Research Article
Effect of Calcination on Mo-Modified Zeolite Catalyst
Department of Fundamental and Applied Sciences, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
INTRODUCTION
One of the main sources of benzene (aromatic) in the world is produced via naphtha cracker or an ethylene cracker. For both the processes the main feedstock is crude oil. The present trend is to move away from crude oil. Natural gas shows promise. For aromatic production using natural gas a single step conversion of methane to aromatics under non-oxidative conditions can be an ideal choice for this gas to liquid technology. Aromatic are more favorable thermodynamically than olefins in this aromatization reaction. This technology selectively breaks C-H bonds of methane at the acid sites of the zeolite, which later recombines to form aromatics in the presence of a Mo carbide catalyst. The active catalyst in this technology is the hcp β-Mo2C supported on HZSM-5 zeolite. The main products from this reaction are aromatic (benzene and naphthalene) and coke. Coke deposition although undesirable is important at the initial stage of MDA because it is one of the active components of the said reaction. The Mo carbide catalyst is generally prepared by impregnation of ammonium heptamolybdate (AHM) onto the HZSM-5 zeolite. The AHM is anchored as Mo7O246- on the zeolite external surface, which on calcination between 450 to 700°C is converted to MoO3.
It has been establish that preparation and pretreatment of the catalyst can affect the channel structure and acidity of zeolite as well as the location of the Mo species on/in HZSM5 as described by Xu and Lin (1999); Lin et al. (1997) and Wang et al. (1997). This chemical environment can be significantly altered as a result of high temperature calcinations (treatment under atmosphere air). Xu et al. (1995a), Solymosi et al. ((1995) and Wang et al. (1997) carried out studies on various calcination temperature between 450 to 700°C for MDA reaction. The Mo carbide modified HZSM5 is a bifunctional catalyst. The MoCx sites convert CH4 into ethane and ethylene, which then react on the acid sites to form C6-C10 aromatic via rapid oligomerisation, cracking and cyclisation reactions.
From the above explanation it can be inferred that the main function of calcinations is the redistribution of the MoO3 species over the catalyst surface and into the channels of the zeolite or pore. This results in stronger interaction between the MoO3 species and the HZSM-5 zeolite. This effect leads to a more stable chemical environment which reduces or prevents the sublimation of MoO3 species as indicated by Borry III et al. (1999).
The present study investigates the influence of calcination temperature on the chemical and physical properties of the Mo-modified HZSM-5 zeolite utilized for MDA reaction.
Preparation of HZSM-5: NH4ZSM-5 with a Al/Si ratio of 41 was obtained from Zeolyst International (batch number = CBV8014G). NH4ZSM-5 was then dried at 150°C in an oven overnight. It was then cooled in a desiccator without desiccant for an hour before being calcined.
Preparation of MoO3/HZSM-5: The wt. % used in this study refers to Mo metal wt. % supported on HZSM-5 assuming that MoO3 is the final phase produced. In this study 10 wt.% Mo/HZSM-5 are prepared by wet impregnation of ammonium heptamolybdate tetrahydrate [(NH4)6Mo7O24.4H2O] ( AHM) onto H-ZSM-5. The AHM used in this study is of A.C.S. reagent grade obtained from Aldrich with formula weight 1235.86 and density of 2.498 and is the source of Mo.
Calcination of MoO3/HZSM-5: The powdered zeolite was packed loosely in porcelain boat and placed in a horizontal quartz tube reactor fed by a constant airflow of 12 mL min-1. This zeolite was then slowly heated up from room temperature to 400°C at a constant heating rate of 10°C min-1. At 400°C the temperature was maintained for 2 h. This is important to slowly remove excess water formed during the reaction and avoided zeolite dealumination due to steaming. Dealumination can result in reduced number of Brønsted acid sites and produce octahedrally coordinated extra-framework aluminium. After 2 h at 400°C, the temperature was then slowly raised to 500 to 650°C as per test requirement at constant rate of 5°C min-1 and kept for 5 h at this temperature to complete the transformation and at the same time remove any other excess water that might be present. It was then allowed to cool to room temperature at a constant rate of 10°C min-1. The zeolite was stored in a desiccator without desiccant. Catalyst calcined at 500, 600 and 650°C will be denoted either as 500, 600 and 650°C, respectively.
Pelletization of calcined MoO3 / HZSM-5: The cool calcined powder was further ground in a glass mortar and pestle. It was then introduced into a pellet maker and pressed into pellets under a pressure of 8 tons. Again the pellets were crushed by the pestle and mortar and sieved to a particle size of 250-425 μm (40-60 mesh).
Catalyst preparation: The calcined catalyst was activated by in-situ method in a fixed bed reactor. The calcined catalyst for the β form was obtained by carburisation and activation with methane gas flowing at a rate of 63 mL min-1 heated slowly from ambient to 700°C and held at the same temperature (700°C) for an hour as per method by Wang et al. (1993). At the end of dwell period of the carburisation stage the activation gas was replaced by helium at a flow rate of 7.5mL min-1, held for 15 min before cooling to ambient temperature and stored in a closed vial container for further testing.
Catalyst evaluation: A 3 vol % Ar (internal standard) in methane (main reactant) was used in the present study. Ar is selected as an internal standard as it is inert in MDA reaction. Ar present in the feed gas is used to constantly monitor the methane flow rate/concentration at the inlet as well as the outlet of the reactor. Quantification of product follows Liu et al. (1997) method. The MDA reaction was carried out using a quartz reactor tube at 700°C with a GHSV of 1500 h-1 over 20 h Time On Stream (TOS). The product was quantified hourly by an on-line GC as describe by Sinnathambi (2005).
RESULTS AND DISCUSSIONS
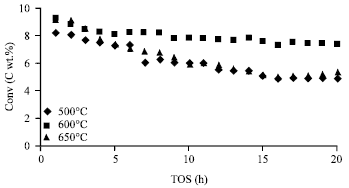
Methane conversion: Figure 1 represents the effect of calcination temperature (500, 600 and 650°C) on the catalytic performance with respect to methane conversion on 10Mo/HZSM-5 catalyst over 20 h TOS.
From the Fig. 1, it can be seen that a calcination temperature of 600°C gives the best methane conversion of the 3 studied. Both at calcination temperature of 600 and 650°C, the methane conversion for 1st h TOS starts at almost the same conversion, but the deactivation rate for 650°C increases at a faster rate than 600°C after 3 h TOS. In the case of 500, methane conversion starts at a lower rate as compared to that of 600 and 650°C, but after 3 h TOS, 500°C has a similar deactivation rate as that of 650°C.
 | |
| Fig. 1: | Effect of calcination temperature on methane conversion |
 | |
| Fig. 2: | Effect of calcination temperature on MDA product distribution |
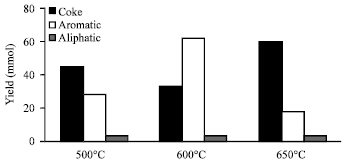
MDA product distribution: The main products from MDA are aromatic, aliphatic hydrocarbon and coke. Figure 2 shows the effect of calcinations temperature on the product yield in mmol over 20 h TOS on 10Mo/HZSM5 catalysts.
From Fig. 2, it can be seen that the catalytic activity for 600°C is highest. Calcinations at 600°C are 123% and 131% more reactive than that at 500 and 650°C, respectively.
Calcination at 600°C produces more aromatic than either 500 or 650°C for the same duration of time. This yield amounts to 227 and 353% more aromatic than that at 500 and 650°C, respectively. The coke yield on the other hand is highest at 650°C and lowest at 600°C.
In the case of selectivity, 600°C has 64% for aromatic and 34% for coke. Both 500 and 650 have 37 and 22% for aromatic and 60 and 75 % for coke, respectively.
From on the above observations, calcination at 600°C has the highest catalytic activity, selective and yield for aromatic and very much lower for coke yield and selectivity as compared to 500 and 650°C. In other words, 600°C is the best calcination temperature for MDA under the specified conditions.
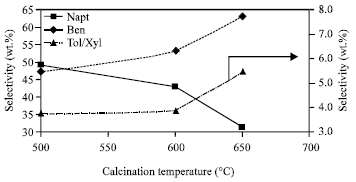
Aromatic product selectivity: Figure 3 represents the effect of calcinations temperature on aromatic product selectivity over 20 h TOS. Benzene and naphthalene represents the major product and the % selectivity is represented on the primary y-axis, while toluene and xylene, the minor product which represent <6 wt. % on the secondary y-axis.
From Fig. 3, it can be seen that both benzene and toluene and xylene group selectivity increases with increase in calcinations temperature, whereas for naphthalene the reverse is true.
Based on the above observation it suggests that both naphthalene and benzene follows a similar intermediate as described by Wang et al. (2003). Increase in calcinations temperature favours benzene selectivity at the expense of naphthalene. From Fig. 2, it was noticed that above calcinations temperature of 600°C, the selectivity for coke increases. From these 2 Fig. 2 and 3, it may indicate that calcinating at 650°C will induce naphthalene condensation resulting to its build up leading to catalytic deactivation. As a result aromatic and naphthalene yield decreases while benzene increases with increase in calcination temperature.
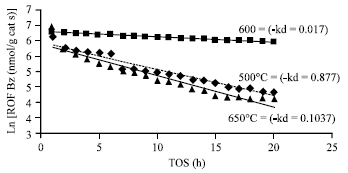
Deactivation rate: The rate of catalytic deactivation was derived by plotting the natural logarithm of rate of formation of benzene against TOS in accordance to Levenspiel (1972) and modified by Wang et al. (2003). The negative gradient gives the rate of deactivation.
 | |
| Fig. 3: | Effect of calcination temperature on aromatic fraction selectivity |
 | |
| Fig. 4: | Rate of deactivation with calcination temperature |
From Fig. 4, it can be seen that the rate of deactivation (kd) is the lowest for calcinations temperature of 600°C and highest for 650°C. This suggests that the catalyst calcined at 650°C undergoes much faster catalytic deactivation than both 500 and 600°C, i.e., 600°C being the more stable catalyst. This is in agreement with all the observations seen in Fig. 2 and 3.
From the above observation it can be suggested that the catalytic activity increases from calcinations temperature 500°C until it reaches a maximum activity at 600°C. As the calcinations temperature approaches 650°C and above the catalytic activity decreases and may probably be destroy thereafter. This is in agreement with findings by Lin et al. (1997) and Xu et al. (1995b).
Surface analysis: Table 1 shows the results from surface analysis of the parent HZSM5 and the effect of calcinations temperature on 10 wt.% Mo modified HZSM5 catalyst. It also shows the changes as the catalyst undergoes from calcinations to activation and finally to the reacted or spent condition after 20 h TOS. The losses in surface properties during these periods as compared to the parent zeolite are shown in brackets.
It can be observed from Table 1, that the BET SA increases with increase in calcination temperature suggesting that spreading of Mo species increases with increasing calcination temperature. This is in agreement with findings by Xu et al. (1995b).
| Table 1: | Effect of calcination temperature on surface properties |
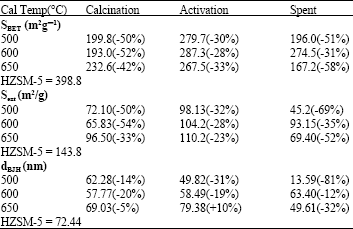 | |
Upon activation from calcination an increase in BET SA is observed for all 3 catalysts. This is expected as the bulky MoO3 species are converted to the smaller and orderly Mo carbide observed by Solymosi et al. (1995) and Wang et al. (1997). From Table 1, the activated 600 and 500°C catalyst has a much higher BET SA than the 650°C. This suggests higher spreading of the small uniform Mo carbide for both 500 and 600°C calcined catalyst, as against 650°C or firmer bonding for the later. Upon reaction 600°C activated catalyst loses less BET SA (3%), than either 500 (21%) and 650°C (25%). This further supports better distribution of the Mo carbide species for 600°C as compared to the other 2. This is apparent from the low carbon deposition observed from catalyst evaluation from Fig. 2.
The same type of effect as BET SA is noticed for the external SA when the catalyst changes from calcinated to activated to reacted. It is interesting to note that catalyst calcined at 600°C has a higher external SA after reaction as compared to those at 500 and 650°C. Since coke is generally deposited on the external surface and in the vicinity of Mo carbide species as described by Xu et al. (1995a), this further suggest the spreading of Mo species at 500 or 650°C is not as highly dispersed as those at 600°C. It may also suggest that calcinations temperature of 500°C is not as efficient as 600°C to well disperse the Mo species.
On examining the calcined catalyst’s pore diameter from Table 1 it can be noticed that all 3 catalyst have lower value than the original HZSM5. This suggests that the pore mouth or channel may be block partially by the migrating Mo species. The calcinated catalyst shows the greatest loss at 600°C and the lowest at 650°C.
On activation the pore diameter of catalyst calcined at 650°C increases more than the HZSM5 zeolite. This may suggests that the pore mouth or channel at 650°C might have been damaged by the excessive Mo species present in the pore channel reacting with the BAS present in the channel on activation or further spreading of the Mo species.
| Table 2: | Effect of calcination temperature on Mo loading |
 | |
As for catalyst at 500°C there is a further decrease in pore diameter suggesting that the blockage at the pore mouth of the catalyst is most probable cause. In the case of 600°C there is a slight increase in pore diameter. This may suggest that probable reduced narrowing of the channel by transformation of the bulky Mo species to smaller Mo carbide species.
When the activated catalysts upon reaction, decrease in pore diameter is observed in all. This is as a result of coke depositions, which are usually associated at the active Mo sites and BAS suggesting heavy carbon deposition at the pore mouth or may be at the pore channels.
Mo content elemental analysis: Table 2 shows the effect of Mo content (measured by ICP-AES) and its changes in temperature (calcinations) and catalyst condition from calcinations to activation and reacted. From same table it can be seen Mo content during calcination treatment results in decrease in Mo content with increase in calcinations temperature. During the calcinations stage, the losses in Mo for both 500 and 600°C are quite similar, but 650°C experience a further loss 5 wt.% more than the others. This may be as a result of increase volatisation of Mo at higher calcinations temperature.
On activation the loss of Mo between the 3 catalysts is between 12-13%. This is expected as explained by to Borry III et al. (1999) and Lin et al. (1997) for more than a monolayer loading for Mo. Further loss of Mo from the reacted catalyst thereafter becomes minimal for all 3 catalyst studied. This indicates a strong bonding between the Mo species and the HZSM zeolite. Further treatment thereafter (activation or MDA reaction) experience minimal Mo loss for all 3 catalysts.
The observation made above further strengthens the findings whereby at higher calcination treatment more Mo species are drawn into the zeolite channels where most of the BAS are found. This is in agreement with Edwars et al. (1990) who reported partial diffusion of Mo species into subsurface regions of the catalyst is highly possible during elevated calcinations temperature conditions.
X- Ray diffraction (powdered): Figure 5 illustrates the XRD patterns for freshly calcined catalyst represented by 10BC500, 10BC600 and 10BC650 is for 500, 600 and 650°C. The arrow head on the XRD pattern represent the MoO3 peak (2θ values at 33.8, 39.0 and 49.3°), Al(MoO4)3 (2θ value at 45.7°) and doublet (2θ at 44 to 47°) are isolated and analyzed further .
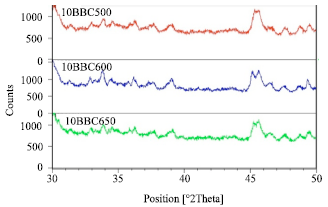 | |
| Fig. 5: | XRD Pattern on Freshly Calcined Catalyst |
From the Fig. 5, it can be seen clearly that MoO3 peaks (2θ values at 33.8, 39.0 and 49.3°)for 10BC600 is much more resolved than either 500 or 650°C. The doublet (2θ = 44 to 47°) split peak resolution for 600°C much better defined than either 500 or 650°C. A well split peak resolution is an indication of retention of the zeolite crystallinity as in the case of 600°C and to a certain extend 650°C. In the case of 500°C the doublet is less resolved and leads or indicates that the zeolite is in the process of changing symmetry from orthorhombic to monoclinic or as a result of sorption of organic molecules or bulky ion occlusion as observed by Borry III et al. (1999) and Liu et al. (2001) could be possible.
Based on calcinations temperature studies for 10MoBM at the set parameters, calcinations at 600°C is the preferred temperature for MDA reaction as it has the highest catalytic activity and produces the highest yield and selectivity towards aromatic and lowest coke yield and selectivity. Further it is most stable catalyst to those studied.
The author would like to thank Universiti Teknologi PETRONAS for their sponsorship. Not to forget the technicians, colleagues and others who were directly or indirectly involved in the project.