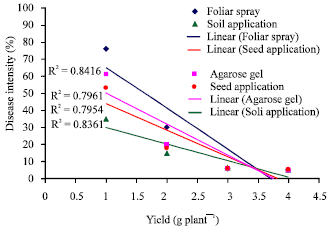
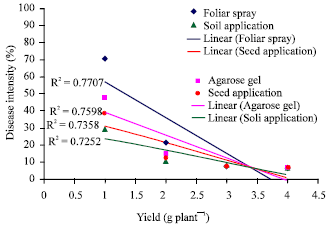
Research Article
The Relative Performance of Different Inoculation Methods with Alternaria brassicae and A. brassicicola on Indian Mustard
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh, 202002, India
Mujeebur Rahman Khan
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh, 202002, India
F.A. Mohiddin
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh, 202002, India