Research Article
Postharvest Changes in Some Physiological Traits and Activities of Ammonia-assimilating Enzymes in Lettuce During Storage
Not Available
Toshiyuki Matsui
Not Available
Haruo Suzuki
Not Available
Yusuke Kosugi
Not Available
Among the leafy vegetables, lettuce (Lactuca sativa L.) is the most important crop over the world both commercially and in home gardens. Undoubtedly, it is the world’s most popular salad vegetable. Moreover, it contains high percentage of minerals and vitamins, such as vitamin A and C and being low in calories is really necessary for any diet. However, this vegetable is highly perishable and soon after harvest start to deteriorate. It losses weight, texture, flavor, nutritive value and appearance (Schulz, 1989). In other word, it losses quality and potential storage life. In this regards, both time and temperature are the important factors in postharvest product deterioration. At 0°C, head lettuce can be held in good condition for 2 to 3 weeks and the storage life at 3.3°C is only about half at that at 0°C (Anonymous, 2004).
Quality degradation of lettuce is a result of combined symptom of some physiological and biochemical characters when the heads are exposed to ambient temperature. These symptoms are mainly characterized by browning of stem butt, wilting, leaf curling and margin chlorosis and in severe cases whole leaf senescence. Again, these visible changes are accompanied by some active metabolic transformations to the nutrients that stored in the leaf during development (Buchanan-Wollaston et al., 2003). In the course of senescence, a mature leaf gradually losses its photosynthetic capacities and instead of providing a source of carbon, becomes a source of nitrogen and other mineral elements (Turgeon, 1989). Although growing plants need to assimilate nitrogen, ultimately in the fully reduced form, excessive concentration of ammonia or ammonium ions are toxic to plants (Givan, 1979). Inorganic nitrogen is assimilated into amino acids, namely glutamate, glutamine and asparagine which play a pivotal role as N- transport compounds in plants (Lea and Miflin, 2003). Enzymes of nitrogen metabolism have generally been shown to lose their activity to a greater or lesser extent during this period (Peoples and Dalling, 1988), the exception being glutamate dehydrogenase (GDH, EC 1.4.1.2) which shows a substantial rise in activity, especially in the dark (Lauriere, 1983). The enzyme glutamine synthetase (GS, EC 6.3.1.2) plays a central role in nitrogen metabolism and is characterized by its high affinity for ammonia (Miflin and Habash, 2002). It also catalyses the first step in amino acid biosynthesis from ammonia (Miflin and Lea, 1980). It binds ammonia as glutamine in the presence of glutamic acid with the hydrolysis of ATP. As a result, the accumulation of ammonia in senescing leaves has been shown some promise with almost complete disappearance of GS (Peters and Van Laere, 1992). The product glutamine is a major source of nitrogen for the synthesis of all other nitrogen containing compound like amino acid and nucleotide. The ability to assimilate and dispose of excessive level of ammonia is no doubt useful to the plant as means of preventing ammonia toxicity. It has been reported that when ammonia accumulate in high concentration that become toxic to plant cell. In such condition, plant tissues undergo appropriate modification in their metabolism to prevent ammonia toxicity or at least converted to an innocuous form (Givan, 1979; Yamata et al., 1984; Loyola-Vargas et al., 1988). In that situation, the possible role of GDH in ammonia detoxification process is supported by the findings that the enzyme is induced by high level of ammonia. This enzyme mediates the combination of α-ketoglutarate with ammonia to yield glutamic acid and also readily catalyses the reverse reaction leading to oxidative deamination of glutamate (Givan, 1979). Moreover, it has long been known that ammonium is highly inhibitory to the TCA cycle and this inhibition is caused by reversal of oxidative deamination of glutamate so that α-ketoglutarate is trapped from the cycle as glutamate (Lehninger, 1964).
It is therefore, the goal of this study is to better understand both physiological and biochemical characteristics which are the basis of deterioration process of lettuce during storage. Here, we report changes in some physiological traits related to the shelf life of lettuce and activities of enzymes in ammonia assimilation, especially GS and GDH and the accumulation of ammonia in the leaf and midrib portion of two lettuce cultivars during storage at 20°C for 10 days. Relationship between these parameters would provide an overall view of the postharvest life of lettuce.
Plant materials: Two popular crisphead lettuce cultivars, ‘Shizuka’ and ‘Cisco’, were grown under commercial conditions in Kagawa prefecture, Japan. Horticulturally mature heads of these two lettuce cultivars were harvested in December, 2004. Immediately after harvest, the heads were brought to the Postharvest Laboratory and stored at 20°C in perforated polyethylene bags. After each storage treatment, fully expanded leaves were chosen from the middle of the heads, eliminating the outermost leaves and immature inner leaves. The midrib tissues were separated from leaves and both the leaf and midrib portions were cut into small pieces (ca. 2x2 cm) and immediately stored at -30°C until needed for analysis.
Weight loss: The percentage of weight loss was calculated by the decrease in weight to the initial weight of the respective days and multiplied by hundred.
Color assessment: Color reading of lettuce heads were performed with a chromameter (Nippon Denshoku Kogyo Co. Ltd.,) equipped with an 8 mm measuring head and a C illuminant. The meter was calibrated using the manufacturer’s standard white plate. Color changes were quantified in ‘L’ ‘a’ and ‘b’ color spaces. ‘L’ refers to the lightness of the head and ranges from black = 0 to white = 100. A negative value of ‘a’ indicates green, while a positive number indicates red-purple color. On the other hand, positive and negative values of ‘b’ indicate yellow and blue color, respectively (McGuire, 1992). The results were expressed as hue angle (h° = tan-1 (‘b’/‘a’) when ‘a’>0 and ‘b,>0 or h° = 180° + tan-1 (‘b’/’a’) when ‘a’<0 and ‘b’>0) and calculated from the ‘a’ and ‘b’ values (Lancaster et al., 1997). On each head, three readings were taken from different portions and averaged the value.
Respiration rate measurement: Respiration rate was measured at 48 h interval by the production of carbon dioxide from an intact lettuce head sealed for 1 h in a 10 L desiccator jar held at 20°C. Before placing the head in the desiccator jar, it was weighed carefully. Ten milliliter gas sample was taken after 1 h and injected to a TCD gas chromatograph equipped with a 1 m activated charcoal column at 70°C (GC-8AIT, Shimadzu Co. Ltd.). The results were expressed as mL CO2 kg-1 h-1.
Enzyme extraction: Approximately 5 g lettuce sample from leaf and midrib portion was homogenized in ice cold condition (ca. 0-4°C) with 1% Polyvinylpolypyrrolidone (PVPP) proportional to the sample weight, 1 g sea sand and 5 mL Buffer A using a mortar and pestle. Extraction was performed as described by Hurst and Clark (1993), in which Buffer A contained 50 mM tris-HCl (pH 7.6), 10 mM MgSO4A7H2O, 1 mM EDTA, 1 mM dithiothreitol (DTT), 12 mM 2-mercaptoethanol, 5 mM L-glutamate and 100 mL L-1 glycerol. The homogenate was squeezed through four layers of cotton cloth. The residual tissues were re-extracted with an additional 5 mL of the same buffer and the filtrate was centrifuged at 11,000xg at 2°C for 10 min. The resulting supernatant was used for enzyme assay.
Enzyme assay: The enzymatic activities were assayed in a total volume of 1 mL assay mixture. The activity of GS was determined with 80 mM Na-L glutamate, 100 mM tricine-KOH buffer (pH 7.0), 6 mM hydroxylammonium chloride, 20 mM MgSO4A7H2O, 1 mM diethylenetriamine pentaacetic acid (DTPA), 8 mM ATP and 8 mM mercaptoethanol. After incubating at 35°C for 8 min, the reaction was stopped by the addition of 1 mL ferric reagent that contains 0.37 M FeCl3, 0.67 M HCl and 0.2 M trichloroacetic acid (TCAA). GS activity was measured using a double beam spectrophotometer (Shimadzu model UV-150-02) at 540 nm and soluble protein contents was determined following the method of Lowry et al. (1951) using bovine serum albumin as the standard.
Both aminating and deaminating activities of GDH were determined spectrophotometrically (Shimadzu model UV-150-02) at 350 nm according to NADH oxidation or NAD+ reduction maintaining a temperature of 30°C. For GDH amination, a total volume of 1.0 mL assay mixture contained 10 mM α-ketoglutaric acid, 100 mM tris-HCl (pH 8.0), 200 mM NH4Cl, 1mM CaCl2 and 0.2 mM NAD(P)H. The reaction was started by adding 200 μL crude extract. Likewise, the 1.0 mL assay mixture for GDH deamination consisted of 100 mM L-glutamate, 100 mM tris-HCl (pH 9.3), 1 mM NAD(P)+ and 0.5 mM CaCl2 . The reaction was started with the addition of 200 μL crude extract. Blank controls were performed omitting individual substrates. One unit of GDH activity is defined as the reduction or oxidation of one micromole of coenzyme (NADPH / NADP, respectively) per min at 30°C.
Ammonia assay: To assess ammonia, 2 g sample from each portion of lettuce head was extracted with 10% trichloroacetic acid at 1:10 ratio in an ice bath (0-4°C) and centrifuged at 11,000xg for 10 min. Ammonia content was assayed as described by Kun and Kearney (1974), where 1 mL assay mixture contained 200 μL 0.5 M tris-HCl buffer (pH 8.0), 100 μL 0.1 M 2-oxoglutarate (pH 7.4), 30 μL 8 mM β-NADH, 150 μL distilled water and 500 μL of neutral extract sample. The decrease of NADH, as determined by the change of extinction at 365 nm, was used as a measure of the reaction.
Statistical analysis: A randomized complete block design was used with three replications. The level of significance was calculated from the F-value of ANOVA. Linear correlation was used to evaluate the relationship between enzyme activities and ammonia contents.
Weight loss: Weight loss was found to be high after 2 days of storage in both the cultivars. This level declined in the next 2 days and after that no significant change was found throughout the storage period (Fig. 1). However, at the end of the storage period, more than 17 and 18% weight losses were recorded in ‘Shizuka’ and ‘Cisco’ to their initial weight at harvest, respectively.
 | |
| Fig. 1: | Weight loss of two lettuce cultivars (‘Shizuka’ and ‘Cisco’) stored at 20°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols |
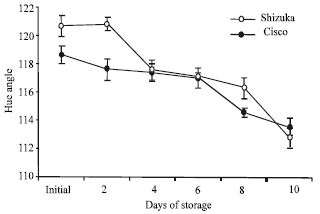 | |
| Fig. 2: | Hue angle change of two lettuce cultivars (‘Shizuka’ and ‘Cisco’) stored at 20°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols |
Hue angle change: The hue angle decreased with storage time in both cultivars (Fig. 2). However, the cultivar ‘Cisco’ showed a lower hue angle value than ‘Shizuka’. The decrease in hue angle value revealed a corresponding decline in green color.
Respiration rate: Both cultivars showed high respiration rate immediately after harvest and declined successively as storage progressed (Fig. 3). Cultivar ‘Cisco’ exhibited relatively high rate of CO2 production than ‘Shizuka’, though its initial rate was low.
Enzyme activities: The activities of GS and GDH in leaf and midrib portion of two lettuce cultivars are presented in Fig. 4 and 5, respectively. In case of GS, leaf portion showed significantly higher activity than midrib. The activity decreased gradually with the progress of storage duration except for a transient increase on 6-day and 8-day in the cultivar ‘Shizuka’ and ‘Cisco’, respectively.
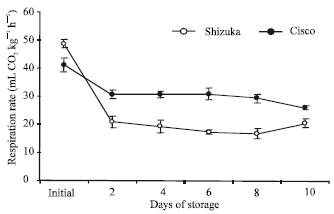 | |
| Fig. 3: | Changes in respiration rate of intact lettuce head of the cultivars ‘Shizuka’ and ‘Cisco’ during storage at 20°C for 10 days. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols |
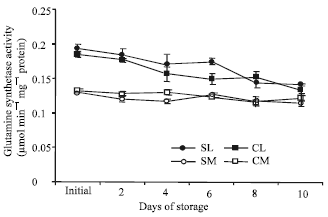 | |
| Fig. 4: | Changes in the activities of glutamine synthetase in the leaf and midrib portions of two lettuce cultivars stored at 20°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols. Legend: SL = ‘Shizuka’ leaves; SM = ‘Shizuka’ midribs; CL = ‘Cisco’ leaves; CM = ‘Cisco’ midribs |
However, no significant change was observed in the midrib portion.
GDH amination activities increased throughout the postharvest period with some fluctuations on day 2 and 4, whereas deamination activities were fluctuating up to the end of the storage period in both cultivars and portions. Moreover, GDH amination activity was remarkably higher than that of GDH deamination. Although ‘Shizuka’ midrib showed comparatively lower activity to its leaf portion, both portions of ‘Cisco’ showed more or less similar pattern. For GDH deamination, ‘Shizuka’ midrid showed the highest activity.
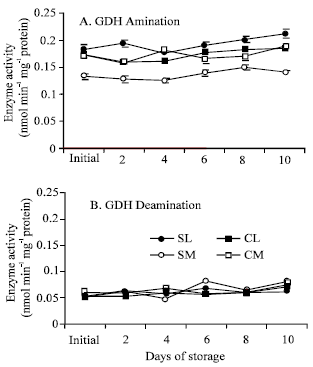 | |
| Fig. 5: | Changes in the activities of (A) GDH amination and (B) GDH deamination in the leaf and midrib portions of the cultivars ‘Shizuka’ and ‘Cisco’ held at 20°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols. Legend as shown in Fig. 4 |
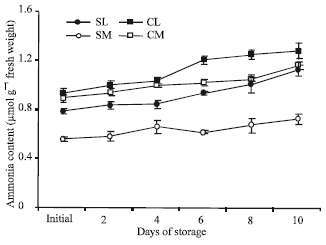 | |
| Fig. 6: | Ammonia content in the leaf and midrib portions of two lettuce cultivars (‘Shizuka’ and ‘Cisco’) stored at 20°C for 10 days. Vertical bars indicate SE, which, when absent, is concealed by the graph symbols. Legend as shown in Fig. 4 |
Ammonia content: The content of ammonia increased over the storage period in both portions of the two cultivars (Fig. 6). However, both portions of ‘Cisco’ showed significantly higher amount of ammonia content than ‘Shizuka’.
| Table 1: | Correlation coefficient (r) values between the enzyme activities and ammonia content in the leaves and midribs portions of two lettuce cultivars stored at 20°C for 10 days |
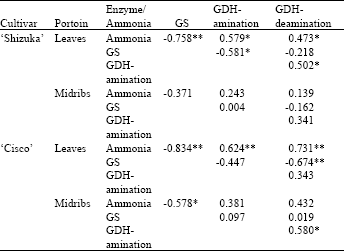 | |
| GS = glutamine synthetase, GDH = glutamate dehydrogenase | |
| *, **denote significant at p<0.05 and p<0.01, respectively; n = 18 | |
In the midrib of ‘Shizuka’, ammonia content was not so high, but followed the same trend to its leaf portion. Accumulation of ammonia in leaf portion was noticeably started after 4 days of storage. The increase of ammonia at the end of the storage period was calculated more than one and a half times to its initial content mainly in leaf portion.
Correlation between enzyme activities and ammonia content: A highly significant negative correlation was observed between the GS activity and ammonia content in the leaf portion of both cultivars (Table 1). A positively significant relationship was found between ammonia content and GDH amination and deamination in the leaf portion of both cultivars, however, the level of significance was higher in ‘Cisco’ than ‘Shizuka’. GS and GDH showed overall an inverse relationship. Again, GDH amination and GDH deamination showed a positive correlation which was significant in the leaf and midrib portion of ‘Shizuka’ and ‘Cisco’, respectively.
Leafy vegetables like lettuce start to deteriorate soon after harvest. In this study, the first symptom of quality deterioration was imposed with the wilting of leaves and loss of turgor due to loss of water at the second day of storage which continued up to the end of the storage duration (Fig. 1). It is likely that the high water content and succulent nature of lettuce favor its susceptibility to weight loss. A remarkable weight loss was also previously reported in lettuce (Siomos et al., 2002) and in chinese cabbage stored at 20°C (Porter et al., 2003) which confer that leafy vegetables are more susceptible to water loss.
Furthermore, both the lettuce cultivars showed a high rate of senescence (Fig. 2) coupled with susceptibility to many physiological disorders. However, the changes in hue angle were not evident until the second day of storage with a lower hue angle value in ‘Cisco’. This result was similar with the result of Garratt et al. (2005) where they found that lettuce head became yellow and necrotic after 3-7 days of storage under 19±2°C. A rapid decline in respiration rate was observed after 48 h of storage and decreased slowly thereafter (Fig. 3). The high initial respiration rate in both cultivars could be a consequence of detachment from the source of respiratory substrate. It is believed that the supply and control of metabolic energy is vulnerable to disruption which quickly leads to loss of normal respiratory function. In addition, Geysen et al. (2005) reported that the respiration rate of stored lettuce is significantly influenced by temperature, carbon dioxide and oxygen concentration.
The activity of GS decreased gradually over the storage period in both portions and cultivars (Fig. 4). The decline of GS activity in leaf portion was calculated as 26 and 28% to its initial level for ‘Shizuka’ and ‘Cisco’ after the end of the storage period, respectively. However, no significant change was observed in the midrib portion of both cultivars. Apart from the determination of GS1 (cytosolic) and GS2 (chloroplastic) separately, it may be described as midrib portion contain very low amount of GS2 which is highly contributing in the total GS activity measured in this experiment. Again, it has been reported that in lamb’s lettuce stored at 20°C, the content of sucrose declined rapidly causing a remarkable loss in carbon substrates (Enninghorst and Lippert, 2003). Hence, the decline in GS activity is a response to harvest and may be a consequence of autophagic processes (Journet et al., 1986) induced by carbohydrate deprivation and/or ammonia accumulation and end product control (Rhodes et al., 1976). Moreover, metabolic regulation of GS expression in plant is controlled by the relative abundance of carbon skeletons versus amino acids (Oliveira and Coruzzi, 1999). Therefore, the scarcity of carbon sources could possibly have a negative role on GS activity. On the other hand, GDH activity increased during the storage period except for a slight decrease in both portions of ‘Shizuka’ on the fourth day. However, the GDH amination activity was remarkably higher than the deamination activity which showed similarity with the experiment on asparagus (Enriquez et al., 2000). The increase in GDH aminating activity could be a result of ammonia produced by protein hydrolysis. In this process, large amount of ammonia may be produced where deamination of amino acid usually happened and the carbon skeleton is then being used as respiratory substrates. Though the increase in GDH deamination is not so high (Fig. 5), the slight increase in the activity indicates that the enzyme also operates in the direction of energy generation by supplying the cell with carbon skeletons (Cammaerts and Jacobs, 1985). It is well known that GDH amination dealt with high level of nitrogen produced and GDH deamination can provide the cell with energy ensuring the continued operation of the citric acid cycle in the face of carbohydrate deprivation (Robinson et al., 1991). The activity of GDH may possibly serve as an adaptation to detoxify the ammonium accumulated during storage when the activity of GS decreased. The rise in GDH activity along with a corresponding decline in GS activity of this experiment was confirmed by similar result in other green vegetables like asparagus spears (Enriquez et al., 2001) and broccoli (Baclayon et al., 2004).
The trend of ammonia accumulation in both cultivars increased with the storage time (Fig. 6). However, the accumulation was only one and a half times higher than the initial level. There are a number of different mechanisms by which ammonia may be produced within the plant cell. In leaf, ammonium ions may also be produced by the reduction of nitrate or by the conversion of glycine to serine in photorespiration (Lea et al., 1989). The initial content and successive accumulation of ammonia was relatively higher in ‘Cisco’ than ‘Shizuka’. The reason could be the cultivar ‘Cisco’ is more efficient in nitrogen uptake and utilization than ‘Shizuka’. As ammonia is thought to be toxic to the plant, this could be one of the reasons for the rapid deterioration of the cultivar ‘Cisco’. The highly significant negative correlation (Table 1) between GS and ammonia in the leaf portion clearly indicates that accumulation of ammonia is an effect of the declining activity of GS. Such relation has also been observed in other green vegetables like asparagus spears (Enriquez et al., 2001) and broccoli (Baclayon et al., 2004). A marked inverse relationship which was demonstrated between GS and GDH strongly suggests that they are subject to concurrent regulation by a common metabolite and is confirmed by the findings of Rhodes et al. (1976).
Based on the above discussion, it can be summarized that lettuce is very susceptible to water loss and showed high rate of senescence during storage. The cultivar ‘Cisco’ exhibited comparatively higher weight loss and respiration rate with a lower hue angle value. The accumulation of ammonia and the decrease in GS activity is a response to significant stress after harvest. The negative correlation between GS and GDH might be influenced by ammonia accumulation in the tissue due to the breakdown of chlorophyll and proteins. Further researches at molecular level are needed for complete understanding of the deterioration process and the expression of genes that are involved in this process during storage.
We are thankful to the Ministry of Education, Culture, Sports, Science and Technology, Japan for financial support under scholarship program for foreign students.