ABSTRACT
Aqueous ginger extract was tested against root knot nematode Meloidogyne javanica. In vitro studies, higher concentrations of extract (100% concentration) suppressed root knot egg hatching and caused juveniles mortality. Pasteuria endospore attachment was greater in 25% concentration extracts compared to those at 100 and 50% concentrations. Plant growth improved and disease severity was reduced when combined applications of the plant extract and P. penetrans were applied. In treatments receiving 25% extract and P. penetrans, 72% females were parasitized by the bacterial antagonist and 9 spores of bacteria per juvenile were found attached with nematode cuticle.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.858.860
URL: https://scialert.net/abstract/?doi=ajps.2003.858.860
INTRODUCTION
Of the various plant parasitic nematodes root knot nematodes Meloidogyne spp. have a wide host range and are considered the greatest threat to global agricultural production (Zaki, 2000; Sasser, 1980). Incorporation of plant parts/extracts into the soil alone or with biocontrol agents have been suggested as an alternative, safe and effective control method for the management of plant parasitic nematodes (Siddiqui and Alam, 1985). Ginger (Zingiber officinale Roscoe) is a biennial plant, bearing sessile aromatic tubers. It has been used in British as well as Indian Pharmacopoeia as a hot remedy for various diseases. It is also reported to cause motor paralysis of central origin after subcutaneous injection in frog (Watt and Breyer-Brandwijk, 1962). Of the potential biocontrol agents, Pasteuria penetrans, an obligate parasite of plant parasitic nematodes has great potential as economically and eco-compatible biological control agent (Dickson et al., 1994). In the present study the nematicidal potential of ginger was tested in vitro and its effect on the efficacy of the bacterial antagonist P. penetrans, against root knot nematodes was evaluated.
Material and Methods
Ginger rhizomes were obtained from a local market, dried and soaked in sterile distilled water (@ 500 g l-1) for 15 days. The filtrate obtained was used as 100% concentration. Further concentrations were prepared by adding the requisite amount of water to the extract. A culture of Meloidogyne javanica (Treub) Chitw. was maintained on tomato cv. Roma VF in the greenhouse. Root knot nematode eggs were extracted from the cultures pot (Hussey and Barker, 1973) using a 0.5% sodium hypochlorite solution. The egg suspension was poured on sieves lined with tissue paper and incubated for 48 hrs at room temperature (28±2oC). Freshly hatched juveniles were collected after 48 hrs and stored in a glass beaker for further use. Pasteuria penetrans (isolate PP3) used in the present study provided by Dr. S. R. Gowen, University of Reading, UK. The bacterium was propagated on M. javanica on tomato (Lycopersicon esculentum Mill cv. Roma VF) in the greenhouse and stored in dried root powder containing infested nematode containing resting spores of the bacterium.
For the hatching assay, egg masses of equal size were collected manually from root knot infested tomato roots, surface sterilized (0.2% sodium. hypochlorite) washed twice with sterile distilled water and kept in glass cavity blocks (d. 2 cm) @ 1 egg mass/cavity block, containing 2 ml of extract concentration (100%). Egg masses in sterile distilled water served as a control. Each treatment had four replicates. The number of hatched juveniles in each treatment was recorded after 24 and 48 hrs.
To study the effect of the rhizome extract of ginger on the mortality of root knot juveniles, 100 μl of nematode suspension (containing about 30-50 juveniles) was added to glass cavity blocks containing 2 ml of extract (100% concentration). Juveniles in sterile distilled water served as a control. Each treatment was replicated four times. The mortality was assessed as the mean percentage of dead nematodes using compound microscope (x10). Nematodes were considered dead if they did not move when probed with a fine needle.
To study the effect of test extract on the attachment of P. penetrans to root knot juveniles, 40-60 freshly hatched juveniles were added to a glass cavity block containing 2 ml extract (100, 50 and 25% concentration) and 200Fl of bacterial spore suspension (4.5x105 spores/1000Fl). A treatment lacking the bacterial spore suspension served as a control. Each treatment was replicated four times. Observations were taken after 24 hrs and 20 juveniles/replicate were observed using compound microscope (x40) for the attachment of endospores of P. penetrans.
Sandy loam soil, pH 7.8, obtained from experimental fields of the Botany Dept., University of Karachi, was used to fill plastic pots (d. 8 cm), each containing 350 cc soil. The soil was amended with P. penetrans root powder @1% w/w (16x105 spores g-1 soil) and drenched with the aqueous ginger extracts @ 25, 50 and 100% concentration @20 ml pot-1. Two days after seedling transplantation, two day old hatched juveniles were introduced in the root zone of the tomato plant (@2000 J2 pot-1). Each treatment was replicated three times. Pots were arranged in a randomized complete block design on greenhouse bench. Pots were watered as needed and sprayed with insecticides once a week. The experiment was terminated 45 days after nematode inoculation. Growth parameters (as plant height, shoot weight), nematode population in soil (200 g) and root (one g) was determined. For the estimation of female infection with P. penetrans, root samples were collected from the bacterial applied pots. Ten mature females/root system, were teased out randomly, put on a glass slide, crushed under cover slip and confirmed the presence of bacterial endospores under compound microscope (x40). Percentage of the female infection by P. penetrans was calculated. Soil samples from bacterial applied pots were sub-sampled. Nematodes extracted from soil by modified Baermann method and P. penetrans spore attachment on 25 juveniles/sample was recorded (Stirling, 1984).
Results obtained were analyzed statistically and subjected to factorial analysis of variance (FANOVA). The follow up of FANOA included least significant difference (LSD) according to Gomez and Gomez, 1984. Standard error difference (SED) was also calculated.
RESULTS AND DISCUSSION
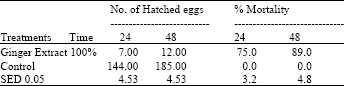
Emergence of root knot larvae was significantly (p<0.001) reduced with plant extract and exposure times (p<0.001). Aqueous extract of ginger significantly (p<0.01) inhibited egg hatching and caused mortality of M. javanica. Fewest eggs were hatched in the extract after 48 h even after transferred to distilled water. Significantly greater mortality was observed at 48 h exposure to plant extract than at 24 h (Table 1).
| Table 1: | Effect of aqueous extrcat of ginger on egg hatching and % juveniles mortality in vitro |
 | |
| Table 2: | Effect of aqueous extract of ginger on attachment of Pasteuria spores to root knot juveniles in vitro |
 | |
Fewest spores (p<0.001) attached to root knot juveniles after 24 h in 100% concentration of extract whereas 50 and 25% concentration had non significant effects on bacterial spore attachment; where more than 14 and 15 spores per juveniles were found attached, respectively compared to the untreated control (>18 spores/J2) (Table 2).
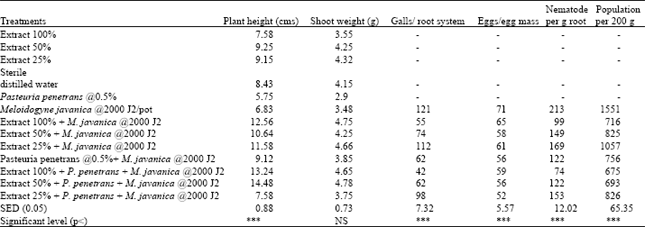
P. penetrans alone provided disease control similar to the treatments where 100 and 50% concentration of the extract were applied. Pots receiving the higher concentrations (100 and 50% concentration) with P. penetrans significantly reduced (p<0.001) gall formation per root system, egg mass production, egg formation per egg mass compared to other treatments and the untreated controls (Table 3). P. penetrans with 50 and 25% extract provided a greater improvement in tomato plant height, fresh shoot weight compared to the other treatments and the untreated control (Table 3).
Bacterial spore attachment was favoured by 50 and 25% concentrations of the extract. More than 9 bacterial spores per juvenile were observed in pots receiving 25% concentration of the plant extract compared to 100 and 50% concentrations. About 72% root knot females were found parasitized by the bacterium in the treatment receiving 25% concentration of the plant extract and P. penetrans followed by other treatments. The treatment of P. penetrans with ginger extract (25% concentration) resulted in the attachment of >9 spores per juvenile Reduction in bacterial spore attachment (>4 spores/J2) was observed in treatments where P. penetrans was applied with the aqueous plant extract at 100% concentration (Table 4).
There is not previous report on the nematicidal activity of ginger against plant parasitic nematodes. In the present study aqueous extracts of ginger were tested against root knot nematodes, M. javanica. The toxicity of the aqueous extract is due to a biologically active constituent(s). The reduction in bacterial spore attachment differed between the test concentrations (100, 50 and 25%) of plant extract.
| Table 3: | Effect of aqueous extract of ginger and P. penetrans in the control of root knot nematode on tomato plants |
 | |
| Table 4: | Effect of aqueous extract of ginger on juveniles and females infection by P. penetrans |
 | |
| **, p<0.01; ***, p<0.001 | |
In the greenhouse experiment, co-inoculations of plant extract and P. penetrans enhanced the growth of tomato plants and the disease was suppressed. The effectiveness of P. penetrans as a biocontrol agent was increased when 25% extract was applied for the management of root knot nematodes. Root knot female infection (72%) by bacteria occurred in the treatment receiving 25% plant extract with P. penetrans. Beside the nematicidal mode of the action, the test extract might induce behavioral effects, such as modifying host recognition by nematode (Zuckerman and Esnard, 1994). Further investigations are required, which could lead to the exploitation of the natural biocidal activity of ginger against root knot nematodes as an environmentally benign control measure.
ACKNOWLEDGMENTS
This work was carried out under the research grant of University of Karachi, which is sincerely acknowledged.
REFERENCES
- Dickson, D.W., M. Ostendorp, R. Giblin-Davies and D.J. Mitchell, 1994. Control of Plant Parasitic Nematodes by Biological Antagonists. In: Pest Management in Subtropics: Biological Control a Florida Perspective, Rosen, D., F.D. Bennett and J.L. Capineera (Eds.). Intercept, Andover, UK., pp: 575-601.
- Sasser, J.N., 1980. Root-knot nematodes: A global menace to crop production. Plant Dis., 64: 36-41.
Direct Link - Zuckerman, B.M. and J. Esnard, 1994. Biological control of plant nematodes, current status and hypothesis. Jap. J. Nematol., 24: 1-13.
Direct Link








