Research Article
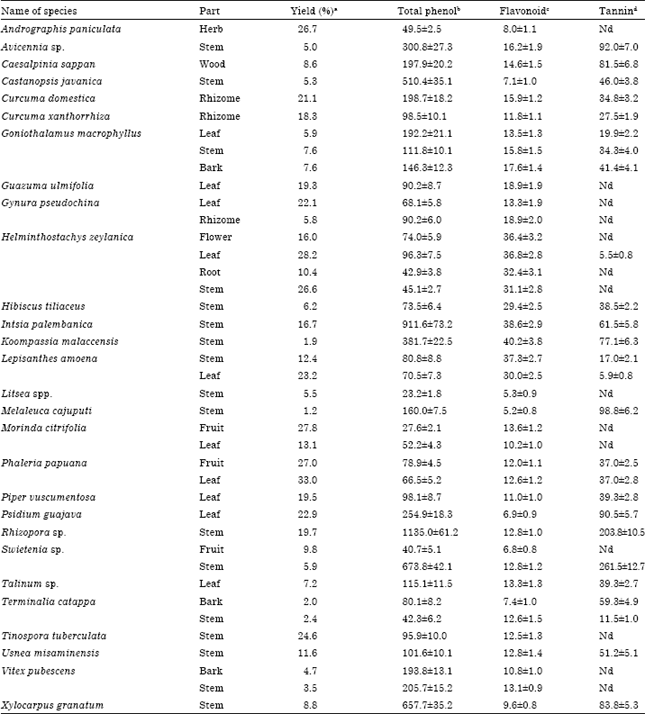
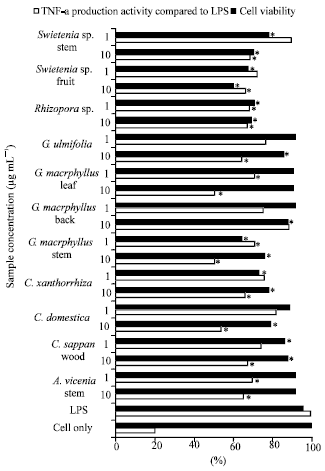
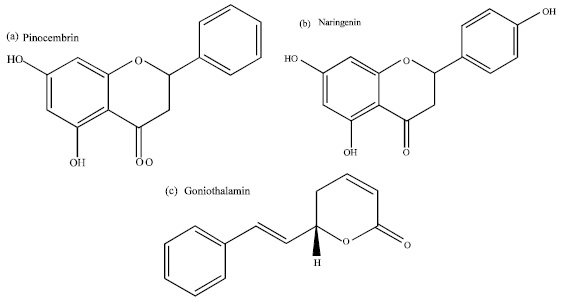
TNF-α Production Inhibitory Activity, Phenolic, Flavonoid and Tannin Contents of Selected Indonesian Medicinal Plants
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, IPB Darmaga Campus, Bogor, Indonesia
S. Kotsuka
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan
K. Yamauchi
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan
H. Kuspradini
Faculty of Forestry, Mulawarman University, Samarinda, East Kalimantan, Indonesia
T. Mitsunaga
Faculty of Applied Biological Science, Gifu University, 1-1 Yanagido, Gifu, 501-1193, Japan
L.K. Darusman
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, IPB Darmaga Campus, Bogor, Indonesia