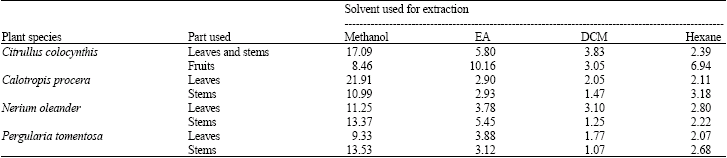
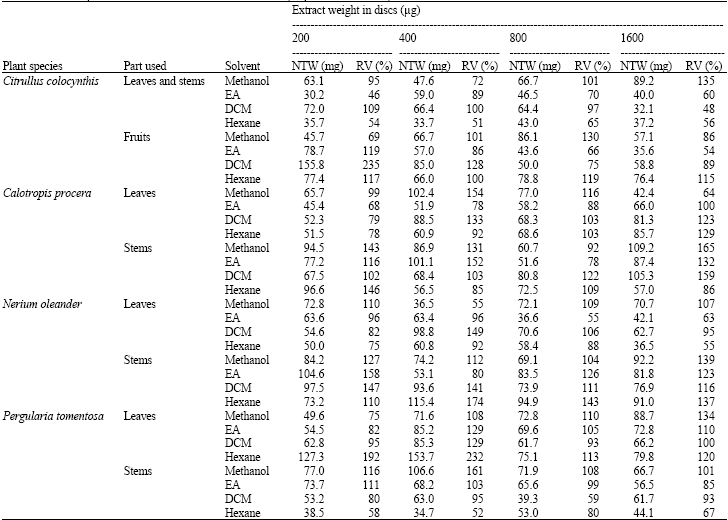
Research Article
Effect of Some Poisonous Plants Extracts on Fusarium oxysporum f. sp. albedinis
Phytochemistry and Organic Synthesis Laboratory, Bechar University, Bechar 08000, Algeria
A. Marouf
Laboratory of Plant Biochemistry and Natural Substances, Oran University, Oran 31000, Algeria
A. Cheriti
Phytochemistry and Organic Synthesis Laboratory, Bechar University, Bechar 08000, Algeria