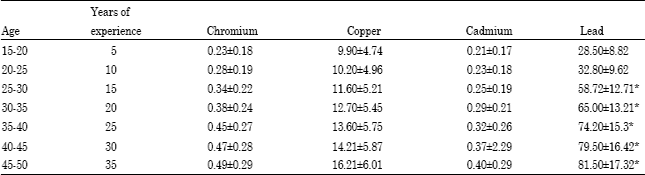
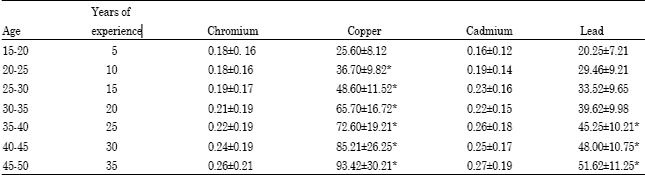
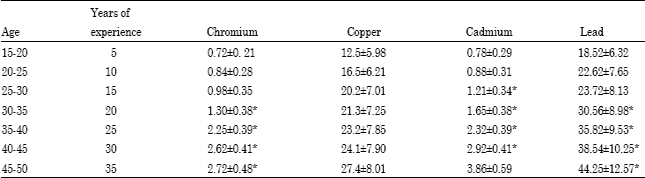
Research Article
Evaluation of Occupational Exposure to Toxic Metals using Fingernails as Biological Indicators
Department of Biotechnology, K.S.R. College of Arts and Science (Autonomous), Tiruchengode, Namakkal District, Tamil Nadu, 637 215, India
J. John Prabakaran
Department of Biotechnology, K.S.R. College of Arts and Science (Autonomous), Tiruchengode, Namakkal District, Tamil Nadu, 637 215, India