Research Article
Utilization of Some Edge-Row Plants as Forage in Nigeria
Department of Animal Science, University of Ibadan, Ibadan, Nigeria
O. J. Babayemi
Department of Animal Science, University of Ibadan, Ibadan, Nigeria
Environmental beautification is gaining attention in rural and urban centers of Nigeria, resulting to persistent planting of ornamental and herbal trees around the residential quarters as edge-rows. In order to prevent such trees and shrubs from growing wild, some branches are often selectively pruned down by the gardeners. Such pruned brush-woods are left under the tree or are allowed to dry and later set ablaze, thereby littering the compound and polluting the entire environment. The rising trend in tree planting can be mutual and beneficial to both small ruminants and the environment at large. Leaf of the trees can be used to feed goats and sheep as protein supplements all year round while the wastes accruing from the animals can as well be used as organic manure for the trees. Tropical trees are highly nutritive and rich sources of protein and minerals (Teferedegne, 2000; Babayemi et al., 2004).
Neem, almond, mango and bitter leaf plants are among the common trees used as ornamental trees as they are well adapted to varying ecological and edaphic features in Nigeria. Mango (Mangifera indica) belongs to the family Anacardiaceae. It is a cultivated plant and the fruit is used as food for human. Mango has a feeding value as fodder for livestock (Isah et al., 1999). It can be used as a standing feed reserve so that livestock can survive the critical period of feed scarcity during the dry season.
Almond (Terminalia catappa) commonly called is a tree that belongs to the family combretaceae. It is a dicotyledonous plant whose fruit is eaten by human. It is a medium-sized genus of tree and shrubs with leaves densely clustered at the end of the branches. Information on Terminalia catappa as livestock feed is limited due to unawareness of its feed potentials and its usefulness in augmenting feed supply of livestock especially in dry season (Babayemi, 2007).
Neem (Azadirachta indica) belongs to the family Meliaceae. It is used as shade tree. There is scanty documentation on the use of neem leaf as feed for livestock. Livestock acceptability of the leaves could be low as this may be due to the acerbic taste and the presence of tannin in it (Babayemi and Bamikole, 2006).
Bitter leaf (Vernonia amygdalina) is a shrub with petiole leaf of about 6mm diameter, green with a characteristic odour and bitter taste, grows under a range of ecological zones in Africa, produces large mass of forage and it is drought tolerant (Bonsi et al., 1995). The leaves are used for human consumption, suggesting that the excess may be utilized as feed.
As the forages differ in chemical composition and secondary metabolites (Babayemi et al., 2004), quick and precise assessment means are therefore, needed to ensure that all materials are properly evaluated before being fed to livestock. The present study was planned to assess the nutrient composition and degradability using in vitro gas methods of neem, mango, almond and bitter leaf in Nigeria.
Collection of feed samples: Two hundred and fifty (250) grams of Neem, almond, mango and bitter leaf were obtained from twenty tree stands each at University of Ibadan campus and its environs in October 2007. Samples of the plants were bulked differently and from the composite, 200 g each was sub-sampled. The samples were oven dried at 105°C until a constant weight was ascertained. The dried samples were milled (1 mm sieve) and kept in cellophane bag pending the laboratory analysis.
Qualitative determination of secondary metabolites: Presence of secondary metabolites was determined qualitatively as described by Larrahondo (1985), Rosales et al. (1989) and as modified by Babayemi et al. (2004). An extract was prepared and colour changes observed following the addition of various reagents, giving an indication of the presence of phenols, steroids and saponins. Briefly, 2 g each of the dry matter of the samples were weighed into extraction bottles. 30 mL of petroleum ether and 30 mL of a 9:1 methanol-water solution was added into the extraction bottles each. The bottles were then placed on a mechanical shaker for the extraction of the samples by revolving at 160 rpm for 90 min. The resulting mixture was then filtered and placed in a separating funnel. The residue was washed with petroleum ether and methanol water to dilute to 50 mL methanol water and 50 mL petroleum ether. It was left to stand until two separate layers can be identified. The lower layer was the methanol-water polar fraction; the top non-polar fraction being formed was the petroleum ether. The two layers were later separated into containers and sealed properly to prevent evaporation. 9 mL of distilled water was added to 1.67 mL of the methanol fraction. 1 mL of this solution was shaken in a small test tube for 30 sec After 15 minutes; the height of foam in the tube was measured, giving an indication of the level of saponin in the samples. The following height of foam was used to determine the levels of saponin: 5 mm or less = Negligible; 5-9 mm = low content of saponins; 10-14 mm = Medium content of saponins; 15 mm or more = sample high in saponins. From the methanol-water fraction, 1 mL was pipette into a test tube with a prepared solution of 1% FeCl3 (w/v) at a level of 1 mL was dispensed into the test tube. The following parameters were used for scoring: No change = No phenols or tannins; Dark blue = water soluble tannins or phenols; Dark green = flavonoids or condensed tannins. Ten (10) mL of the non-polar fraction in a test tube was evaporated in a water bath to concentrate the solution. 0.5 mL of chloroform, 0.25 mL acetic anhydride and 0.125 mL concentrated sulphuric acid was added. Changes in colour indicated the presence of steroids as follows: Blue or green = steroids; Red, pink or purple = Triterpenoids; Light yellow = saturated steroids or triterpenoids.
In vitro gas production procedure: The residues of the plants obtained from qualitative determination of secondary metabolites were respectively oven dried to a constant weight and then prepared for in vitro gas production with whole samples (not extracted). In vitro gas production was carried out as described by Menke and Steingass (1988). 200 mg DM each of the samples were weighed and introduced carefully into the cylinder of the syringe. Each of the samples was replicated three times. The blank which contained no substrate was also prepared. The piston (greased with vaseline to ensure easy movement and precise fitting) was then pushed down the cylinder gently to avoid thrusting out of the sample through the cylinder tube. The silicon rubber tube attached to the capillary attachment (needle) of the syringe was closed with the steel clip.
Rumen liquor was collected prior to the early morning feeding of the goats. The goats were previously fed with 60% concentrate (40% corn, 10% wheat offal, 10% palm kernel cake, 20% groundnut cake, 5% soybean meal, 10% dried brewers grain, 1% common salt, 3.75% oyster shell and 0.25% fish meal) and 40% Gliricidia sepium. The rumen fluid (about 120 mL) in equal proportions was collected through stomach tube by suction method from three West African dwarf goats and was bulked into a thermos flask. The rumen fluid was then taken to the laboratory and filtered through four layer cheese cloth into a warm flask (kept in a bucket of water at 37-39°C). The liquor was maintained by flushing with stream of CO2 and gradually stirred using an automatic stirrer.
The McDougall’s buffer (g/L) consisted of NaHCO3 (9.8), Na2HPO4 (2.77), KCl (0.57), NaCl (0.47), MgSO4.7H2O (0.12) and CaCl2.2H2O (0.16) was prepared. The rumen liquor-buffer solution was mixed in the ratio 1:4 (v/v). The plastic syringes containing the substrates were placed in a water bath at 38-39°C 1 h before incubation started. The steel clip on the syringe was loosened gently to allow for inoculation of the syringes. 30 mL of the rumen fluid-buffer mixture was pipette with a syringe into each of the pre-warmed plastic syringes. This was followed by drawing most of the air from the syringes. The syringes were shaken gently to make sure that the substrate is mixed with the liquid and that the remaining air bubbles was taken out from the syringes. The zero time (i.e. the time when injection of the rumen/buffer mixture into the syringes and placement into the incubator) of the incubation was recorded. The level of the piston of the syringes (which was about 30.0 mL) and the time when the filling of the syringe with the rumen liquor/buffer mixture was finished were also recorded. The syringe was then incubated at 39 + 1°C in an incubator. The blanks contained only the 30 mL of the rumen-buffer solution.
The incubation lasted for 24 h and the accumulated gas was read every 4 h interval. The gas produced was read by measuring the space formed between top of the piston and the liquid in the syringe. After each reading, the content in the syringe was shaken properly to allow proper mixing of the substrate. About 10 molar NaOH was prepared and introduced into the syringe after the end of the 24 h incubation. 4 mL of the 10 M NaOH solution was introduced into each of the incubated syringes through the silicon tube fixed to the capillary attachment after opening the steel clip. After the absorption of CO2, the clip was closed again and the level of methane (CH4) was read.
Chemical composition analysis: The CP, CF, EE, ash contents of the whole plants and CP ash of the extracted ones were also determined according to AOAC (1990). Crude protein analysis was by the process of Kjeldahl. It was effected through the breaking down of 2 g sample (n = 2) in 25 mL concentrated H2SO4 plus selenium, using Gerhardt Kjeldahtherm (Gerdart GmbH + Co. KG Fabrik fur Laborgerate Postfach 1628 D53006 Bonn) until an opaque colour was obtained. The digested sample was rested for 12 h, diluted with distilled water and made up to the mark of 250 mL volumetric flask. Five mL of the digest was taken and distilled with 40% NaOH and the ionised ammonium was trapped by boric acid. The distillate was immediately titrated (n = 3) with 0.01N HCl. In order to obtain the percentage CP, the amount of N was multiplied by a factor 6.25. Neutral detergent fibre, acid detergent fibre and acid detergent lignin were determined as reported by Van Soest et al. (1994).
Calculation and statistical analysis: The average of the volume of gas produced from the blanks was subtracted from the volume of gas produced from each sample. This gave the net Gas Produced (GP) for each sample. Graphs of the volume of gas produced every 2-h interval of the 2 replicates of each sample was plotted against the incubation time. From the graph, the degradation characteristics were estimated as defined in the equation, Y = a + b (1-e-ct) (‘Ørskov and McDonald, 1979) where Y = degradability at time (t); a = intercept (or initial gas produced); b = potentially degradable fraction; c = rate of degradation of b; t = incubation time. The asymptote represents (a + b) or the potential degradability. The intercept of the curve represented by ‘a’ gives the DMD (dry matter disappearance) value at time zero hour. The ‘b’ value can be calculated as the difference between the asymptotic DMD and the intercept ‘a’. To calculate the rate of degradation, the above equation needs to be transformed:
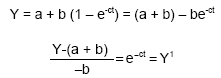 |
Take the natural logarithmic derivation of both sides.
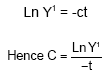 |
In order to get a good estimate of ‘c’, select ‘Y’ i.e. DMD% at time t, when the curve of the graph is changing most rapidly. The gas produced on incubation of 200 mg feed DM after 24 h of incubation together with the levels of other chemical constituents was used to predict digestibility of organic matter determined in vivo and metabolisable energy.
Metabolisable energy (ME, MJ/kg DM) = 2.20 + 0.136 GP + 0.057 CP; Organic matter digestibility (OMD, %) = 14.88 + 0.889 GP + 0.45 CP + 0.0651XA, where CP = crude protein; XA = ash and GP = the net gas production in ml from 200 mg dry sample after 24 hour of incubation and after correction for the day-to-day variation in the activity of rumen liquor using the Hohenheim standard. The data obtained from the in vitro gas production was subjected to ANOVA of the SAS (1988) and the level of significance of the means was tested using the Duncan’s Multiple Range F-test.
Chemical composition of the browse plants is presented in Table 1. The dry matter content of the browse plants ranged from 16.8% in bitter leaf to 46.5% in mango. Organic matter content of the browse plants ranged from 88.0% in bitter leaf to 94.0% in neem. Crude protein content ranged from 10.5% in mango to 21.8% in bitter leaf. The neutral detergent fiber ranged from 34.5% in neem to 38.5% in almond. The acid detergent fiber in the browse plants ranged from 21.0% in almond to 26.3% in neem. The acid detergent lignin ranged from 6.5% in almond to 15.5% in bitter leaf. The browse plants used in this present study have crude protein that ranged between 10.5 and 21.87%. This is reasonable and it is in accordance with Espinosa (1984), that tree leaves possess high protein content of 18-26% CP on the average.
The browse plants in the present study have CP contents above 8%, a minimum requirement for ruminants. According to Norton (2003), feed containing less than 8% CP cannot provide the minimum ammonia levels required by rumen microorganisms to support optimum activity.
| Table 1: | Chemical composition (g/100 g DM) of neem, almond, mango and bitter leaf plants |
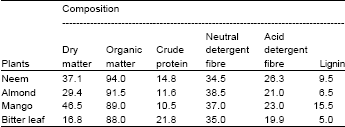 | |
Thus, these browse plants are beneficial and therefore can be used for supplementing the low protein pastures and crop residues especially during the dry season. The low fibre fractions in the present study was explainable as the level of fibre always depend on the stage of growth (Babayemi, 2007). The age of the browse leaves used in the present study was not known. It has been established that the lower the fibre content, the better the feed intake and the digestibility since high lignin is reported to physically encrust structural carbohydrates, preventing enzyme attack and inhibiting attachment of rumen microorganisms (Coombe, 1981).
Table 2 shows the secondary metabolites contained in the browse plans. All the browse plants tested negative to the presence for saponin except for mango leaf that has a level of 2 mm height indicating an amount of saponin content in it. Although the height of saponin in mango leaf was not supposed to reckon with according to the spot test procedure (Larrahondo, 1985), but its presence had effect on gas production and more importantly the methane production. A further analysis for qualitative determination of secondary metabolites, especially the saponin, is essential in order to ascertain its level in mango leaf. All browse plants contained flavonoid or phenol but according to the spot test method used, condensed tannins were specifically implicated. The browse plants indicated the presence of steroids. An overview analysis by qualitative evaluation, apart from being beneficial as a field or spot test in lieu of its low requirement for laboratory equipment and facilities may be more applicable as initial detection for screening when samples are large (Babayemi et al. 2004). Secondary metabolites such as tannins, saponins and steroids were qualitatively analyzed in the browse plants. Tannins have some special properties such as capacity to bind strongly with proteins, polysaccharides, nucleic acids, steroids, alkaloids and saponins (Mueller-Harvey and McAllan, 1992; Haslam, 1981). Although very negligible, the amount of saponin found in mango leaves may be an advantage to ruminants. An amount of saponins was also found in tropical fruits, which suppressed fermentation and methanogenesis in faunated and defaunated rumen fluid (Hess et al. 2003). The browse plants in the present study would therefore protect the dietary protein against microbial attack when included as supplements. A low saponin observed in the mango species might be important when fed to ruminants, to suppress the methane production thereby conserving the energy and simultaneously decreasing methane emission (Babayemi et al. 2004).
Table 3 represents the cumulative gas productions from the four different browse plants. There was consistent increase in gas production by all the forages with increasing incubation period (Fig. 1).
| Table 2: | Qualitative analysis evaluation of secondary metabolites of neem, almond, mango and bitter leaf plants in Nigeria |
 | |
| Table 3: | Gas production from the in vitro fermentation of the neem, almond, mango and bitter leaves incubated for 24 h |
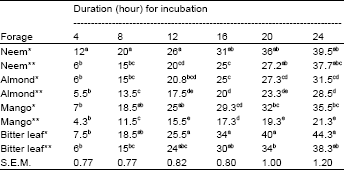 | |
| * and ** represented extracted and whole plants respectively; a,b,c,d = Means within the column with different superscripts are highly significant (p> 0.05); S.E.M.: Standard error of mean | |
This is expected being characteristics of tropical browse trees (Fadare and Babayemi, 2007). There were significant differences in the volume of gas produced between the forages. The bitter leaf, either extracted or not maintained the highest gas production value followed by neem than other plants. Characteristically, bitter leaf and neem plants are naturally bitter which might be due to the presence of tannin. On the other hand, the high crude protein and low lignin contents observed (Table 1) might have contributed to the proliferation of microbes that degraded the forages, resulting to their outstanding gas production. It was observed in Table 3 from the in vitro fermentation that the gas production from the extracted browse plants was higher compared to those that were not extracted (whole leaves). The improved gas production might be due to the absence of tannins which has been removed during extraction that would have inhibited the microbial degradation of the proteins contained in them.
Presented in Table 4 are the in vitro gas production characteristics of the extracted and whole browse plants. There were no significant differences observed in the ‘a’ and ‘c’ although all the extracted leaves had higher mean values than the unextracted. Significant differences (p<0.05) occurred for ‘a+b’, ‘b’, ‘Y’ and ‘t’ neem and bitter leaf plants were similar in these parameters but higher than those of almond and mango leaves. The importance of secondary metabolites was also glaring as being potent to stem down degradation of feedstuffs (Babayemi and Bamikole, 2006).
| Table 4: | In vitro gas production characteristics of extracted and whole neem, almond, mango and bitter leaf plants in Nigeria |
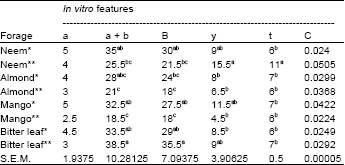 | |
| * and ** represented extracted and whole plants respectively; a,b,c,d = Means within the column with different superscripts are highly significant (p> 0.05); S.E.M.: Standard error of mean | |
| Table 5: | Metabolizable energy (ME = MJ/kg DM), organic matter digestibility (OMD = %) and methane (CH4 = ìmol/200 mg DM) of extracted and whole neem, almond, mango and bitter leaves |
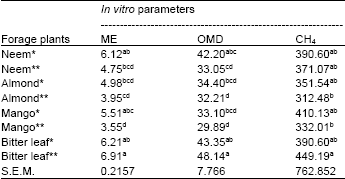 | |
| * and ** represented extracted and whole plants respectively; a, b, c, d = Means within the column with different superscripts are highly significant (p> 0.05); S.E.M = Standard error of mean | |
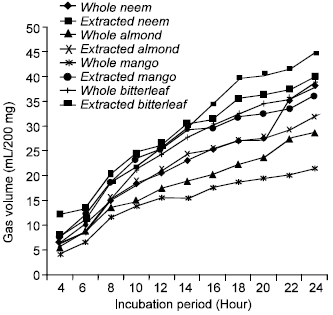 | |
| Fig. 1: | Gas producation of whole and extracted browse trees |
Presented in Table 4 is the predicted metabolizable energy, organic matter digestibility and measured methane production. Effect of extraction of the secondary metabolites from the browse leaves was not significantlyobvious in ME, OMD and CH4 values. However, there were significant (p<0.05) variations between browse leaves for ME, OMD and CH4 values. Although the variations for CH4 between the extracted and whole leaves were not significant, the differences were meaningful, suggesting positive effects of some antinutritional factors to suppress high methane production. In feed formulation, manipulation to reduce preponderance of methane is encouraged.
Conclusion: Results showed that edge-row plants can also be used as browse plants for ruminants. These browse trees are high in crude protein and contain certain antinutrirional factors also known as secondary metabolites. The metabolizable energy is high but low in organic matter digestibility.