ABSTRACT
The pregnancy complicated by uncontrolled maternal diabetes affected the maternal health and the placenta experiences a diversity of structural and functional changes. The study aims to explore the possible adverse effects of Gestational Diabetes Mellitus (GDM) on the morphometric of umbilical cord and its vessels. The study carried on fifty placentas with its cords of normal pregnancy were without any known pre-existing disease and 50 placentas with its cords from mothers complicated by GDM (untreated). Gestational diabetes mellitus group was selected from Gynecology and Obstetrics Department of AL-Hilla Teaching Hospital, Iraq. The gross examination showed that the number of cords with Single Umbilical Artery (SUA) was 13 out of 50 cords in pregnant women with gestational diabetes compared with normal pregnancy. All the histomorphmetric parameters of cord with SUA as the circumference, diameters, cross-section area of cord, CSA of whrton’s jelly and histomorphometric umbilical vessels of SUA and vein of GDM group were decreased when compared to the normal group. The GDM has adverse perinatal outcome as respiratory distress, cardiovascular anomalies and still birth. It can be concluded that the lean cord with single umbilical artery were related with GDM and associated with adverse fetal outcome.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2015.346.351
URL: https://scialert.net/abstract/?doi=pjbs.2015.346.351
INTRODUCTION
Pathological variations in placenta in maternal diabetes are the chief cause for reduced placental functions; here after they can consequence in increased fetal complications (Nagi, 2011). Diabetes Mellitus (DM) in pregnant women may be categorized into clinical diabetes or pregestational diabetes (women previously diagnosed with type 1 or type 2 diabetes) and gestational diabetes (GDM). Which is stated as any degree of glucose intolerance with commencement or first recognition during pregnancy (Forsbach-Sanchez et al., 2005; American Diabetes Association, 2009). Late pregnancy complications of uncontrolled diabetes effect in neonatal morbidity and mortality. A number of epidemiological studies show that despite good glycemic control the number of congenital malformations in diabetic mothers is still higher than in non-diabetics (Evers et al., 2004). Fetal macrosomia, congenital malformations and intrauterine growth retardation are commonly seen in poorly controlled diabetes (Verma et al., 2010). The umbilical cord is hereditarily a part of the fetus and normally contains two artery one vein buried with wharton’s jelly (Heifetz, 1984). The reported occurrence SUA was 5.9% in the first trimester of pregnancy (at 11-14 weeks gestation) (Rembouskos et al., 2003). The most frequent abnormality of the umbilical cord is a single umbilical artery that has an incidence of 0.2-2.0% of all pregnancies (Lubusky et al., 2007). The single umbilical cord SUA is believed to be caused by atrophy of a previously normal artery, the presence of the original artery of the body stalk. The intrauterine development of fetus is dependent on one vital organ "The placenta" umbilical cord connects the fetus with fetal surface of placenta that attaches to uterine endometrium. So, umbilical cord anomalies fiercely affect the fetal outcome (Kinare, 2008). The SUA is related to other fetal anomalies, the corporated anomalies are genitourinary followed by cardiovascular malformation (Gornall et al., 2003). The alterations in the vascular tree in placentas from pregnancies complicated by diabetes mellitus and preeclampsia are not well defined Maly et al. (2005). The aim of the present study was to determine whether, the GDM related to single umbilical artery and the effects of GDM on measurements of umbilical cord and vessels.
MATERIALS AND METHODS
Patients: Fifty mother’s placenta of normal pregnancy without any known pre-existing disease, or its complications and fifty mothers with diabetic pregnant female (uncontrolled by insulin) were selected from indoor patients of the Department of Gynecology and Obstetrics, Al-Hilla Teaching Hospital. The age of these mothers ranges between 20 and 38 years. Every patient had undergone the investigations, such as, plasma glucose and glycoslyted heamoglobin (HbAIc). The women in the study formally consented to sample collection and the study goals.
Exclusion and inclusion criteria: The multiple pregnancy and overt diabetes have been excluded. The women with GDM diagnosed by a specialized physician during the late days before delivery (they did not know that had suffered from GDM and they do not attend the pregnancy care unit at the primary health center).
Morphometric examinations: After delivery, umbilical cords were tied and cut 5 cm away from placental end. The umbilical cord with the placenta was collected within 37-40 weeks of gestation from selected hospital. Systematic random samples of umbilical cord taken for this study were given as:
| • | The mean diameter of the umbilical cords was measured by vernier calipers [(Maximum diameter+minimum diameter)/2] in millimeters (Gill and Jarjoura, 1993) |
| • | Circumference and Cross-Section Area (CSA) of the umbilical cord were measured in millimeters and sq. millimeters |
| • | CSA of the umbilical vessels was measured in sq. millimeters |
| • | Thickness of vessel wall measurements expressed the whole thickness of the vessel wall from endothelium to wharton’s jelly by microscope equipped with a scale |
| • | The CSA of the Wharton's jelly was calculated in sq. millimeters by subtracting the measured total CSA of umbilical vessels from the measured CSA of the umbilical cord (Togni et al., 2007) |
Light microscopy: The tissues were taken from the umbilical cord at placental junction and cut end to determine the incidence of single umbilical cord in fetuses. A 2 cm wedge of tissue was taken and fixed in 10% formalin for one week. The tissue was dehydrated and followed by embedding in paraffin and 7 micron serial sections were generated with the help of rotator microtome. The tissue sections were stained with hematoxylen and eosin. Histological appearance of the umbilical cord was assessed and various histomorphometric parameters were measured.
Vascular corrosion casting: Cast models of the cord and placental vessels were created from full-term placentas by the corrosion casting technique that was used similar to the before published protocols (Mu et al., 2001). The placentas were treated within 5 min after delivery. The data obtained from the study was compiled and expressed as mean and standard deviation. The p value (p<0.05) was taken as significant.
RESULTS
Figure 1a shows the normal insertion (centric insertion) in normal group, while the Fig. 1b, displays abnormal insertion (marginal insertion) in GDM group with SUA. As shown in Fig. 2a-d the casts of the vasculature of full-term placentas provided information about the morphological criteria of branching umbilical, chorionic blood vessels with its arborization.
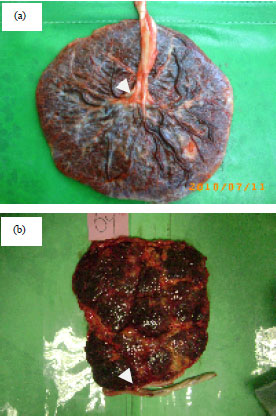 | |
| Fig. 1(a-b): | (a) Central insertion (control) and (b) Marginal insertion with SUA of GDM |
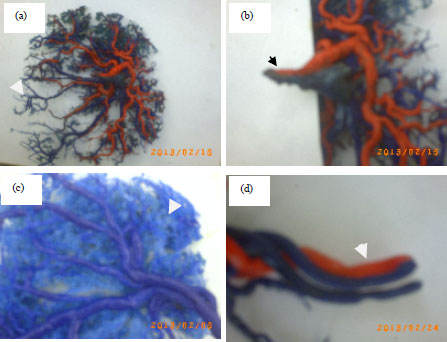 | |
| Fig. 2(a-d): | (a) Cast section blood vessels of placenta with SUA of GDM group shows decrease in arborization of placenta blood vessels, (b) Main branching of umbilical blood vessels (single artery and one vein)of GDM group, (c) Cast section of normal group shows normal arborization of blood vessels and (d) Main branching of umbilical blood vessels (two arteries and one vein) of normal group |
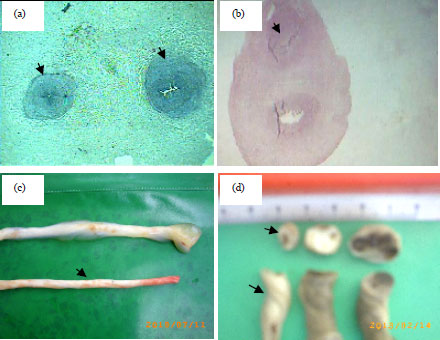 | |
| Fig. 3(a-d): | Comparison of lean umbilical cord of GDM group with normal cord (a) Cross section of normal umbilical cord show two umbilical arteries, (b) SUA with one vein (full section), (c) Lower cord with SUA of GDM group shows lean umbilical cord in comparison with upper normal cord and (d) Left cord with SUA shows the cross section area of lean cord less than 1 cm in comparison with cords without SUA |
Figure 3a showed smaller Cross-Section Area (CSA) of umbilical cord under (4x) magnification completely appeared with SUA and vein of GDM group while, the whole cross-section area of normal umbilical cord not appeared, because the normal umbilical cord is larger as in Fig. 3b. As revealed in Fig. 3d there was a differences in size, circumference and diameter between lean cord with SUA of GDM (left), normal and umbilical cords of GDM without SUA (blood clots in vessels in the right one of this figure). Figure 4a and b 10x shows the CSA of normal artery with its normal number of the folds.
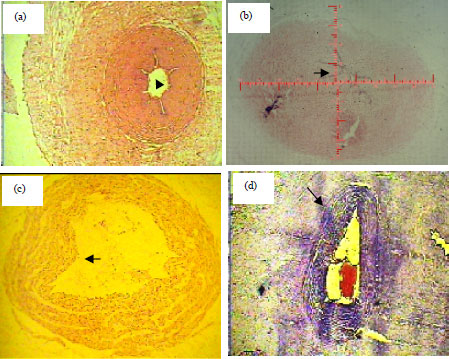 | |
| Fig. 4(a-d): | (a) Cross section of normal artery(4x)shows normal six folds, (b) Normal cord shows scale, (c) Cross section of SUA show one fold, splitting of internal elastic lamina and sloughing of endothelial layer and (d) Normal vein of normal group |
| Table 1: | Histomorphometric variables of the umbilical cord in the two groups |
 | |
| *p<0.05 was considered as the level of significance | |
The study concludes that GDM causes pathological and structural changes in SUA including splitting of internal elastic lamina, sloughing of endothelial layer, the wall thickness of the SUA (from endothelium to wharton’s jelly) thinner than the wall thickness of normal artery and reduces the number of folds as shows Fig. 4c. The histomorphometric parameters of normal umbilical arteries compared to single umbilical artery represented in Table 1, which showed that the mean cord area was (51.55±2.32 mm2 vs 40.04±2.1 mm2). The jelly area was (42.26±2.11 mm2 vs 30.11±1.81 mm2). In arteries total area of artery-1 was (2, 08±0.17 mm2 vs 1.99±0.30 mm2), total luminal area of artery-1 (0, 36±0.08 mm2 0.25±0.09 mm2), wall thickness of artery-1 was (487.93±26.62 μm vs 350.90±24.60 μm). The artery-2 was absent in umbilical cord with SUA. The mean number of Hoboken folds, or valves were 4.50±21.52 vs 2.40±20.42. The outcome in the GDM group was 10 (40%) with cardiovascular, still birth was 6 (24%) and 16 (64%), respiratory distress.
DISCUSSION
The umbilical cord connected the fetus and the placenta, any abnormality in the cord may have harmful effects on the life of the fetus. The present study concentrated on the study of histological and morphological alterations of the umbilical cord in GDM, which may lead to the possibility of complication with adverse effects on both the placenta and fetus. The intrauterine development of the fetus depends on the placental vascular system. In the current study, SUA was more frequent in GDM group, concerning the pathogenic mechanism, it is thought that primary agenesis of one umbilical artery are the most frequent mechanism. The SUA was considered to be a developmental umbilical cord accident associated with disturbance of fetal blood flow. Changes in the structures of placenta, using morphological criteria of villous bifurcating are divisible into diverse topological regions of placenta based on villous capacity and vessel form (Mayhew et al., 2004). It is mostly agreed that the normal placenta shape at delivery is ovoid with centric insertion (Jauniaux et al., 2003; Chaddha et al., 2004; Salafia and Maas, 2005). Observations the placenta of the mothers of GDM groups with lean cord and SUA recorded that the insertion of umbilical cord becomes more marginal in nature. This was come in agreement with Verma et al. (2010), who studied the GDM but they don’t study SUA. In our study marginal insertion found among pregnant women with GDM, other reports declar that marginal insertion related with the mother of hypertensive group (Majumdar et al., 2005) and low birth weight (Rath et al., 2000) but they don’t study SUA. The resin cast considered the most suitable casting medium for the umbilical cord vessels, due to its low viscosity, rapid and evenly polymerization and minimal shrinkage during hardening that allows the infusion of small vessels, including the capillaries. The lean cord with SUA of GDM group due to missing area of the second artery and deficiency of the wharton’s jelly due to lack of glycogen in the cord tissues. This may be resulted from mucous connective tissue degeneration, which may reflect the effects of maternal diabetes on the wharton’s jelly. Another study by Horn et al. (2006) reported the link of hypercoiling and thinning with consecutive constriction of the umbilical vessels (thin cord syndrome; TCS) and intrauterine fetal death. They found that there is an obvious difference in the frequency or TCS in the spontaneous abortion group and the TCS was the causative of intrauterine demise in 9% of cases.
The present histomorphometric study (Table 1) shows significant decrease (p<0.05) in wharton’s jelly area that is supported by biochemical studies. It is mentioned that the sulphated glycosaminoglycan (SAGs) accumulated in the outer matrix of wharton’s jelly have an effect on the biology of the cord tissue (Bankowski et al., 1996). In this study, wall thickness luminal area and total umbilical vein area were decreased compared to normal vein, such observation may be due to hypoplastic effect through different factors. Inan et al. (2002) mentioned a decrease in wall thickness of vein, a total vein area with a narrow lumen of the vein, if abnormal changes in doppler umbilical flow waveform in pre-ecliptic pregnancies. The total umbilical artery wall thickness and fold’s of SUA significantly decreased p<0.05 than normal cord artery such change may be due to decrease in both tunica intima and media. The GDM causes pathological changes in tunica intima including splitting of internal elastic lamina and sloughing of endothelial layer and the tunica media, include reducing in the number of smooth muscle fiber and folds. These findings may be attributed to hypoplasia in smooth muscle fiber, which lead to a reduction in the folds number of SUA. In contrast with Junek et al. (2000) who, found 15% increased thickness of arterial wall in pre-eclamptic group as compared to control group. Our study display the mean of diameter, circumference and the Cross Section Area (CSA) of the umbilical cord in the GDM with SUA group was decreased than in control group due to decrease of wharton’s jelly, missing the one artery and decrease all the previous measurement criteria. Ghezzi et al. (2001) found strong correlation between CSA of umbilical cord and the CSA of the wharton’s jelly (r = 0.97, p<0.001) but he was study GDM without SUA. Luoto et al. (2011) mentioned that there was a risk of adverse outcomes as macrosomia, caesarean sections, respiratory distress associated with maternal glucose intolerance. Hadar and Hallak (2003) mentioned that GDM is associated with stillbirth. Further investigation is needed to confirm these findings. To the best of our knowledge, we are the first to demonstrate the measurements of umbilical cord with single umbilical artery in pregnancies complicated by untreated GDM.
CONCLUSION
The results confirmed the possibilities of reflecting effects of GDM on missing of one umbilical artery (SUA) and lean umbilical cord. We recommended the systematic doppler with scanning for the numeral of umbilical arteries in the 20th week of gestation as a good investigation method. The finding of an SUA should attentive the ultra-sonographer to examination for related malformations and signs of chromosomal deficiencies (trisomy 18), such as cardiac abnormalities, overlapping fingers, spina bifida and facial cleft at the16-22 week scan.
ACKNOWLEDGMENTS
Thanks to all the clinicians, staff of labor and delivery room and patients for their cooperation in the study.
REFERENCES
- American Diabetes Association, 2009. Diagnosis and classification of diabetes mellitus. Diabetes Care, 32: S62-S67.
CrossRefDirect Link - Bankowski, E., K. Sobolewski, L. Romanowicz, L. Chyczewski and S. Jaworski, 1996. Collagen and glycosaminoglycans of Wharton's jelly and their alterations in EPH-gestosis. Eur. J. Obstet. Gnaecol. Reprod. Biol., 66: 109-117.
CrossRefPubMedDirect Link - Chaddha, V., S. Viero, B. Huppertz and J. Kingdom, 2004. Developmental biology of the placenta and the origins of placental insufficiency. Semin. Fetal Neonatal Med., 9: 357-369.
CrossRefDirect Link - Evers, I.M., H.W. Valk and G.H.A. Visser, 2004. Risk of complications of pregnancy in women with type 1 diabetes: nationwide prospective study in the Netherlands. BMJ, Vol., 328.
CrossRefDirect Link - Ghezzi, F., L. Raio, E. di Naro, M. Franchi, D. Balestreri and V. D'Addario, 2001. Nomogram of Wharton's jelly as depicted in the sonographic cross section of the umbilical cord. Ultrasound Obstet. Gynecol., 18: 121-125.
CrossRefDirect Link - Gill, P. and D. Jarjoura, 1993. Wharton's jelly in the umbilical cord. A study of its quantitative variations and clinical correlates. J. Reprod. Med., 38: 611-614.
PubMedDirect Link - Gornall, A.S., J.J. Kurinczuk and J.C. Konje, 2003. Antenatal detection of a single umbilical artery: Does it matter? Prenat. Diagn., 23: 117-123.
CrossRefPubMedDirect Link - Hadar, A. and M. Hallak, 2003. Single umbilical artery and umbilical cord torsion leading to fetal death. A case report. J. Reprod. Med., 48: 739-740.
PubMedDirect Link - Heifetz, S.A., 1984. Single umbilical artery. A statistical analysis of 237 autopsy cases and review of the literature. Perspect. Pediatr. Pathol., 8: 345-378.
PubMedDirect Link - Horn, L.C., R. Faber, H. Stepan, E. Simon, R. Robel and C. Wittekind, 2006. Umbilical cord hypercoiling and thinning: A rare cause of intrauterine death in the second trimester of pregnancy. Pediatr. Dev. Pathol., 9: 20-24.
CrossRefPubMedDirect Link - Inan, S., M. Sanci, D. Can, S. Vatansever, O. Oztekin and S. Tinar, 2002. Comparative morphological differences between umbilical cords from chronic hypertensive and preeclamptic pregnancies. Acta Medica Okayama, 56: 177-186.
PubMedDirect Link - Jauniaux, E., J. Hempstock, N. Greenwold and G.J. Burton, 2003. Trophoblastic oxidative stress in relation to temporal and regional differences in maternal placental blood flow in normal and abnormal early pregnancies. Am. J. Pathol., 162: 115-125.
CrossRefDirect Link - Junek, T., O. Baum, H. Lauter, K. Vetter, D. Matejevic and R. Graf, 2000. Pre-eclampsia associated alterations of the elastic fibre system in umbilical cord vessels. Anatomy Embryol., 201: 291-303.
CrossRefDirect Link - Mayhew, T.M., D.S. Charnock-Jones and P. Kaufmann, 2004. Aspects of human fetoplacental vasculogenesis and angiogenesis. III. Changes in complicated pregnancies. Placenta, 25: 127-139.
CrossRefDirect Link - Luoto, R., T.I. Kinnunen, M. Aittasalo, P. Kolu and J. Raitanen et al., 2011. Primary prevention of gestational diabetes mellitus and large-for-gestational-age newborns by lifestyle counseling: A cluster-randomized controlled trial. PLoS Med., Vol. 8.
CrossRef - Majumdar, S., H. Dasgupta, K. Bhattacharya and A. Bhattacharya, 2005. A study of placenta in normal and hypertensive pregnancies. J. Anat. Soc. India, 54: 34-38.
Direct Link - Maly, A., G. Goshen, J. Sela, A. Pinelis, M. Stark and B. Maly, 2005. Histomorphometric study of placental villi vascular volume in toxemia and diabetes. Hum. Pathol., 36: 1074-1079.
CrossRefDirect Link - Lubusky, M., I. Dhaifalah, M. Prochazka, J. Hyjanek, I. Mickova, K. Vomackova and J. Santavy, 2007. Single umbilical artery and its siding in the second trimester of pregnancy: Relation to chromosomal defects. Prenat. Diagn., 27: 327-331.
CrossRefDirect Link - Mu, J., T. Kanzaki, T. Tomimatsu, H. Fukuda and E. Fujii et al., 2001. A comparative study of intraplacental villous arteries by latex cast model in vitro and color doppler flow imaging in vivo. J. Obstet. Gynaecol. Res., 27: 297-303.
CrossRefDirect Link - Rath, G., K. Garg and M. Sood, 2000. Insertion of umbilical cord on the placenta in hypertensive mothers. J. Anat. Soc. India, 49: 149-154.
Direct Link - Rembouskos, G., S. Cicero, D. Longo, C. Sacchini and K.H. Nicolaides, 2003. Single umbilical artery at 11-14 weeks' gestation: Relation to chromosomal defects. Ultrasound Obstet. Gynecol., 22: 567-570.
CrossRefDirect Link - Salafia, C.M. and E. Maas, 2005. The twin placenta: Framework for gross analysis in fetal origins of adult disease initiatives. Paediatr. Perinat. Epidemiol., 19: 23-31.
CrossRefDirect Link - Verma, R., S. Mishra and J.M. Kaul, 2010. Cellular changes in the placenta in pregnancies complicated with diabetes. Int. J. Morphol., 28: 259-264.
Direct Link - Togni, F.A., E. Araujo Junior, F.A.P. Vasques, A.F. Moron, M.R. Torloni and L.M.M. Nardozza, 2007. The cross-sectional area of umbilical cord components in normal pregnancy. Int. J. Gynecol. Obstetr., 96: 156-161.
CrossRefPubMedDirect Link








