Research Article
Production of α Amylase by Flocculant Yeast
Department of Biology, Faculty of Sciences, University of Isfahan, Isfahan, Iran
Giti Emtiazi
Department of Biology, Faculty of Sciences, University of Isfahan, Isfahan, Iran
Industrial Saccharomyces cervisiae are unable to metabolize starch, a relatively cheap carbon source. The induction of such a capability is a marked improvement in both the baking and brewing industry as well as in producing ethanol, maltose syrups or single cell protein (scp) (Marin et al., 2001).
α-amylase (1, 4-α-D-glucan glucanohydrolase, EC 3.2.1.1) catalyzes the endoamylolytic cleavage of α-1,4-glucosidie linkages of starch and similar substrates releasing maltose, oligosaccharides and limited dextrins. Typically commercial amylases are crude preparations obtained from species of the filamentous fungus Aspergillus. These enzymes are commonly added to flour to enhance baked products. However, their use increases the cost of the final product and those preparations act as potent allergens capable of sensitizing many of the workers who are exposed to them (Alvarez et al., 1996).
Previously, the α-amylase and glucoamylase genes from Aspergillus oryzae were used to construct a starch-utilizing baker’s yeast. α-Amylase cDNA was introduced into baker’s yeast via the episomal plasmid (Yep) (Randez et al., 1995) or by employing an integrative cassette targeted to the ribosomal rDNA locus (Nieto et al.,1999) Glucoamylase cDNA has also been introduced in to baker’s yeast targeted to the δ-sequence of the yeast retrotransposon Ty1 (Saito et al., 1996)
Shigechi et al. improved ethanol productivity from starchy materials by constructing two recombinant yeast strains co-expressing glucoamylase and α-amylase. Plasmids for cell surface expression and for secretory expression of Bacillus stearothermaphilus α-amylase were constructed and cotransformed into the flocculent yeast strain YF207 along with the plasmid for cell-surface expression of R. oryzae glucoamylase (Hisayori et al., 2002). In this work, 3 flocculant yeasts were isolated from saliva and the production of α-amylase and utilization of starch were investigated.
Isolation and media: Yeast were isolated from Saliva and the isolated yeasts were grown in the following medium (gl–1): Starch 10, KH2PO4, 2; MgSO4 0.04, 0.004; yeast extract or (NH4)2SO4 pH was adjusted to 6.0. When necessary, media were solidified by the addition of 2.5% agar. The media was incubated at 25°C in a flask on α shaker.
Enzyme assay: a-amylase was estimated from reducing sugar liberation in 2 ml reaction mixtures containing 1% soluble starch. 0.1 ml phosphate buffer pH 7.0 and 0.5 ml cell free culture fluid was incubated for 30 min at 37°C (Busch and Stutzenbtger, 1997). One unit of the enzyme activity descripted as 1mmol maltose was released per h under the assay condition (25°C pH 7).
Starch hydrolyzed test: Starch hydrolysis was evaluated in liquid medium by using 50 ml of culture supernatant as a loss of Iodine (0.5% (w/v) KI, 0.15% (w/v) I2) staining capacity (Abarca et al., 1989). Measurement of flocculation ability (Callejia and Johnson, 1976).
Estimation by visual observation: Flocculation was estimated by eye. This consisted of viewing the sides and bottom of the culture flasks against the ceiling light. Then expressed as (-), when no gross floc was observed, (+) for a culture which showed any flocs at all and (+) followed by some number from 1-4 for highest degree of flocculation.
Turbidimetric method: Culture cells were harvested by centrifugation, washed four times in 10 mM EDTA, followed by washing the cells three times with deionized water. Cell suspension in acetate buffer (pH 4.5) was distributed in tubes. Flocculation was determined by measuring the absorbance of the suspension at 660 nm.
Growth rate: Growth rate was determined by measuring the absorbance of the suspension at 600 mm.
Six yeast were isolated on starch agar and 3 of them were starch positive with Iodine Fig. 1. The production of α amylase and the growth rate were investigated in the media with 1% starch as the only source of carbon and the media with 1% starch and yeast extract or ammonium as nitrogen as is shown in Fig. 1- 4. Although the B strain has maximum α-amylase activity, strain E used 1% starch in 17 days. However, strain B only used 50% of this starch in 17 days. The strain E had maximum α amylase activity when 10 ml of the saliva were added to starch media (Fig. 5).
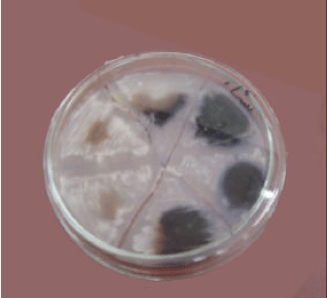 | |
| Fig. 1: | Isolation of six yeasts on starch agar, three of them were positive with Iodine |
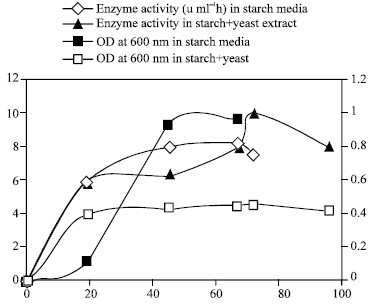 | |
| Fig. 2: | Production of amalayse and growth rate of Strain F |
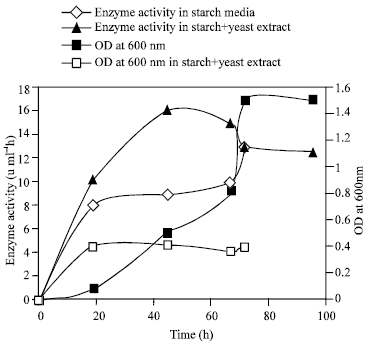 | |
| Fig. 3: | Production of amylase and growth rate of yeast strain B |
As is shown in Fig. 5, strain E has maximum 28 unit –amylase activity. Enzyme of strain E was stable at 25, 37 and 60°C, however, strain B was stable at 25°C (Fig. 6).
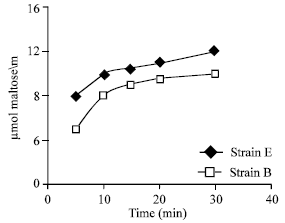
As is shown in Fig. 6, the α-amylase activities in strain B was reduced by addition of temperature, however, the enzyme activities of strain E were stable at 25 to 60°C. The production of maltose (μmol ml–1) by strain E and B are shown in Fig. 7. As is shown, the sugar was released from starch by the enzyme of strain E and B.
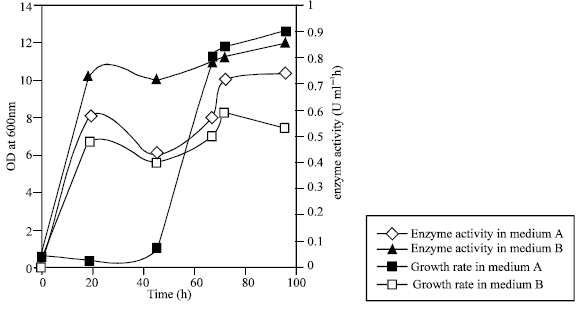 | |
| Fig. 4: | Production of amylase and growth rate of strain (E). A=starch media, B=Starch+yeast extract |
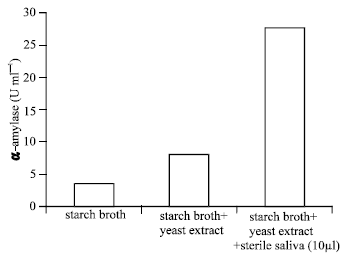 | |
| Fig. 5: | The effect of growth factor on maximum α-amylase production by strain E. One unit is enzyme activity which releases 1 μmol of reducing equivalents (assayed against a maltose standard) per h under the assay condition (25°C, pH 7.0) |
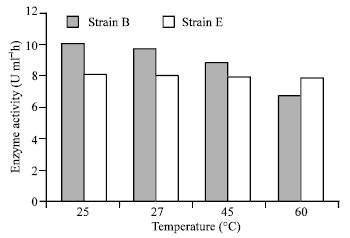 | |
| Fig. 6: | The effect of temperature on enzyme |
 | |
| Fig. 7: | Production of maltose by amylase from strain E and B |
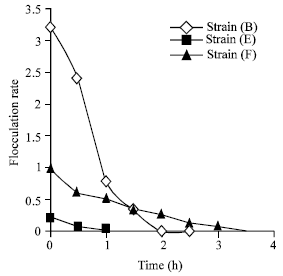
 | |
| Fig. 8: | The rate of flocculation after maximum amylase production. The measurement was according to the decreases of OD at 600 nm by time |
The E strain was the best flocculant yeast (Fig. 8). As is shown in Fig. 8, the entire cells have settled down in one h.