Short Communication
Development of A Low Energy Thermodynamic Water Suction Pump
Nuclear Institute for Agriculture and Biology, Faisalabad, Pakistan
Hafiz A. Bilal
Government College of Technology, Faisalabad, Pakistan
Different types of available water pumping machines include (i) cylinder piston pump ii) centrifugal pump, (iii) turbine or rotary pump (iv) submersible pump and (v) air lift pump. The cylinder piston pump works on the principle that to and fro motion of piston or plunger in a cylinder causes mono directional expansion so water rushes into the system and pushed out in the next motion. In centrifugal pump the air pressure creates vacuum that sucks sub-soil water. The effective water extraction depth is 28 feet maximum. In turbine or rotary pump, there is a long shaft fitted with blades at pre-fixed distances. When rotating the blades throw up the water to upper set consecutively until the water leaves the outlet. Its effective water extraction depth may be 100 feet or even more. The submersible pump is installed in the water pipe and has long life. In air lift pump there are two pipes installed. From one pipe air is pressurised from the pump and pushed to the system. The aerosol formed, being lighter than water, rushes out from the second pipe. However, this is used for sampling purpose as the expenses on compressor are more and also it is difficult to maintain the high pressure constantly. All available pumps consume mechanical power derived from electrical or fuel involving high costs and increase entropy of the system. Therefore, a water pumping set is introduced for utilising wasted/surplus thermal energy in the form of smoke and exhaust gases from industry, domestic or municipal garbage burning chambers or solar heat collectors at negligible working cost.
Theoretical Development: Hot exhaust gases, released from a burning fuel, contain (i) high thermal energy, (ii) carbon-di-oxide, water vapours and other gases. These gases have low density and may displace downward the cold water/'gases (at high density) already present in a process container elevating the inner temperature. Now a fine jet of a cold water enters the system. On cooling and absorption of water soluble gases suction is created that is used to perform useful work. Cold (ground) water is used as a sink for heat and water soluble gases simultaneously. There is no appreciable undesired increase in the temperature of product water.
Operation: Schematic diagram of the pump is shown by Figure 1. The pump works in two cyclic processes as follows:-
| Process 1: | Inlet A opens the hot gases/steam entry in the process tank and water/cold gases are expelled out of process tank from outlet C. Inlet B remains closed. |
| Process 2: | Inlet A and C are disconnected. Inlet B opens the cold ground water entry into the process tank. Absorption of water soluble gases, heat and steam in the cold water reduces the inner temperature and pressure. External water, at atmospheric pressure, rushes into the process tank under the pressure difference till the internal pressure increases and approaches to atmospheric pressure. |
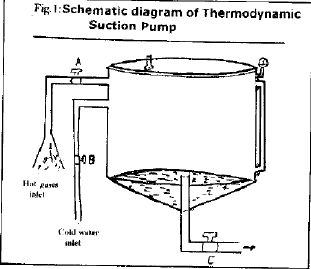 | |
| Fig. 1: | Schematic diagram of Thermodynamic Suction Pumnp |
Numerical Model: Let the gaseous mixture at T1, containing /, m and n partial pressure of a gaseous mixture of dry air, Carbon dioxide (and other water soluble gases) and water vapours at initial state functions of P1, T1 be cooled down to temperature T2 and pressure P2 in a tank of volume V.
According to Law of Partial Pressure
| (1) |
The change in partial pressure for each component can be derived separately as
| i) | Dry air(da) component: |
| (2) |
| ii) | Carbon di-oxide and other water soluble gases component: Pressure change is also accompanied by solubility of CO2 and other water soluble gases in water at cold water temperature. Therefore, the volume fraction is reduced to (m - c) or even less. Therefore, net change in pressure will be: |
| (3) |
c - solubility of water soluble gases at To
Water vapours (stearn) component: The water vapours will be converted from steam to water at the same temperature at the expenditure of latent heat of vaporization. The vapour pressure of pure steam at 760 mmHg (at 100°C) that falls to maximum of 23.74 mmHg at 25°C and even zero at 0°C. In case if the partial pressure of steam in the hot gases is less than
| (4) |
23.74 for all n>0.04 volume fraction at T2-25°C
The remaining vapours (n - 23.74) will be converted to water droplets condensing in the tank. Substituting the values from Equations 2, 3 and 4 in Equation 1 the final pressure of the system is given by
Pfinal = Pda + PCO2 + Psteam
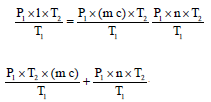 | (5) |
*value does not exceed the vapour pressure of steam at T2
Relative decrease in partial pressure = (P1-P2)/P1 Special cases:
Case 1: For complete dry air: Subsitituting/ = 1, m = n - 0, c = 0.002 (say) and final temperature T2 = Normal ground water (Table 1).
| Table 1: | Relative decrease in internal pressure (%) of dry air system |
 | |
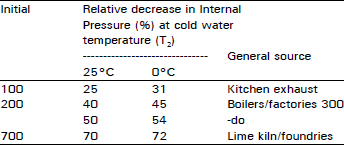
Case 2: For completely burnt gases air mixture:
Substituting / = 0.8, m = n = 0.1, c = 0.002 (say) and T2 = 25°C and 0°C (Table 2).
| Table 2: | Relative decrease in internal pressure of burnt gases/air system |
 | |
Case 3: For pure steam system:
Substituting / = m = 0 and n = 1 and final temperature of 25°C
Reduction in pressure with cold water
| at 25°C | = | 97% | |
| and | at 0°C | = | 100% |
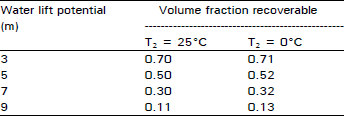
Working Capacity of system under different water tables: The reduction in internal pressure of the tank is converted to useful work. The maximum recoverable work in the form of volume fraction (f) of water in the process tan connected to the water table at depth (h) is calculated in Table 3 for pure steam system.
| Table 3: | Maximum volume fraction recoverable from 100 percent steam system at initial temperature on 100°C cooled with ground water |
 | |
Recommendations: The system is recommended for (i) Out lifting the water from low elevation for agriculture, industrial and domestic use and ii) to reduce the health hazards associated with the exhaust gases released from industry, domestic or municipal/industrial garbage burning chambers.