ABSTRACT
A hydroponic experiment was carried out to see the effects of salinity by using a randomized complete block design with four salinity treatments and five replicates. The salinity levels tested were 0, 50, 100 and 150 mol m-3 NaCl. CaCl2 was also applied to the salinity treatments. Green areas of leaf three and leaf four were not significantly affected by salinity at harvest one and harvest two, respectively. Green area of all other plant parts was significantly decreased with increase in salinity at both harvests. Salinity significantly decreased dry weight of leaf five, tiller two and roots at harvest one. At harvest two, dry weights of all plant parts except leaf 5 and dead leaves were significantly affected by salinity.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.244.248
URL: https://scialert.net/abstract/?doi=ajps.2005.244.248
INTRODUCTION
Salinity inhibits growth and development of most plants. Crops grown in salt affected soils may suffer from drought stress, ion toxicity and mineral deficiency leading to reduced growth and productivity[1]. Dry matter formed prior to anthesis has been estimated to contribute 3-30% of the grain dry matter at maturity[2-6]. Growth and yield in crop plants are affected to varying degrees by salinity[7-9]. The growth of plant is influenced by leaf area and by amount of dry matter produced per unit leaf area. Inhibition of shoot and root development is the primary response to the stress. Growth, morphology, anatomy and physiology of root and shoot are affected by salinity[8]. Changes in water and ion uptake and production of hormonal signals that communicate information to the shoot might induce changes in development[8,9]. Since root growth is usually less sensitive to salt stress than shoot growth, an increased root/shoot ratio is often observed when plants are subjected to salt conditions[10-12]. One of the main objectives of the present work is to study the effects of salinity on growth and dry matter production of wheat. Previous experiments[13,14] have shown large effects of salinity on leaf expansion. Therefore, this experiment examined the effects of NaCl on dry matter production parameters before anthesis.
MATERIALS AND METHODS
The experiment was carried out using a Randomized Complete Block Design. There were four salinity treatments and five replicates. The salinity levels tested were 0, 50, 100 and 150 mol m-3 NaCl. CaCl2 was also applied to the salinity treatments (50, 100 and 150 mol m-3 NaCI) in the ratio of 20:1 (moles Na:Moles Ca) in order to increase to increase the potassium/sodium ratio.
The experiment was initiated on 13 May and terminated on 29 June at Aber Farm, University of Wales, Bangor, UK. Spring Wheat variety Wembley was used in this experiment. The experiment was carried out in glasshouse with no control of temperature and without supplementary lighting.
In this experiment, large containers having 25 L water holding plastic containers (63x35 cm wide and 18.5 cm deep) were used. Prior to seeding, eight 7 mm (for air supply) and one 9 mm (for solution changes) holes were made in the front, sides and bottom of the containers. The holes were plugged with rubber bungs to facilitate easy changes of nutrient solutions and to fix air supply needles (No. 16: Terumo Europe, Belgium). The containers were arranged along the sides of large work benches, again to facilitate easy access for maintenance and measurements. Instead of polyurethane tubing, silicon tubing (Scientific Services, Chester, UK) was used to facilitate sealing of holes created by needles in it. The silicon tubing (5 mm Internal Diameter (ID), 8 mm Outer Diameter (OD)) was fixed along the sides of the workbenches and then connected to the air regulator. Air from the silicon tubing to the containers was supplied via narrow (0.58 mm ID, 0.96 mm OD) polythene capillary tubing (Portex Ltd. Hythe, Kent, England), which was cut into the silicon tubing and the bungs fitted in the containers. This system allowed a more uniform and efficient distribution of air in each container and avoided the air blockage problem encountered when using the 3-way air regulators and aerators in previous experiment[13].
Plants were germinated and grown in P180 Plugtrays (Cookson Plantpak Ltd., Maidon, Essex, UK). Prior to seeding, capillary matting was fixed at the bottom of each plugtray with copydex. Another P180 plugtray was stacked on top with the one containing the capillary matting at the bottom. This provided increased strength to the plugtray to withstand plant weight gained later during the experiment. The pairs of plugtrays were then placed on the top of the 25 L containers, which were filled with nutrient solutions (0.4 g LG1 Phostrogen). All the cells of the P180 plugtrays were filled with vermiculite. Seeds were presoaked over night in a muslin bag suspended under a slow running tap. The next morning the seeds were sown on the moist vermiculate in the P180 plugtrays with one seed per cell and a total of 180 seeds per tray (51.5x30.0 cm). The seeds then covered with newspapers and kept moist until the radicles and coleoptiles were seen to be emerging. The newspapers were later removed. The containers were then filled with tap water containing 0.4 g LG1 Phostrogen and the trays placed over the containers. The seedlings were later thinned to 90 per tray by uprooting alternate rows along the width of the tray, as 9 rows of 10 plants at a distance of 60x30 mm, which is equivalent to a plant population of 583 plants m-2.
Preparation of nutrient based salt solution: In this experiment, for ease of preparation and solution changes, Phostrogen based nutrient and salt stock solution were used for each treatment. After necessary calculations for each salt treatment being made, Phostrogen and all the micronutrients were added to each salt stock solution and the volume was made to 10 L and then stored in a cold room for further use. This technique facilitated the maintenance of a uniform supply of salt stress throughout the course of experiment.
Prior to salt stress (12 days after seeding), when the plants had 2 emerging leaves, the containers were drained out and refilled. The stock salt and nutrient solutions were drip fed into the plant containers from polyurethane containers, which were fixed to retort stands. One needle, connected to polythene capillary tubing was inserted into the bottom side of the polyurethane container and the other into the 25 L container holding the plants. The concentration of stock solutions varied for each treatment so that final desired concentration would be achieved. By this method salinity was introduced gradually and continuously over a 2 days period in each treatment. Salinisation was completed at 1+2 leaf stage (one fully expanded and two expanding leaves).
Harvest for growth analysis was made when leaf 5 and leaf 7 were fully expanded (ligule emerged). Leaf 5 was fully expanded on 14 June (18 DAS) whereas leaf 7 was fully expanded on 19 June (23 DAS) at 100 and 150 mol m-3 NaCl and 26 June (30 DAS) at 0 and 50 mol m-3 NaCl, respectively. At each harvest, 10 plants were harvested from the second and third rows of the right side of the replicate containers of each treatment concerned. The plants were kept large polythene bags and taken to the laboratory adjacent to the glasshouse. The dead leaves on the mainstern, Leaf 1 and 2 or plus 3 were grouped together. All the expanded and emerging leaves on the mainstern were also detached separately at the point joining the sheath. The primary and secondary tillers, their leaves and stem emerging from the axils of leaf I were considered as Tiller 1. Most plants did not produce more than 2 primary tillers. Therefore, the primary and secondary tillers, their leaves and stems emerging from the axils of leaf 2 and 3 were considered as tiller 2. The leaves of tiller I and tiller 2 were detached from the point joining the sheath and stems were cut into small portions to facilitate smooth feeding through the automatic area meter. Similarly main stem were also cut into small portions. The areas of all the detached green parts except those of dead leaves were recorded using an automatic area meter. After recording leaf area of all shoot parts, detached and all shoot parts were prepared for dry weight.
RESULTS AND DISCUSSION
Green areas of leaf three and leaf four were not significantly affected by salinity at harvest 1 and harvest 2, respectively (Table 1). Green area of all other plant parts was significantly decreased with increase in salinity at both harvests.
Salinity significantly decreased dry weight of leaf 5, tiller 2 and roots at harvest 1 (Table 2). Dry weight of all other plant parts was not significantly affected but was decreased with salinity in dead leaves, stems and tiller 1 while it was inconsistently affected in other plant leaves. At harvest 2 dry weights of all plant parts except leaf 5 and dead leaves was significantly affected by salinity. It was consistently decreased with salinity in expanding flag leaf, leaf 7, leaf 6, stems, tiller 2 and roots but increased in leaf 4 and dead leaves. Total plant dry weight was consistently decreased with salinity at both harvests. Leaf growth was affected more than root growth (Table 3). These results confirm the findings of earlier workers that leaf growth is often more reduced in common with dry soil[8,9,15]. It is probably due to factors associated with water stress rather than a specific effect. This is supported by the evidence that Na+ and ClG are always below toxic concentrations in the growing tissues, The underlying mechanism involved in the inhibition processes of root and shoot growth is not clearly established.
| Table 1: | Effect of different salinity levels on green area ( cm2/plant) of spring wheat |
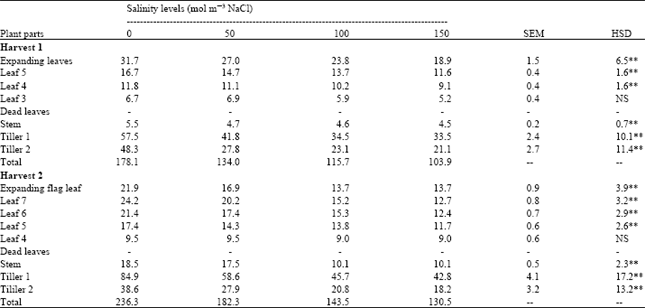 | |
| Table 2: | Effect of different salinity levels on dry weight (mg/plant)of spring wheat |
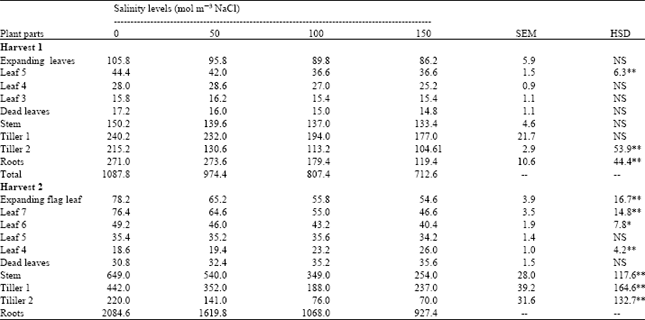 | |
| SEM = Standard error of means, HSD = Honestly significant difference, NS= Not significant, ** = Significant at 1% probability levels, respectively - = Not applicable, -- = Data not analyzed | |
Restriction of root growth by salinity reduces the soil volume that can be explored by the root and hence the availability and uptake of water and essential minerals[8]. The diminished supply of nutrient to the shoot may contribute to growth reductions[16-20]. The increase in root/shoot ratio under stress, which might diminished the demand for element supply to the shoot, has a potential to increase the ability of the root to supply those elements and hence might present an adaptive advantage[10]. A potentially negative effect of such a change is the decreased ability of the shoot to supply assimilates to the root and the growing tissues, which is likely to affect plant development and survival particularly under long-term salinization[21,22]. Hu and Schmidlialter[23] showed that in wheat growing in 120 mM NaCl with a 25 per cent reduction in growth rate, Na+ in the growing cells of leaves was only 20 mM at maximum and ClG 60 mM.
| Table 3: | Effect of different salinity levels on percent decrease in green leaf area and dry weight per plant of spring wheat |
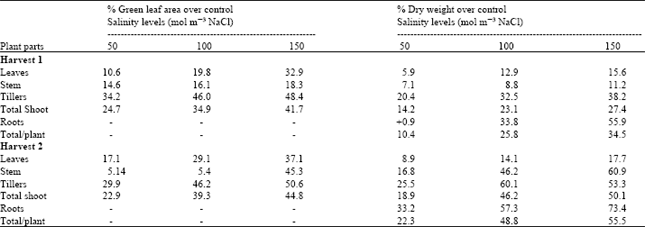 | |
| =- Not applicable | |
Whether water status, hormonal regulation or supply of photosynthate exerts the dominant control over growth of plants in dry or saline soil is an issue that has been debated for the last decades and not yet resolved. Over the time scale of days, there is much evidence to suggest that hormonal signals rather than water relations are controlling growth in saline soils[9]. Two closely related wheat genotypes that differed in rates of Na+ accumulation had the same growth reduction for the first 4 weeks in 150 mM NaCl. However, visual differences appeared long before that; the genotype with the higher rate of Na+ uptake showed faster leaf senescence, with injury appearing in the oldest leaf after 2 weeks[24]. Later, when the proportion of dead leaves increased above about 20% of the total, the rate of new leaf production slowed down dramatically and some individuals died. The initial growth reduction is due to the osmotic effect of the salt-sensitive plant from a more tolerant one is the inability to prevent salt reaching toxic levels in the transpiring leaves[9].
REFERENCES
- Munns, R., 2002. Comparative physiology of salt and water stress. Plant Cell Environ., 25: 239-250.
CrossRefDirect Link - Cheeseman, J.M., 1988. Mechanism of salinity tolerance in plants. Ann. Rev. Plant Physiol., 87: 547-550.
Direct Link - Rawson, H.M., R.A. Richards and R. Munns, 1988. An examination of selection criteria for salt tolerance in wheat, barley and triticale genotypes. Crop Pasture Sci., 39: 759-772.
CrossRefDirect Link - Hu, Y. and U. Schmidhalter, 1988. Spatial distribution and net deposition rates of mineral elements in the elongating wheat (Triticum aestivum L.) leaf under saline soil conditions. Planta, 204: 212-219.
Direct Link - Munns, R., J. Guo, J.B. Passioura and G.R. Cramer, 2000. Leaf water status controls day-time but not daily rates of leaf expansion in salt treated barley. Aust. J. Plant Physiol., 27: 949-957.
Direct Link








