, but their tolerance levels and rates of growth reduction at lethal concentrations of salt vary widely among different plant species. Salt stress affects all the major processes such as growth, photosynthesis, protein synthesis and energy and lipid metabolism (Ramoliya et al., 2004; Parida and Das, 2005). Many studies have demonstrated that inoculation with the AM fungi improves growth of plants under the of salinity stress conditions (Diallo et al., 2001; Burke et al., 2003; Tian et al., 2004). Recently, Rabie (2005) suggested that the AM fungi protected the host plants against the detrimental effects of salt. When mung bean (Vigna radiata) plants were irrigated with different dilution of seawater. The AM plants showed higher growth than the non-AM plants at all the levels of irrigation. Mycorrhizae were involved in protection against salt stress, via better access to nutritional status (Zandavalli et al., 2004) and modification of plant physiology i.e., osmotic modifications (Roa and Tak, 2002) and photosynthesis (Merguihur et al., 2002). To some extent, these AM fungi have been considered as bio-ameliorators of saline soils (Yano-Melo et al., 2003; Tian et al., 2004). Therefore, the present study was conducted to evaluate the efficacy of the AM fungus, Glomus clarum, in the development of salinity tolerance of the berseem clover (Trifolium alexandrinum) plant under salinity stress.
MATERIALS AND METHODS
A pot experiment was conducted in a glasshouse during December to February in 2006-2007 at Ramin Agricultural and Natural Resources University, Ahwaz, Iran (31° 36’ N, 48° 53’ E). This area is situated at an elevation of 50 m above sea levels. Agricultural topsoil obtained from a surface layer (0-20 cm) was used. According to soil analysis results, the soil (pH 7.5), which was dried then ground and sieved (2.0 mm) contained 70.9% sand, 12.5% silt and 16.6% clay, 8.4 g kg-1 organic carbon, 6.3 mg kg-1 available nitrogen, 7 mg kg-1 available phosphorus, 240 mg kg-1 available potassium and 1.7 dS m-1 EC. The soil was autoclaved at 110°C and 240 kPa pressures for 1 h on two occasions over a two-day period to ensure all possible mycorrhizal propagules had been destroyed (Asghari, 2004). Trifolium alexandrinum seeds were used as an experimental material. They were sterilized with a mixture of 1 part sodium hypochlorite (12.5% w/v) and 2 parts of reverse-osmosis water and soaked for 10 min in diluted sodium hypochlorite and then rinsed three times (Asghari, 2004). Spores consisted of the mycorrhizal inoculum’s mycelium and root segments of the Glomus intraradices isolate propagated with berseem clover (T. alexandrinum) roots for 4 months (Rabie and Almadini, 2005). Each used pot was inoculated with 20 g inoculum for the mycorrhizal treatments, or 20 g sterilized inoculum with 20 mL filtrates free from mycorrhizal propagules from the inoculum for the non-mycorrhizal treatments. The inoculum was placed 3 cm below the surface of the soil before sowing to produce mycorrhizal plants. The berseem clover (Trifolium alexandrinum) was used as the model plant. The experiment was a 3x2x3 complete factorial which was comprised of three salinity levels and two inoculation treatments with three replications. It was carried out with the following treatments: non-mycorrhizal control and inoculation with Glomus intraradices inoculum, each at three salt levels (three NaCl concentrations) of 2.2 (control), 5.0 and 10.0 dS m-1. Nutrient solution containing N at 100 mg L-1, P at 50 mg L-1 and K at 50 mg L-1 was added to the soil at a rate of 100 mL every week. Twenty seeds were sown into each pot and were thinned to twelve seedlings per pot after emergence. Tap water was supplied daily and the pots were weighted every week to adjust water content. Plants were harvested after 63 days. At the end of the experiment, dry matter was measured. Shoot dry weight was recorded after drying in an oven at 70°C (Alamgir-Kabir et al., 2006). Plant materials were digested with concentrated H2SO4 and H2O2 (Tian et al., 2004). Phosphorous was extracted by nitric-perchloric acid digestion and measured using the vanadonomolybdophosphoric calorimetric method (Jackson, 1967). K and sodium (Na) of shoot system were assayed using a flame spectrophotometer, while calcium (Ca) and magnesium (Mg) of plant shoots were determined by atomic absorption (Allen et al., 1984). Immediately after harvest, part of the root system of non-AM and AM plants was washed carefully in 4°C water to remove the adhering soil particles (Rabie and Almadini, 2005). Roots were cut into 1 cm segments and thoroughly mixed and samples of 0.5 g was cleared with 10% (w/v) KOH at 90°C for 2 h and stained with acid fuchsin (Tian et al., 2004). The mycorrhizal colonization and root length were tested by the grid line-intersect method described by Giovannetti and Mosse (1980). Mycorrhizal Growth Response (MGR) was calculated using the individual total plant dry weight (DW) of M and mean dry weight of NM plants at a given NaCl level (Asghari, 2004):
Statistical analysis: All data were analyzed with SAS Institute Inc. 6.12. All data were first analyzed by ANOVA to determine significant (p≤ 0.05) treatment effects. Significant differences between individual means were determined using grouped in Duncan multiple comparison test.
RESULTS
Plant growth and mycorrhizal growth response (MGR): Mycorrhizal infection significantly stimulated the berseem clovers’ growth (Table 1). At the end of experimental period, a significant effect of mycorrhizal symbiosis was observed in the root and shoot dry weights of salinity-stressed plants with respect to the non-mycorrhizal stressed plants. Control (mycorrhizal and non-mycorrhizal) plants showed a higher plant height compared with salinity stress-treated plants (Table 1). With increasing NaCl levels, the mycorrhizal growth responses of berseem clovers’ showed decreasing trend. The biggest MGR was at 2.2 dS m-1.
Root length: Mycorrhizal colonization had significant effect on T. alexandrinum root length. The results showed that total root length mycorrhizal inoculated plants was much more than non-mycorrhizal plants. This result is in agreement with the earlier study of Largewerff and Holland (1960). With increasing salinity levels, root length significantly decreased particularly at non-mycorrhizal plants.
Colonization: In this experiment no colonization by AM fungi in control plants. Inoculated plants had colonization percentages ranging from 63 to 34%. The colonization rate declined with increased NaCl level, indicating that salinity suppressed the formation of arbuscular mycorrhizae.
| Table 1: | Effect of salinity levels on shoot and root dry weight, height, root length, % Mycorrhizal Growth Response (MGR) and % colonization of Mycorrhizal (M) and Non-Mycorrhizal (NM) berseem clover |
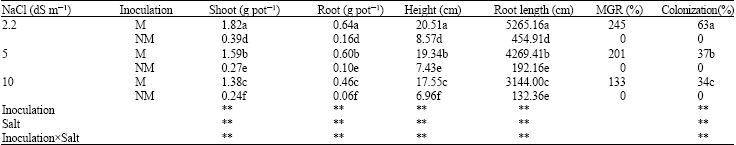 | |
| **Significant difference at p<0.01 by ANOVA. Mean values with different letter(s) are significantly different at p<0.01 | |
| Table 2: | Effect of salinity levels on P, Na, K, Ca and Mg concentrations in roots of Mycorrhizal (M) and Non-Mycorrhizal (NM) berseem clover |
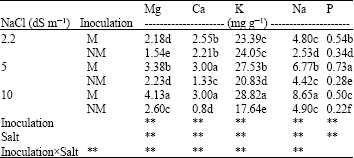 | |
| **Significant difference at p<0.01 by ANOVA. Mean values with different letter(s) are significantly different at p<0.01 | |
| Table 3: | Effect of salinity levels on P, Na, K, Ca and Mg concentrations in shoots of Mycorrhizal (M) and Non-Mycorrhizal (NM) berseem clover |
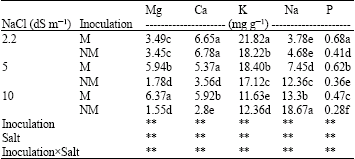 | |
| **Significant difference at p<0.01 by ANOVA. Mean values with different letter(s) are significantly different at p<0.01 | |
Nutrient uptake: Phosphorus concentrations in shoots and roots of M plants were much higher than NM plants. The P concentration of M and NM plants declined with increasing NaCl level.
Inoculated plants had higher root and shoot K and Mg concentrations than NM plants (Table 2, 3). Na concentration in roots of M plants was higher than their shoots.
DISCUSSION
AM fungi efficiency can be measured in terms of host plant growth under different environmental conditions (Ruiz-Lozano et al., 1996). In this sense, the inoculation of Glomus intraradices used in this study maintained the growth in colonized clover in both control and stress conditions compared to non-mycorrhizal plants (Table 1). This effect was more pronounced in aerial biomass than in root biomass (Table 1) which may be because of arbuscular mycorrhizal colonization, caused a proportionally greater allocation of carbohydrates to the shoot than to the root tissues (Schwab et al., 1982). Earlier research has shown that salinity may reduce mycorrhizal colonization by inhibiting the germination of spores (Hirrel, 1981), inhibiting growth of hyphae in soil and hyphal spreading after initial infection had occurred (McMillen et al., 1998) and reducing the number of arbuscules (Tian et al., 2004).
Saline stress induces P deficiency. AM fungi increased P uptake and saline stress in plants was thereby alleviated. This is consistent with previous findings that the main mechanism for enhanced salinity tolerance in mycorrhizal plant was the improvement of P nutrition (Al-Karaki et al., 2001), although in some cases, however, saline tolerance of mycorrhizal plants appears to be independent of plant P concentration (Feng et al., 2002).
Experimental evidences from this study indicated that increased treatment of NaCl induced significant increase in Na+ and decrease in K+, Mg2+ and Ca2+ levels in shoot system of non-AM berseem clover plants (Table 3). These results are in agreement with the previous observations (Aziz and Khan, 2001; Ferreira et al., 2001; Parida et al., 2004). These researchers reported that high salt (NaCl) uptake competes with the uptake of other nutrient ions, especially K+, leading to K+ and other ions deficiency. The ions deficiency displayed by salinity stress, particularly by NaCl uptake indicated a nutritional imbalance. It has been generally accepted that AM fungi would enhance nutrient uptake by infected plants under salinity conditions (Rao and Tak, 2002; Yano-Melo et al., 2003; Zandavalli et al., 2004). It appears from the present study that AM berseem clover plants contained significant higher levels of K+, Mg2+ and Ca2+ ions than non-AM plants at all salinity levels. Some studies have reported that AM fungal colonization had no significant effects on plant K contents or concentrations (Al-Karaki, 2000; Mohammad et al., 2003), but results of this study indicate that root K concentrations increased even at high salinity level. Based on these results and on existing literatures, the greater salt tolerance of AM plants may be the result of the plant nutrition improvement under salinity stress. It is also noteworthy that Na+ concentration in shoot system of AM berseem clover plants at high salinity level was comparable to that of non-AM plants at moderate salinity level. These results are consistent with the earlier study (Zandavalli et al., 2004; Rabie et al., 2005) and suggest that AM fungi may protect shoot system, mainly leaves, from Na+ toxicity either by regulating Na+ uptake from the soil or by accumulating it in root thereby delaying its translocation onto shoot system of infected plants. The role of K+ and Ca2+ in salt adaptation of plants has been previously discussed by several authors. Parida and Das (2005) reported that when under salt stress; plants maintain high concentrations of K+ and low concentrations of Na+ in the cytosol. They do this by regulating the expression and activity of K+ and Na+ transporters and of H+ pumps that generate the driving force for transport. In addition, externally supplied Ca2+ reduces the toxic effects of NaCl, presumably by facilitating higher K+/Na+ selectivity. High salinity also results in increased cytosolic Ca2+ that is transported from the apoplast and intracellular compartments. The resultant transient Ca2+ increase potentialities stress signal transduction and leads to salt adaptation. Based on these data and existing literatures it is conceivable to conclude that AM symbioses may regulate the expression and activate K+ and Na+ transporters and H+ pumps that generate the driving force for transport. Besides, it may increase the transient Ca2+ from apoplast and intracellular compartments. This inference needs further investigation to support it.
CONCLUSION
In summary, the results indicate that, inoculation with mycorrhizal fungi increased plant growth and nutrient uptake of the T. alexandrinum in saline conditions even with very low root colonization. These results may be of practical importance, as they highlight the potential of using mycorrhizal fungi inoculation in legumes to revegetate saline lands, especially where indigenous mycorrhizal populations are low or do not result in colonization of legumes. More work to determine factors that promote the AM colonization under field conditions is required before this potential could be realized.
REFERENCES
- Al-Karaki, G.N., 2000. Growth of mycorrhizal tomato and mineral acquisition under salt stress. Mycorrhiza, 10: 51-54.
CrossRefDirect Link - Al-Karaki, G.N., R. Hammad and M. Rusan, 2001. Response of two tomato cultivars differing in salt tolerance to inoculation with mycorrhizal fungi under salt stress. Mycorrhiza, 11: 43-47.
CrossRefDirect Link - Benlloch-Gonzalez, M., J.M. Fournier, J. Ramos and M. Benlloch, 2005. Strategies underlying salt tolerance in halophytes are present in Cynara cardunculus. Plant Sci., 168: 653-659.
Direct Link - Burke, D.J., E.P. Hamerlynck and D. Hahn, 2003. Interactions between the salt marsh grass season. Spartina patens, arbuscular mycorrhizal fungi and sediment bacteria during the growing. Soil Biol. Biochem., 35: 501-511.
Direct Link - Diallo, A.T., P.I. Samb and H. Roy-Macauley, 2001. Water status and stomatal behaviour of cowpea, Vigna anguiculata (L.) Walp, plants inoculated with two Glomus species at low moisture levels. Eur. J. Soil Biol., 37: 187-196.
Direct Link - Feng, G., F.S. Zhang, X.L. Li, C.Y. Tian, C. Tang and Z. Rengel, 2002. Improved tolerance of maize plants to salt stress by arbuscular mycorrhiza is related to higher accumulation of soluble sugars in roots. Mycorrhiza, 12: 185-190.
Direct Link - Ferreira, R.G., F.J. Tavora and F.F. Hernandez, 2001. Dry matter partitioning and mineral composition of roots, stems and leaves of guava grown under salt stress conditions. Pesquisa Agropecuaria Brasileira, 36: 79-88.
CrossRefDirect Link - Giovannetti, M. and B. Mosse, 1980. An evaluation of techniques to measure vesicular-arbuscular infection in roots. New Phytol., 84: 489-500.
CrossRefDirect Link - Hirrel, M.C., 1981. The effect of sodium and chloride salts on the germination of Gigaspora margaria. Mycology, 43: 610-617.
Direct Link - Katerji, N., J.W. Hoorn, A. Hamdy, M. Mastrorilli, M. Nachit and T. Oweis, 2005. Salt tolerance analysis of chickpea, faba bean and durum wheat varieties: II. Durum wheat. Agric. Water Manage., 72: 195-207.
Direct Link - McMillen, B.G., S. Juniper and L.K. Abbott, 1998. Inhibition of hyphal growth of a vesicular-arbuscular mycorrhizal fungus in soil containing sodium chloride limits the spread of infection from spores. Soil Biol. Biochem., 30: 1639-1646.
Direct Link - Mohammad, M.J., H.I. Malkawi and R. Shibli, 2003. Effects of arbuscular mycorrhizal fungi and phosphorus fertilization on growth and nutrient uptake of barley grown on soils with different levels of salts. J. Plant Nutr., 26: 125-137.
Direct Link - Munns, R., 2002. Comparative physiology of salt and water stress. Plant Cell Environ., 25: 239-250.
CrossRefDirect Link - Parida, A.K., A.B. Das and B. Mittra, 2004. Effects of salt on growth, ion accumulation, photosynthesis and leaf anatomy of the mangrove, Bruguiera parviflora. Trees, 18: 167-174.
CrossRefDirect Link - Parida, A.K. and A.B. Das, 2005. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf., 60: 324-349.
CrossRefPubMedDirect Link - Pascal, S.D., A. Maggio and G. Barbieri, 2005. Soil salinization affects growth, yield and mineral composition of cauliflower and broccoli. Eur. J. Agron., 23: 254-264.
Direct Link - Rabie, G.H., 2005. Influence of arbuscular mycorrhizal fungi and kinetin on the response of mungbean plants to irrigation with seawater. Mycorrhiza, 15: 225-230.
CrossRefDirect Link - Ramoliya, P.J., H.M. Patel and A.N. Pandey, 2004. Effect of salinization of soil on growth and macro-and micro-nutrient accumulation in seedlings of Salvadora persica (Salvadoraceae). For. Ecol. Manage., 202: 181-193.
Direct Link - Rao, A.V. and R. Tak, 2002. Growth of different tree species and their nutrient uptake in limestone mine spoil as influenced by Arbuscular Mycorrhizal (AM)-fungi in India arid zone. J. Arid Environ., 51: 113-119.
CrossRefDirect Link - Ruiz-Lozano, J.M., R. Azcon and M. Gomez, 1996. Alleviation of salt stress by arbuscular-mycorrhizal Glomus species in Lactuca sativa plants. Physiol. Plant., 98: 767-772.
CrossRefDirect Link - Tian, C.Y., G. Feng, X.L. Li and F.S. Zhang, 2004. Different effects of arbuscular mycorrhizal fungal isolates from saline or non-saline soil on salinity tolerance of plants. Applied Soil Ecol., 26: 143-148.
CrossRefDirect Link - Yano-Melo, A.M., O.J. Saggin and L.C. Maia, 2003. Tolerance of mycorrhized banana (Musa sp. cv. Pacovan) plantlets to saline stress. Agric. Ecosyst. Environ., 95: 343-348.
CrossRefDirect Link - Zandavalli, R.B., L.R. Dillenburg and P.V. de Souza, 2004. Growth responses of Araucaria angustifolia (Araucariaceae) to inoculation with the mycorrhizal fungus Glomus clarum. Applied Soil Ecol., 25: 245-255.
CrossRefDirect Link - Alamgir-Kabir, M., A.T.M. Rafiqul-Hoque and M. Kamaluddin, 2006. Acclimation of acacia hybrid (Acacia mangium x Acacia auriculiformis) vegetative propagules to soil water deficits. Dendrobiology, 56: 37-43.
Direct Link - Aziz, I. and M.A. Khan, 2001. Experimental assessment of salinity tolerance of Ceriops tagal seedlings and saplings from the Indus delta, Pakistan. Aquatic Bot., 70: 259-268.
CrossRef - Rabie, G.H. and A.M. Almadini, 2005. Role of bioinoculants in development of salt-tolerance of Vicia faba plants under salinity stress. Afr. J. Biotechnol., 4: 210-222.
Direct Link








