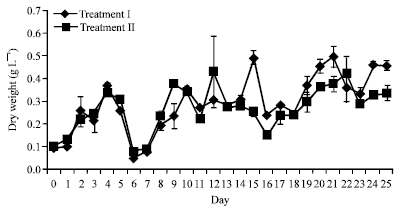
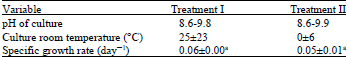Research Article
Large-scale Culture of a Tropical Marine Microalga Chaetoceros calcitrans (Paulsen) Takano 1968 at Different Temperatures Using Annular Photobioreactors
Laboratory of Marine Biotechnology, Institute of Bioscience, Universiti Putra Malaysia, 43400, Serdang, Selangor, Malaysia
F.M. Yusoff
Laboratory of Marine Biotechnology, Institute of Bioscience, Universiti Putra Malaysia, 43400, Serdang, Selangor, Malaysia
M. Shariff
Laboratory of Marine Biotechnology, Institute of Bioscience, Universiti Putra Malaysia, 43400, Serdang, Selangor, Malaysia