ABSTRACT
In cocoa cotyledon, storage albumin is abundantly present compared to other proteins. Two-dimensional electrophoresis (2-D IEF/SDS-PAGE) and native isoelectric focusing (native-IEF) techniques were used to analyse some of its biochemical characteristics. 2-D IEF/SDS-PAGE analysis by using commercially available immobilised pH gradient (IPG) gels in the first dimension for IEF exhibited reproducible patterns and high resolution of polypeptide patterns. The storage albumin (22 kDa) exhibited as a single form on 2-D IEF/SDS-PAGE with pI at around 4.6. An albumin was not resolved into multiple forms in IEF. The study found that there was similar analytical identity in storage albumin for all cultivars. This indicates that the storage albumin did not reflect genetic differences.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.958.962
URL: https://scialert.net/abstract/?doi=ajps.2003.958.962
INTRODUCTION
Cotyledon storage proteins have been classified as either albumin, globulin, prolamin or glutelin, according to the extent of their solubility in water, dilute salt solutions, aqueous alcohol and dilute alkali or acid, respectively (Osborne, 1924). Proteins constitute 10-16% of the dry weight of cocoa cotyledon. Cocoa cotyledon protein can be fractionated into albumin, globulin, glutelin and prolamin (Zak and Keeney, 1976). Albumin is abundantly present in cocoa cotyledons and comprises about 51% of total cotyledon protein (Voigt et al., 1993). It has been reported that the biological activity of albumin fraction is similar to protease inhibitor (Dodo and Furtek, 1994). Moreover, albumin was not significantly degraded compared to VCG during cocoa fermentation (Amin et al., 1997; 1998). The VCG of cocoa cotyledons were degraded to produce the specific-cocoa aroma precursors during In vitro incubation with cocoa cotyledon proteases or during aseptic bean incubation or during commercial fermentation provided that the pH-value is in the range of 5.0–5.5 (Voigt et al., 1994). By using a one-dimensional electrophoresis (SDS-PAGE) technique, it was clearly shown that cocoa cotyledon albumin has one predominant polypeptide of 21 kDa, while VCG has two predominant polypeptides with apparent molecular weights of the range 45-49 and 31-37 kDa (Voigt et al., 1993). The VCG subunits revealed at least 8 multiple forms had pIs in the range of 6.1 to 6.8 (Amin et al., 2002a, b). This multiple forms may indicate that the VCG occurs in the cocoa cotyledons was not of single gene-origin. In contrast to VCG, albumin was not degraded during fermentation, meaning that it remained unaffected by the action of active proteases. Therefore, the study is conducted to investigate the influence of cultivars of cocoa trees on cocoa cotyledon albumin polypeptide pattern. In addition, there is a dearth of study on polypeptide composition of albumin by 2-D IEF/SDS-PAGE, thus some of its biochemical characteristics using 2-D IEF/SDS-PAGE and native isoelectric focusing is also investigated.
MATERIALS AND METHODS
Cocoa fruits: The cocoa fruits of Forastero (PBC 140 clone) were purchased from Golden Hope Plantation, Sitiawan, Perak, Malaysia. Criollo and Trinitario were obtained from the Kalawining Experimental Garden of the Indonesian Coffee and Cocoa Research Institute, Jember, Indonesia. The fresh beans were extracted from the fruits immediately after arrival and the cotyledons were shock-frozen in liquid nitrogen and freeze-dried (Labconco, USA; -40°C, 120 bar).
Preparation of acetone dry powder: After removing the radiculae, the lyophilised cotyledons were crushed with a mortar and pestle. To remove the cocoa butter, 5 g of crushed cotyledons were extracted with 200 ml of petroleum-ether (b.p. 40-60°C) for 8 h in a Soxhlet apparatus. After evaporating the residual petroleum-ether at room temperature under fume hood, then the crushed defatted cotyledons were then ground with a mortar and pestle. An acetone dry powder (AcDP) of defatted cocoa cotyledons was prepared as described by Voigt et al. (1993) with slight modifications. In order to remove the polyphenol compounds, 5 g of the defatted cocoa cotyledon powder was added to 100 ml of 80% cold aqueous acetone containing 0.1% thioglycollic acid in a 250 ml polypropylene centrifuge tube. The mixture was stirred with a magnetic stirrer for 30 min in an ice-bath (2±1°C). The mixture was centrifuged at 10,000 x g for 15 min at 4°C. The supernatants (solvent phases) were discarded and the resultant pellet was re-extracted twice with 80% and four times with 70% cold aqueous acetone following the same procedure as described above. The white dry powder was kept in a sealed glass container and stored at–20°C before being used.
Extraction of Albumin: An albumin of cocoa cotyledons was extracted according to the modified method of Voigt et al. (1993). Ten grams of AcDP was added with Buffer A which was prepared from 50 mM Tris-HCl, pH 7.5 containing 10 mM NaCl, 2 mM EDTA and 7 mM 2-mercaptoethanol. The mixture was shaken for 1 h in a cold room (5±1°C) at 200 rev min-1 using an orbital shaker. The mixture was then centrifuged at 17,000 x g for 30 min at 4°C. The pellet was re-extracted three times with the same buffer. The ratio of AcDP to extraction buffers was 1:20. The supernatant extracted with Buffer A was pooled and kept for the next step. The supernatant of Buffer A containing the bulk quantity of albumin fraction was dialysed against distilled water to remove the salt ions present in the extraction buffer before further analysis. Protein content was determined according to Hatree (1972) using γ–globulin as a standard.
Two-dimensional electrophoresis: All the steps for two dimensional polyacrylamide gel electrophoresis (2-D IEF/SDS-PAGE) were carried out according to the modified method described by Pharmacia Biotech (Anonymous, 1996a). 2-D IEF/SDS-PAGE was run horizontally using a Multiphor II Flatbed electrophoresis unit (LKB, Pharmacia, Sweden) connected with a Multi-Temp as a cooling system.
Ten μl samples were mixed with 40 μl of sample solution which was prepared from 9 M urea, 2% Triton X-100, 2% β-mercaptoethanol, 0.9% Biolyte of pH 3-10 and a few grains of Bromophenol Blue (BPB). The samples were transferred into an Eppendorf tube in small portions and stored at – 20°C until being used. Isoelectric focusing (IEF) was performed in a ready-made Immobiline DryStrip precast polyacrylamide gel strips (70x0.5 mm) containing an immobilised pH gradients (IPG) (Pharmacia Biotech, Uppsala, Sweden). Running conditions were as follows: 300 V for 1 h and 2000 V for 15 h. After IEF, the IPG gel strips were equilibrated with equilibration solution (0.05 M Tris-HCl buffer, pH 6.8 containing 6 M urea, 30% glycerol and 1% SDS) for 10 min. The equilibrated IPG gel strips were placed on a piece of filter paper and turned up on one of its edges to help it drain excess equilibration solution. The IPG gel strips were transferred onto the surface of the ready-made SDS-PAGE gels (110x245x0.5 mm) (Pharmacia Biotech, Uppsala, Sweden). Electrophoresis was carried out at a constant current of 20 mA for 30 min and continued with 50 mA for another 75 min. Then the gel was stained with silver nitrate according to the method essentially developed by Heukeshoven and Dernick (1985) with some modifications.
Native isoelectric focusing: Native isoelectric focusing (Native-IEF) was performed in ready-made Ampholine PAGplate (pH 3.5–9.5) polyacrylamide gel (T=5%, C=3%) on a polyester support film with (245x110x1 mm, l x w x d) dimensions (Anonymous, 1996b). The native-IEF was run horizontally using the Multiphor II electrophoresis unit connected to a cooling system. The anodic and cathodic electrolytes were 1 M solution of phosphoric acid and 1 M solution of sodium hydroxide, respectively. The broad pI calibration kit (pH 3-10) was run in parallel with the samples. The broad pI marker used were amyloglucosidase (3.50), soybean trypsin inhibitor (4.55), β-lactoglobulin A (5.20), bovine carbonic anhydrase B (5.85), human carbonic anhydrase B (6.55), horse myoglobin-acidic band (6.85), horse myoglobin-basic band (7.35), lentil lectin-acidic band (8.15), lentil lectin-basic band (8.65) and trypsinogen (9.30). They were all purchased from Amersham Pharmacia Biotech (Uppsala, Sweden). Native-IEF was carried out at 1500 V for 90 min. The temperature of coolant was set at 16°C. The silver staining was carried out according to the method of Heukeshoven and Dernick (1985) with some modifications.
RESULTS AND DISCUSSION
An attempt has been made to compare the polypeptide composition of storage albumin by 2-D IEF/SDS-PAGE. Fig. 1 and 2 shows the 2-D IEF/SDS-PAGE pattern of storage albumin extracted from cotyledon of Forastero (PBC 140 clone) and co-migrated storage albumin from three different cultivars (Forastero, Criollo and Trinitario).
Based on the apparent molecular weight, SDS-band A of 22 kDa is considered to be a polypeptide of storage albumin. A polypeptide obtained corresponding to the storage albumin (22 kDa) in PBC 140 and other genotypes was analytically similar (Fig. 1 and 2). A similar result was obtained for the albumin fraction from seeds of different sunflower (Raymond et al., 1991) and barley varieties (Curioni et al., 1995). In addition, isoelectric point (pI) of cocoa cotyledon storage albumin was more acidic (3.5 to 4.55) compared to that of other seeds such as Amaranthus hypochondriacus (pI 7.5) (Marcone et al., 1994).
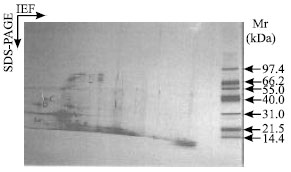 | |
| Fig. 1: | 2-D IEF/SDS-PAGE patterns of an albumin extracted from cocoa cotyledon PBC 140 clone (Forastero type). First dimension: IEF at pH 3-10L using IPG gel strips (11.0 cm x 3.0 cm x 0.5 mm), left to right. Second dimension: SDS-PAGE, top to bottom, gradient gel concentration (8-18%). Approximately 5 μg protein per gel was loaded onto first dimension. The major SDS-bands are indicated. Gel was stained with silver nitrate. Mr: broad molecular weight markers |
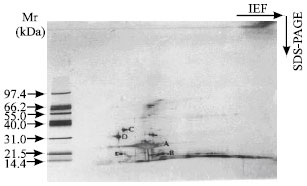 | |
| Fig. 2: | 2-D IEF/SDS-PAGE patterns of co-migrated cocoa cotyledon albumin from 3 different cultivars. First dimension: IEF at pH 3-10 L using IPG gel strips (11.0 cm x 3.0 cm x 0.5 mm), left to right. Second dimension: SDS-PAGE, top to bottom, gradient gel concentration (8-18%). Approximately 5 μg protein per gel protein was loaded onto first dimension. The major SDS-bands are indicated. Gel was stained with silver nitrate. Mr: broad molecular weight markers |
The pI of cocoa cotyledon storage albumin was similar to the pH value (pH 4.0) obtained by MacDonald et al. (1994) after isoelectric focusing of purified albumin in agarose gels. Storage albumin which was indicated as A did not split during IEF to give multiple forms. The result obtained was in contrast to that of cocoa cotyledon VCG (Amin et al., 2002a,b). It indicates that the storage albumin of cocoa cotyledons present in various genotypes had similar analytical identity and thus did not reflect genetic differences.
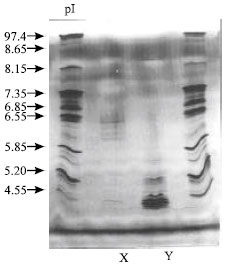 | |
| Fig. 3: | Native Isoelectric Focusing of Vicilin (7S)-Class Globulin (VCG) and Albumin from PBC 140 Clone. The protein samples were subjected to Ampholine PAGplate (Pharmacia Amersham Biotech, Sweden) in the range between 3.5 to 9.5. Lane x: VCG (0.3 mg protein) and y: Albumin (0.1 mg protein). The gel was stained with silver nitrate. The pI markers are indicated |
Most probably an albumin is a product of one gene. However, further studies on storage albumin gene or mRNA of cocoa cotyledon would be useful to provide a clearer indication with respect to a product of one gene.
From these patterns (Fig. 1 and 2) not only was a polypeptide of storage albumin indicated as A identical but so too were other polypeptides as labeled B, C, D and E. There were other polypeptide spots detected along with storage albumin with apparent molecular weights less than 40 kDa which were focused at different pH values. Three polypeptide spots C, D and E with apparent molecular weights of 34.9, 29 and 18.6 kDa were present in the gel (Fig. 1 and 2). These polypeptide spots could be the degradation fragments of cocoa cotyledon enzymes or of albumin impurities from the cytoplasmic compartment. According to Voigt et al. (1995), purified cocoa cotyledon aspartic endoprotease contained two polypeptide subunits with apparent molecular weights of 29 and 13 kDa. In addition, these two subunits were believed to be derived from self-digestion of 42 kDa polypeptide (Voigt et al., 1995). Sarkkinen et al. (1992) found those similar polypeptides with apparent molecular weights of 32 and 16 kDa to be constituents of the aspartic endoprotease from barley grains.
A 29 kDa polypeptide was resolved into two different isoelectric points (pI); one has pI less than 3.5 and another one around 4.6. However, the latter polypeptide isomers had an apparent pI similar to the pI of aspartic endoprotease from buckwheat seeds (4.2) (Belozersky et al., 1989). Moreover, plant seeds aspartic endoprotease so far investigated have been found to have pI in the range of 4.2 to 6.3. Baunmgartner and Chrispeels (1977) found vicilin peptidohydrolase (sulphydryl proteases) from mungbeans to have an apparent pI of 3.75. In this present study, there is no direct evidence to show that these polypeptides were exclusively corresponding to the polypeptide subunits of the cocoa cotyledon proteases. In addition, no work has been published in comparing the properties (pI and number of polypeptide isomers) of aspartic endoproteases subunits using 2-D IEF/SDS-PAGE electrophoresis analysis.
Using native IEF, it has been showed that the storage albumin, in which a good separation of its protein bands in the IEF gels (Fig. 3; lane y) was attained, at least five predominant plus eight faint bands were detected. The storage albumin protein bands had migrated to the more acidic end of the IEF gels. Five predominant protein bands of albumin were focused at pH between of 3.50 to 4.55 in native IEF (Fig. 3). As in 2-D IEF/SDS-PAGE the storage albumin (22 kDa) was not separated to give more multiple forms in IEF, indicating that the storage albumin was merely one of these bands. However, other protein bands in the cluster may represent other albumins.
The study found that the electrophoretic pattern of cocoa cotyledon albumin polypeptide was very similar for different Theobroma cacao L. cultivars namely Forastero, Criollo and Trinitario. Based on 2-D IEF/SDS-PAGE analysis, an albumin did not resolve into different multiple forms in IEF, indicating that most probably it did not reflect genetic differences.
ACKNOWLEDGMENTS
The authors would like to acknowledge the financial assistance provided by the Ministry of Science, Technology and Environment of Malaysia (Project No. 01-02-04-111) and the laboratory facilities of University Putra Malaysia.
REFERENCES
- Amin, I., S. Jinap, B. Jamilah, K. Harikrisna and B. Biehl, 2002. Analysis of vicilin (7S)-class globulin in cocoa cotyledons from various genetic origins. J. Sci. Food Agric., 82: 728-732.
CrossRefDirect Link - Amin, I., S. Jinap, B. Jamilah, K. Harikrisnan and B. Biehl, 2002. Oligopeptide patterns produced from Theobroma cacao L. of various genetic origins. J. Sci. Food Agric., 82: 733-737.
CrossRefDirect Link - Belozersky, M.A., S.T. Sarbakanova and Y.E. Dunaevsky, 1989. Aspartic proteinase from wheat seeds: Isolation, properties and action on gliadin. Planta, 177: 321-326.
CrossRefDirect Link - Curioni, A., G. Pressi, L. Furegon and A.D.B. Peruffo, 1995. Major proteins of beer and their precursors in barleyelectrophoresis and immunological studies. J. Agric. Food Chem., 43: 2620-2626.
CrossRefDirect Link - Dodo, H.W. and D.B. Furtek, 1994. Cloning and sequencing of a gene encoding a 21 kDa trypsin inhibitor from Theobroma cacao L. Cafe Cacao, 38: 113-117.
Direct Link - Hartree, E.F., 1972. Determination of protein: A modification of the lowry method that gives a linear photometric response. Anal. Biochem., 48: 422-427.
CrossRefPubMedDirect Link - Heukeshoven, J. and R. Dernick, 1985. Simplified method for silver staining of proteins in polyacrylamide gels and the mechanism of silver staining. Electrophoresis, 6: 103-112.
CrossRefDirect Link - MacDonald, H.M., K.E. Master and G.L. Pettipher, 1994. Partial purification of cocoa seed proteins and studies into the degradation of cocoa storage protein. Cafe Cacao, 38: 119-124.
Direct Link - Marcone, M.F., F.K. Niekamp, M.L. Maquer and R.Y. Yada, 1994. Purification and characterization of the physicochemical properties of the albumin fraction from the seeds of Amaranthus hypochondriacus. Food Chem., 51: 287-294.
Direct Link - Raymond, J., V. Inquello and J.L. Azanza, 1991. The seed proteins of sunflower: comparative studies of cultivars. Phytochemistry, 30: 2849-2856.
Direct Link - Sarkkinen, P., N. Kalkkinen, C. Tilgmann, C. Siuro, J. Kervinen and L. Mikola, 1992. Aspartic proteinase from barley grains is related to mammalian lysosomal cathepsin D. Planta, 186: 317-323.
CrossRefDirect Link - Voigt, J., S. Kamaruddin, H. Heinrichs, D. Wrann, V. Senyuk and B. Biehl, 1995. Development stage-dependent variation of the level of globular storage protein and aspartic endoprotease during ripening and germination of Theobroma cacao L. seeds. J. Plant Physiol., 145: 299-307.
Direct Link - Voigt, J., B. Biehl, H. Heinrichs, S. Kamaruddin, G. Marsoner and A. Hugi, 1994. In vitro formation of cocoa specific aroma precursors: Aroma-related peptides generated from cocoa seed protein by co-operation of an aspartic endoprotease and a carboxypeptidase. Food Chem., 49: 173-180.
Direct Link - Voigt, J., B. Biehl and S. Kamaruddin, 1993. The major seed proteins of Theobroma cacao L. Food Chem., 47: 145-151.
Direct Link - Zak, D.K. and P.G. Keeney, 1976. Extraction and fractionation of cocoa proteins as applied to several varieties of cocoa beans. J. Agric. Food Chem., 24: 479-482.
CrossRefDirect Link








