Research Article
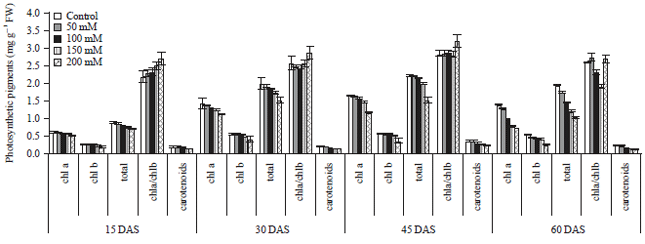
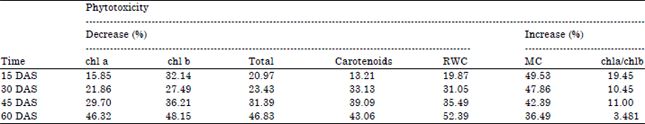
Effect of Salt Stress on Growth Parameters, Moisture Content, Relative Water Content and Photosynthetic Pigments of Fenugreek Variety RMt-1
Department of Biotechnology, Kumaun University, Bhimtal Campus, Bhimtal, 263136, Uttrakhand, India
Veena Pande
Department of Biotechnology, Kumaun University, Bhimtal Campus, Bhimtal, 263136, Uttrakhand, India