ABSTRACT
Pollination and seed production in Portulaca oleraceae were investigated. Evidence from pollen and ovule number suggest that the species is autogamous. Pollen grain germination of P. oleraceae could be assessed on stigmas based on the difference in morphology between germinated and non-germinated pollen grains. Pollen showed a high germination percentage on stigmatic surfaces (>90%). P. oleraceae had two types of flowers with a different status of the pistils at full opening of the flowers : one with full spreading and the other with including stigma lobes of the pistils. Length of styles differed between these two types of flowers. Although the mechanism of pollination differed markedly between these two types of flowers, these flowers achieved >80% success in fertilization in a pollinator-free environment. In a type of flower, pollination that occurred at closure of the flowers also contributed to the high percentage of fertilization. Plants from three area had more flowers with including stigma lobes than those with exerted stigma lobes. Small number of days (10-11 days) were required for maturation of seeds that also contributed to the vigorous reproduction in this plant species.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.226.232
URL: https://scialert.net/abstract/?doi=ajps.2006.226.232
INTRODUCTION
Although Portulaca oleraceae are cosmopolitan weeds that significantly affect agriculture, the ecology of this species is not satisfactorily clarified. It is hard to control because individual plants produce large numbers of seeds (Mulligan, 1972; Zimmerman, 1976; Matthews et al., 1993). Seed production in P. oleraceae were investigated with special reference to pollination mechanisms because I found that the plants bore two types of flowers, some with delayed autogamy and others with autonomous autogamy. Seed production was compared between the flowers because they self-pollinated at different times. Also it is important to assess the exact percentages of pollen germination for the study of seed production. Actually, the assessment of pollen germination on the stigmas is difficult. Therefore, agar or gelatin media are used to assess pollen germination in practice. However, pollen germination depends on the composition of these media. It is questionable whether pollen germination on these media represent that on stigmatic surfaces. In the present study, pollen germination was assessed on stigmatic surfaces using a microscope with or without Ultraviolet (UV) source. I also examined the number of ovules and pollen grains in each plant because they are indicators of plant breeding system (Cruden, 1977). The number of days from pollination to maturation of seeds is an essential theme in the study of reproductive biology in P. oleraceae because the time from pollination to scattering of mature seeds may contributes to the rapid cycle of reproduction taking into account the high germination ability in seeds (>90%) (Zimmerman, 1977). Therefore, present study investigated the number of pollen grains and ovules, pollen grain germination on stigmatic surface, pollination in the two types of flowers and the increase in length and weight of developing seeds.
MATERIALS AND METHODS
Experiments were done in the Faculty of Agriculture of Miyazaki University in 2001, 2002 and 2003. In 2001 and 2002, 10 non flowering possible sibs that were naturally established in a field of Miyazaki University and had two to three lateral shoots were transplanted into 4 L plastic pots stuffed with volcanic ash soil obtained from the study site. A 14N-2.2P-5.8 K liquid fertilizer was applied every two week (5 g L-1). Pots were kept in a plasticfilm greenhouse with its sides kept open throughout the study. In 2003, plants were obtained from a field of Miyazaki University, Miyakonojyo city, Miyazaki and Kyoto University, Kyoto. In these fields, P. oleraceae are common weeds. Plants were transplanted into the field of Miyazaki University.
Number of pollen grains and ovules and pollen germination: In August 2001, the number of ovules and pollen grains per flower in 20 flowers from 10 plants (two flowers per plant) were counted using a binocular microscope (Nikon, SMZ-U). Pollen grains were taken from anthers before dehiscence. Pollen to ovule ratio was calculated for each flower. Percentages of mature pollen grains were assessed for five flowers from five plants by staining with acetocarmine solution immediately after opening. Germination of pollen grains on the stigmatic surfaces was assessed as follows. Shoots with flowers were cut from the plants when the petals emerged from the flower buds in the early morning. Petals and stamens were removed from the flowers using a binocular microscope. Following the emasculation, no pollen grains were confirmed to be on the stigmatic surfaces. Twenty pollen grains from the same flowers were placed on a stigmatic surface with distances of more than five times the diameters of the pollen grains between these grains when the pistils fully expanded. Shoots with the pollinated pistils were placed in a plastic case with the cut ends immersed in distilled water. Pistils were put in FAA at two four and six hours after pollination. Five pistils were used for each time. After more than two days, pistils preserved in FAA were placed on slide glasses. A drop of a solution of 0.1% aniline blue dissolved in 0.1N K3PO4 was then placed on a pistil and the pistil was softly squashed under a cover slip and was observed using a microscope (Nikon, Optiphoto 2x100) with an ultraviolet (UV) source or a light source from a halogen lamp. Morphologies of germinating and non-germinating pollen grains observed under a UV source were contrasted with those under a light source from a halogen lamp. Germinating and non-germinating pollen grains were counted under the both types of light source. A pollen grain was considered to have germinated when the length of the pollen tube was equal to or longer than the diameter of the grain. Germination percentage of the pollen grains was defined as the number of pollen grains germinating/20x100. Percentage data were arcsin-transformed and analyzed by analysis of variance (ANOVA) of a fixed model.
Seed production in flowers with two types of pistils: In August 2003, flowers with two types of pistils were observed and photographed. The lengths of styles and stamens were measured for 20 flowers from each type of pistil. The lengths of styles and stamens were compared between the flowers with each type of pistil by the Student's t-test. For assessment of the difference in the number of pollen grains on the stigmatic surfaces between the two types of flowers, plants were covered by a mosquito net. At full opening of the flowers, the number of pollen grains on the stigmatic surface was counted for 20 pistils per flower with each type of pistil. On sunny days in August, the flowers with two types of pistils were treated as follows: 1) styles were removed when petals began to emerge from the buds; 2) hand-selfed when the flowers were fully opened; 3) emasculated when the flowers were fully opened and 4) allowed to open under a mosquito net covering. Although some kinds of pollinators visited P. oleraceae, the visitation was occasional. Therefore, open pollination was not established. All flowers treated were tagged. Ten flowers from ten plants were used for each combination of pistil type and treatment. On the next day of pollination, the ovaries were cut open and the number of enlarged growing seeds (fertilized) and withered ovules (unfertilized) were counted under a binocular microscope. Percentages of fertilized ovules were calculated for each flower. Percentage data were arcsin-transformed and analyzed by the two-way analysis of variance. Comparison between pistil types in each treatment was performed by the Student's t-test. On rainy days in August 2002, two flowers in each of 10 plants were tagged when petals emerged from the buds. Petals could be visually recognized, but these flowers did not open. On the next day, the ovaries were cut open and the number of enlarged developing seeds was counted. This procedure was done on two rainy days. The percentage of seed set was calculated. In July and August 2003, the number of opening flowers with exserted and included stigma lobes in seven plants from three different areas was counted daily from anthesis to 30 days after anthesis. Counts were skipped on the days when the flowers did not open due to rain. The total number of flowers with each type of pistil that opened by 30 days after anthesis was calculated.
Development of seeds: In August 2001, the flowers were pollinated with self pollen grains and were tagged. Opening flowers and capsules at different days after pollination were taken and the ovules or developing seeds were excised. Length of ovules or developing seeds was measured using a binocular microscope. More than 20 ovules or developing seeds were measured for each day after pollination. The procedures to measure the weights of the developing seeds were as follows: flowers or capsules were cut at the peduncles and the ovary walls or pericarps of these were removed. The weights of these flowers or capsules (a) were measured. The ovules or growing seeds were removed from these flowers or capsules immediately after this measurement. The weights (b) of these flowers or capsules were measured again. The number of ovules or growing seeds in these flowers or capsules was counted. The weight of an ovule or a growing seed was estimated by (a - b)/(number of ovules or growing seeds). This procedure was replicated 10 times each day after pollination. Also, the colors of the seed coats were observed.
RESULTS
Number of pollen grains and ovules and pollen germination: The number of ovules and stamens varied among flowers; the numbers of ovules, stamens per flower and pollen grains per anther were 81±15, 9.6±1.2 and 81±18 (average±SD), respectively. The average of pollen to ovule ratio was 8.8. The pollen grains were uniform in their morphologies with diameters of 70±5.5 μ (average±SD) and with 100% maturity. At four hours after pollination, the appearance of germinated pollen grains was clearly different from that of non-germinating grains (Fig. 1). Non-germinated pollen grains were larger and fluoresced. Germinated pollen were smaller and did not fluoresce. Two hours after pollination, most (82%) but not all pollen grains had germinated (Fig. 2). Percentage of germinated pollen grains after 4 and 6 h was equivalent (94, 93%, respectively), thus pollen grain germination could be assessed after 4 h. Pistils became more transparent and soft by preserving in FAA and made observations easier without using a clearing or softening agent (Fig. 3).
| Table 1: | Length of styles and stamens in flowers with exserted and included stigma lobes of Portulaca oleraceae |
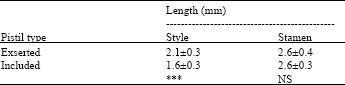 | |
| ***p<0.001, NS, not significant (p>0.05) | |
| Table 2: | Percentages of fertilized ovules in flowers with exserted or included stigma lobes following four treatments in Portulaca oleraceae. Treatment: (a) styles were removed when petals emerged from flower buds. (b) flowers were hand-selfed. (c) stamens were removed when flowers fully opened. (d) flowers opened and closed under a mosquito net covering |
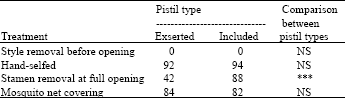 | |
| ***p<0.001, NS, not significant (p>0.05) | |
| Table 3: | Development in length and weight of P. oleraceae seeds with the days after pollination. Weights are the sum of 100 ovules or developing seeds |
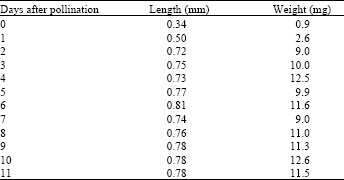 | |
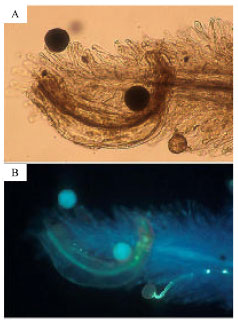 | |
| Fig. 1: | (A) Pollen grains of P. oleraceae on a stigma at 4 h after pollination. One of the three grains withered. Two grains were the same in morphology as those at pollination. (B) The identical stigma and pollen grains observed under an ultraviolet source. A pollen tube is elongating from a withered pollen grain. Two grains were fluorescing |
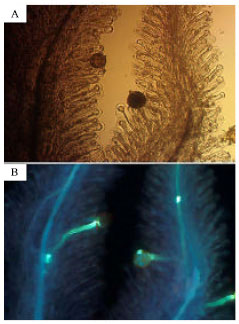 | |
| Fig. 2: | (A) Pollen grains of P. oleraceae on a stigma at 2 h after pollination. A pollen grain in the center is the same in morphology as that at pollination. (B) The identical stigma and pollen grains observed under an ultraviolet source (lower). A pollen grain in the center with fluorescence is germinating |
 | |
| Fig. 3: | (A) A stigma observed at four hours after pollination without preserving in FAA. (B) A stigma preserved in FAA at four hours after pollination and observed three days after pollination |
 | |
| Fig. 4: | A Campsomeris schulthessi visiting a flower of a P. oleraceae |
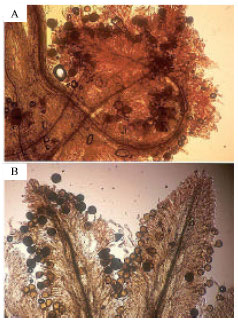
Seed production in flowers with two types of pistils: In the field, Campsomeris schulthessi, Pieris rapae, Zizeeria maha, Halictus aerarius foraging for P. oleraceae were observed (Fig. 4). The time of flower opening differed in different seasons. On sunny days from August to Sept., the flowers began to open at 06:00-07:00 h; they continued opening for 3-4 h and closed by noon. Light affected not only the opening of the flowers but also the closing of them. Flowers immediately closed when the plants were set in a room with decreased light. Anthers dehisced if the buds were physically stimulated the day prior to opening.
 | |
| Fig. 5: | (A) A flower with exserted stigma lobes. (B) A flower with included stigma lobes |
 | |
| Fig. 6: | (A) Reproductive organs in flowers with exserted stigma lobes. (B) Reproductive organs in flowers with included stigma lobes. A stigma in a flower with a folding pistil is contacting the anthers and it autonomously selfed with a large number of pollen grains |
Usually, anthers dehisced when the edges of the petals began to detach from each other as the flowers opened.
 | |
| Fig. 7: | A closing flower of a P. oleraceae. Petals are covering stamens and a pistil |
 | |
| Fig. 8: | An ovule in a flower at flowering (left). A withered ovule as a consequence of failure in fertilization (middle). A growing ovule that succeeded in fertilization sampled the next day of pollination (right) |
Generally on rainy days, petals emerged from the buds, but the flowers did not open. On cloudy days, flower opening depended on the intensity of the light. If flowers opened, it occurred later on cloudy days than on sunny days. Seed set in the flowers that did not open was 78%. Plants bore two kinds of flowers (Fig. 5). In one the styles elongate and the stigma lobes spread above the anthers. In the second the style does not elongate and the stigma lobes do not spread and remained within and contacted the open anthers. Self pollination was minimal in the former and common in the latter (Fig. 6). There were significant differences in length of styles between the two types of flowers (Table 1). The numbers of pollen grains on the stigmatic surfaces of the former and the latter were 14±7 and 178±85 (Mean±SD), respectively. These were significantly different at p = 0.01 by the t-test. Petals bent inside covering sexual organs at closure of both type of flowers (Fig. 7). In flowers with exserted stigma lobes, stigma lobes were pressed into anthers by petals. After closure of flowers, stigmas were dusted with pollen grains. One day after hand self-pollination, most ovules were approximately double in size compared to ovules at flower opening (Fig. 8). In contrast, ovules in flowers with their styles removed before opening had withered the next day. Therefore, successful fertilization could be judged the day after flowering. Analysis of variance for the percentage of enlarged ovules indicated significant differences between pistil types and among treatments (p<0.001). The interaction between treatments and pistil types was also significant. Hand-self pollination produced >90% seed set and seed set in bagged flowers was 84 and 82% (Table 2). However, the percentage of fertilized ovules was significantly different between the two types of flowers when the stamens were removed. The numbers of flowers that opened over a 30 day period were 1080±290, 780±270 and 997±292 (Average±SD) per plant (n = 6) for plants from Miyazaki Univ., Miyakonojyo city and Kyoto, respectively, of which 83, 66 and 87% had included stigma lobes, respectively.
Development of seeds: Seeds of P. oleraceae developed rapidly following pollination, the length and fresh weight of the developing seeds became maximum at three days after pollination and then the increase terminated (Table 3). Developing seeds were white until 5 days after pollination. The developing seeds then changed into light brown to dark brown. They were black at 10-11 days after pollination and were ready to scatter.
DISCUSSION
The pollen to ovule ratio suggested that P. oleraceae were autogamous by Cruden's indicator (Cruden, 1977). In P. oleraceae, germination of the pollen grains could be assessed on the stigmatic surfaces using a microscope with UV source and with a light source from a halogen lamp based on the difference in the morphologies between germinating and non-germinating pollen grains. Because some germinating pollen grains were difficult to discriminate from non-germinating ones at two hours after pollination, assessment of germination at two hours after pollination was not recommended. Assessment at more than four hours after pollination was recommended. In this experiment, pollen grains of P. oleraceae showed considerably high viability in germination on stigmatic surfaces.
In P. oleraceae, flowers with different status of pistils at full opening of flowers coexisted in each plant. The styles of flowers with included stigma lobes were significantly shorter than those of flowers with exserted stigma lobes; stigma lobes can not spread in the former flowers because anthers with sticky pollen grains stick stigma lobes, whereas stigma lobes freely spread over the anthers in the latter flowers. The difference in pollination success between these flowers was easily recognized by the situations of the stigma lobes and the anthers in the flowers. Only a slight difference in the percentages of fertilized ovules was found between flowers with included stigma lobes that opened and closed in bags and those of which the stamens were removed at full opening of the flowers. In contrast, the percentage of fertilized ovules in flowers with exserted stigma lobes that opened and closed in bags was approximately double that of those with removal of the stamens at full opening of the flowers. Therefore, flowers with included stigma lobes autonomously selfed with a large number of self pollen grains by the time of full opening of the flowers. On the other hand, self-pollination was not effectively done in flowers with exserted stigma lobes through the stage from flower buds to full opening of the flowers. Some plant species insure seed production under pollinator-free environment by autogamy (Kalin-Arroyo, 1973; Rathcke and Real, 1993; Klips and Snow, 1997; Solbrig and Rollins, 1977; Kalisz et al., 1999). Autonomous self-pollination is performed at the bud stage (Nickrent and Musselman, 1979; Gandawidjaja and Arditti, 1982), or at late stages of anthesis (Klips and Snow, 1997; Kalisz et al., 1999; Arathi et al., 2002). In tribe Collinsieae, flowers that autonomously self at early and at late stages of anthesis coexist to increase the outcross (Armbruster et al., 2002). In flowers with exserted stigma lobes of P. oleraceae, a small number of self pollen grains stuck to the stigmatic surfaces by full opening of the flowers and another self-pollination occurred when the flowers closed. Thus, P. oleraceae increase the possibility of outcross by flowers with exserted stigma lobes. In Verbascum thapsus, delayed selfing under pollinator exclusion resulted in only 75% of the seed set obtained with natural pollinators (Donnelly, 1998). In Hibiscus laevis, delayed autonomous selfing resulted in production of a half of the number of seeds compared to hand-pollination. Similarly, delayed selfing did not effectively produce seeds compared to hand-selfing in P. oleraceae (Table 2), but seed set was doubled by delayed selfing in flowers with exserted stigma lobes. Because visitation by pollinators are occasional, P. oleraceae assure seed production by boring flowers that autonomously self and those with delayed selfing. Mulligan (1972) found no pollinators for P. oleraceae in a study at Ottawa, Canada, over three years. Mulligan (1972) and Zimmerman (1976) inferred that cross-pollination is achieved by wind. Matthews et al. (1993) were negative to that idea because pollen grains of P. oleraceae were sticky. I also observed that pollen grains of P. oleraceae were very sticky and usually formed clusters. The characteristic did not fit to wind pollination. Actually, I observed pollinators in my experiment.
Light strongly affected flower opening. However, opening of petals was not indispensable for seed production. Because flowers that did not open petals on rainy days produced seeds and no evidence of reproduction by apomixis was found (Zimmerman, 1977; Kim and Carr, 1990), P. oleraceae could produce seeds cleistogamously. In P. oleraceae, the time from pollination to maturity of the seeds was rather short. On the other hand, development of seeds of P. oleraceae is affected by temperature; the seeds develop faster at higher temperatures (Nouguchi and Nakayama, 1974). Therefore, the speed of development of the seeds may differ in different areas or in different seasons. Thus, P. oleraceae had an outstanding ability to produce seeds with a large number of flowers that surely succeed in pollination in each individual, a number of ovules in each flower, a high germination ability of pollen grains and rapid maturation of seeds. This highly vigorous seed producing system is a factor that enables P. oleraceae distribute worldwidely as a hard-to-control weed. On the other hand, it is necessary to investigate the fate of the mass-produced seeds: seed dispersion, germination and seedling establishment, to figure out the reproductive ability of P. oleraceae.









Dan Eisikowitch Reply
Dear Sir
I read just the abstract and I wonder if the two types of flowers mentioned in the paper, are on the same plant or on two different plants.
Sincerely
Dan Eisikowitch