ABSTRACT
A bacterium isolated from old tire sample in Alexandria, Egypt, was shown to be able to utilize natural rubber and other isoprenoide compounds as a sole source for carbon and energy. Taxonomic analysis of the strain based on morphological and physiological studies indicated that the bacterium belongs to nocardioform actinomycete Nocardia sp. strain-MBR. Degradation behavior as well as SEM examinations indicated that the strain grow adhesively and depends on direct contact with the rubber substrate so belongs to the CMN group. Semicontinuous degradation of NR granules resulted in approximately 7% increase in percent CO2 release during growth. Schiff`s reagent staining revealed that the bacterium showed a higher colonization efficiency on small and treated pieces of NR-latex gloves, while a lower colonization efficiency when grown on large and nontreated NR-latex gloves. Formation of bacterial films and occurrence of compounds containing aldehyde groups during cultivation was also confirmed. Degradation of synthetic rubbers as well as other acyclic isoprenoids and compounds of analogous structure namely, phytol, squalene, squalane and prostane was recognized indicating that the bacterium has the metabolic capability to utilize these compounds as sole carbon source.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/biotech.2006.234.239
URL: https://scialert.net/abstract/?doi=biotech.2006.234.239
INTRODUCTION
Biodegradation is a natural process by which organic compounds in the environment are converted to simpler compounds, mineralized and redistributed through the elemental cycles. Interestingly, microorganisms play a central role in the process of biodegradation. Natural Rubber (NR) is a biopolymer (polyisoprene) that is synthesized by many plants and some fungi. It has been commercially produced from Hevea brasiliensis trees at a level of several million tons per year. Narutal rubber is mineralized biologically and many reports on its biodegradability have been published (Rook, 1955; Leeflang, 1963; Tsuchii et al., 1985; Heisey and Papadatos, 1995; Subramananiam, 1995; Tsuchii, 1995; Jendrossek et al., 1997; Linos and Steinbuchel, 1998; Berekaa et al., 2000; Linos et al., 2000; Arenskotter et al., 2001; Linos et al., 2002; Arenskotter et al., 2004; Rose and Steinbuchel, 2005). With regard to the decomposition strategies, two different groups of rubber-degrading bacteria can be distinguished (Linos et al., 2000; Arenskotter et al., 2004). While bacteria forming clear zones (translucent halos) on latex-containing mineral agar have been repeatedly described (Jendrossek et al., 1997), only few representatives of the second, adhesive growing group were so far isolated and described (Tsuchii et al., 1985; Linos et al., 1998; Linos et al., 1999) and were classified into the so-called CMN group (Corynebacterium, Mycobacterium, Nocardia). Gordonia, Mycobacterium and Nocardia were identified as non-clear zone forming rubber-degrading bacteria that are dependent on direct contact to the substrate (Linos et al., 2000). Compared to clear zone forming rubber-decomposing actinomycetes, the adhesively growing bacteria represent the more powerful rubber-degrading bacteria. In the past they were perhaps not well recognized due to the methods applied for enrichment and screening.
It is assumed that degradation of the rubber backbone is initiated by oxidative cleavage of the double bond (Tsuchii et al., 1985; Tsuchii and Takeda, 1990; Linos et al., 2000; Bode et al., 2001; Rose and Steinbuchel, 2005; Braaz et al., 2005). Recently, similar mechanism of degradation was suggested by Bode et al. (2001) after analysis and identification of the degradation product by HPTLC and GPC during growth of gram-positives (Nocardia sp. DSMZ 43191 and Streptomyces coelicolor 1A) and gram-negative (A. colcoaceticus and Xanthomonas sp.) bacteria on natural and synthetic rubber.
Degradation of acyclic isoprenoids and compounds of analogous structure was intensively investigated because it comprises some basic reactions of isoprenoid degradation (Seubert et al., 1960; Yamada et al., 1975; Yamada et al., 1988; Berekaa and Steinbuchel, 2000). The degradation of acyclic isoprenoid compounds by members of the genus Pseudomonas was investigated by Seubert et al. (1960) who proposed citronellol pathway for degradation of these compounds. This pathway is based on the substitution of the β-methyl group with a carbonyl-group, generating a suitable substrate for β-oxidation. Recently, the degradation of squalane, squalene and pristane by members of the genus Mycobacterium was investigated by Berekaa and Steinbuchel (2000). They mentioned that the degradation of polyisoprenoid substrate is not crucial to the degradation of saturated and branched hydrocarbons such as squalane and pristane.
In this study, a bacterium that is able to utilize natural and tire rubber as a sole carbon source was isolated and characterized. Furthermore, the degradation and colonization of the bacterial isolate on treated as well as nontreated NR-latex gloves and NR granules and the effect of size of rubber substrate on colonization efficiency were investigated. Examination of the degraded rubber substrates was performed using SEM and staining with schiff’s reagent. Special emphasis was given to the potency of the nocardioform actinomycete Nocardia sp. strain-MBR to utilize different types of synthetic rubber materials and some acyclic isoprenoids namely, phytol, squalene, squalane and pristane.
MATERIALS AND METHODS
Culture medium: Cultivation was carried out in screw-capped Erlenmeyer flasks, with internal glass container, containing Mineral Salts Medium (MSM) and rubber as sole carbon source (Linos and Steinbuchel, 1998). The rubber materials were added in concentration of 0.5% (w/v). Natural Rubber (NR) granules were added directly and the entire media was autoclaved. All cultures were inoculated with cells obtained from 4-6 days preculture in Luria-Bertani complex medium which were washed twice with sterile saline before use. During incubation at 30°C, the cultures were agitated at 150 rpm on a rotary shaker. Squalane, pristane or squalene were added to the medium at final concentrations of 0.5% (w/v). To test growth on other acyclic isoprenoids namely; phytol, linalool, farnesol, cis-trans citral, citronellal, acetonylacetone and geranic acid, cells were exposed to a vapor of the respective compound delivered from sterile filter paper containing 50-100 μL of this compound and placed in the lid of Petri plate. Inoculated plates were incubated in an inverted position with the lid at the bottom at 30°C separately and in closed containers (Berekaa and Steinbuchel, 2000).
Microorganism: The bacterial strain used in this work was enriched and isolated from old tire rubber. Sample of old tire rubber was cutted into small pieces and incubated in 50 mL MSM with natural rubber granules (NR) as a sole C-source in 250 mL Erlenmeyer flasks that were incubated under shaked conditions. Growth was monitored by increase in turbidity and the change in substrate nature and color. For enrichment, 1 mL of the grown culture was transferred to a fresh medium containing the rubber material and left for incubation and the process was repeated several times. At the end, serial dilutions were made and inoculated on starch nitrate agar plates. The resulted colonies were further subcultured on starch nitrate agar medium for purification. Single pure colonies were subsequently tested for growth on MSM with NR granules as a sole C-source. The bacterium was characterized morphologically and physiologically as described in Bergey’s manual of systematic bacteriology (Lechevalier, 1989).
Mineralization of rubber substrate: Degradation and mineralization of rubber substrate, carried out in screw-capped Erlenmyer flasks with internal glass container, was estimated by determination of the amount of CO2 released during cultivation of cells on the rubber substrate. The released CO2 was trapped in Ba (OH)2 solution resulting in precipitation of CO2 as BaCO2. The decrease in alkalinity was determined by titration with 0.25 N HCl and compared to a non-inoculated control as described previously (Linos and Steinbuchel, 1998).
Staining of rubber-degrading colonies: The actively growing colonies of Nocardia sp. strain-MBR on the rubber surface were visualized clearly by staining with Schiff’s reagent (Ehrlich et al., 1948). At the end of the incubation period the rubber material was removed from flask, washed several times with saline and stained with the reagent (Tsuchii et al., 1985).
RESULTS AND DISCUSSION
Isolation and characterization of the rubber-degrading microorganism: The rubber-degrading isolate was enriched and isolated from old tire rubber material from Alexandria (material and methods). Morphologically, the cells are non-motile rod-shaped, with no aerial mycelia, that tends to form aggregates during early stages of growth. During incubation the cells are separated and in later stages the cells turns to coccoidal forms. The cells are gram-positive and tend to form red pigments. The cells showed adhesive growth on solid substrates and penetrating agar media such as starch nitrate agar or nutrient agar media.
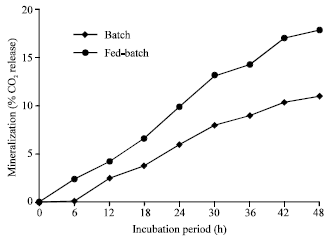 | |
| Fig. 1: | Fed-batch cultivation and mineralization of natural rubber granules by Nocardia sp. strain-MBR cells during different time intervals |
The bacterium was catalase- and oxidase-positive, grew optimally at 30°C. It grew on glucose, mannitol and mannose, producing acid. These results collectively indicated that the bacterium belongs to the group of nocardioform actinomycetes and nominated Nocardia sp. strain-MBR. The use of old tire rubber as a source for enrichment of the rubber-degraders has been recorded (Linos et al., 2002). The results also indicated that the bacterium could be classified among the first CMN group of bacteria that shows adhesive growth on rubber material and grow in direct contact to the rubber substrate (Linos et al., 2000).
Semicontinuous degradation (Fed-batch cultivation) of natural rubber: For mineralization and optimal degradation of natural rubber by nocardioform actinomycete Nocardia sp. strain-MBR, semicontinuous cultivation experiment was carried out. Two sets of flasks containing MSM and amended with natural rubber granules as a sole C-source were run paralle. Mineralization was determined in the first set after know time interval while, in the other set MSM was replaced by fresh medium after each time interval and the mineralization of the rubber material (expressed as % CO2 release) was estimated. Results presented in Fig. 1 indicated that the % CO2 release due to batch and fed-batch cultivation of Nocardia sp. strain-MBR was approximately 11 and 17.8%, respectively, with approximately 7% increase in mineralization of the rubber material for fed-batch cultivation in comparison to batch cultivation. Therefore, the positive contribution of the semicontinuous or fed-batch cultivation may be promising to establish biotechnological processes to the disposal of products made from synthetic or natural rubbers (Tsuchii et al., 1997; Kajikawa et al., 1991).
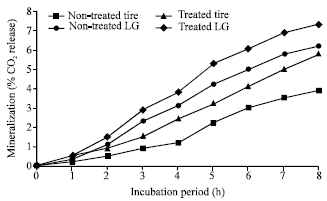 | |
| Fig. 2: | Mineralization (expressed as: %CO2 release) of treated and nontreated raw natural rubber and tire rubber by Nocardia sp. strain-MBR |
Mineralization of the NR-latex gloves as well as tire rubber substrates: In this experiment the ability of Nocardia sp. strain-MBR to degrade NR-latex gloves as well as some forms of tire rubber was investigated. For this purpose, 250 mL Erlenmeyer mineralization flasks with screw and containing 50 mL MSM were prepared and the rubber substrate was added at concentration of 0.5% (w/v). NR-latex glove substrate (treated and nontreated) was cutted in the form of small pieces of 1 cm2 in diameter and tire rubber (treated and nontreated) was added as large piece of 3 cm3 before addition to the MSM and autoclaving. The flasks were inoculated with 2 mL of a 6-days old preculture of Nocardia sp. strain-MBR cells that was previously washed with sterile saline solution 0.9% (w/v) and the mineralization was estimated at different time intervals.
The results presented in Fig. 2 showed that Nocardia sp. strain-MBR was able to degrade and mineralize NR-latex gloves and tire rubber. It was also recognized that the optimum mineralization level of purified latex gloves was 6.2 and 7.4, respectively, with approximately 1.2% increase in %CO2 release that was reached after 8 weeks of incubation. Indeed, results in Fig. 2 showed that the strain recorded 5.8 and 3.9% CO2 release during growth on treated and nontreated tires, respectively.
Colonization on NR-latex gloves: Colonization of the rubber-degrading strain Nocardia sp. strain-MBR on the surface of pre-treated and non-treated NR-latex glove was examined by Schiff,s reagent. Results in Fig. 3 showed that the actively growing colonies on the rubber surface were clearly visualized by staining the reagent and gave a clear purple color thus providing evidence for the occurrence of degradation products containing aldehyde groups during the degradation.
 | |
| Fig. 3: | Enhanced colonization and degradation of small sized NR-latex gloves (right) and large sized NR-latex gloves (left) by Nocardia sp. strain-MBR |
It is assumed that degradation of the rubber is initiated by oxidative cleavage of the double bond resulting into oligomeric derivatives with aldehyde and keto groups at their respective ends that are presumably degraded by reaction involving β-oxidation. Tsuchii and Takeda (1990) found that the degradation of cis-1,4-polyisoprene rubber is by oxygenative attach of the polymer. Furthermore, all possible mechanisms of rubber degradation were intensively discussed by Rosa and Steinbuchel (2005) and mentioned the role of oxygenases in degradation of rubber and other isoprenoid compounds. Recently, Heme-containing enzyme was isolated by Braaz et al. (2005), supporting the fact that most of oxygenases are belong to the heme-containing proteins. Further, colonization efficiency was moderately increased after pretreatment and removal of antimicrobicidial agents from NR-latex gloves. Interestingly, Berekaa et al. (2000); recorded the enhancement of rubber degradation by the removal of antioxidants and other toxic compounds prior to introducing of the rubber substrate to microbial degradation. Therefore, the degradation of rubbers after removal of antimicrobial substances that were added for different purposes by the manufacturing companies will provide a new approaches for a future microbial treatment of rubber waste by combining chemical and biological methods.
Effect of size of NR-latex gloves on colonization efficiency: It is well known that the growth of microorganisms in presence of insoluble C-source is commonly different than growth in presence of soluble one. Therefore, the effect of size of the rubber substrate may be critical in availability of the rubber substrate to microbial degradation. Staining with Schiff’s reagent was also used to examine the effect of size of rubber substrate on colonization efficiency. In this experiment, Nocardia sp. strain-MBR cells were cultivated in MSM and pre-treated NR-latex gloves as a sole source for carbon. Two different sizes of NR-latex gloves (with diameter of 1 and 4 cm2 in length and width, for each of the tested sizes) were used. At the end of incubation period, colonization was tested by Schiff,s reagent. Results in Fig. 3 showed that colonization efficiency was clearly influenced with the size of rubber substrate. Considerable increase in colonization efficiency was recorded when rubber substrate was applied in small pieces (diameter; 1 cm2) while; lower colonization efficiency was recorded in case of large size substrate (diameter; 4 cm2). A similar finding was observed during growth of Nocardia sp. 835A (Tsuchii et al., 1996) and Achromobacter sp. strain-NRB (Berekaa et al., 2005) on NR-latex gloves pieces.
SEM’s photos to show colonization of Nocardia sp. strain-MBR on NR granules and NR-latex gloves: To study colonization of NR substrate (NR granules and NR-latex gloves) by Nocardia sp. strain-MBR, scanning electron microscopy examination (SEM) was performed. For this purpose the rubber-degrading strain was cultivated in MSM with NR granules or NR-latex gloves as sole source of carbon. At the end of 3 weeks incubation period rubber substrates were subjected to examination. Results showed that the NR substrate turned orange due to bacterial growth on the rubber substrates. For further inspection, NR granules were subjected to SEM examination. SEM photos of the rubber granules in Fig. 4a (A and B) showed that the 3 weeks of incubation were sufficient to obtain dense microbial masses in the form of punches on the surface of NR granules in comparison with control (Fig. 4a, C). Furthermore, a compact biofilm was distinguished due to growth of the actinomycete cell on the surface of NR-latex gloves as shown in Fig. 4b (A and B) in comparison with the control (Fig. 4b, C). Similarly, a group of potent rubber degraders belongs to the nocardioform actinomycetes, genus: Gordonia, has been isolated and intensively investigated (Linos et al., 2000; Arenskotter and Steinbuchel, 2004). Member of this group exhibited a similar growth behavior to the nocardioform actinomycete, Nocardia sp. strain-MBR, especially during growth on the solid substrates.
Degradation of synthetic rubbers: In this experiment the ability of Nocardia sp. strain-MBR to degrade different types of synthetic rubbers was tested.
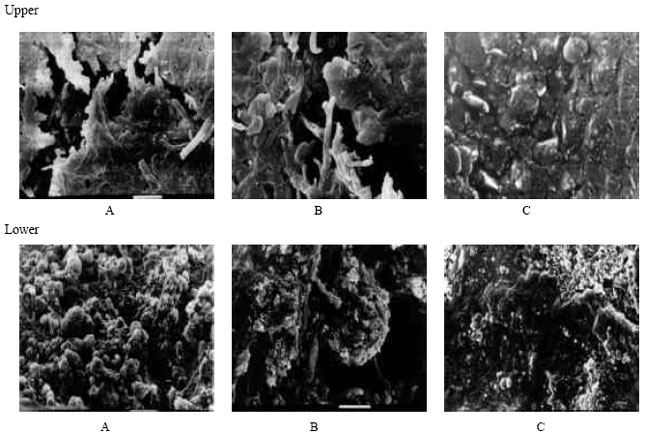 | |
| Fig. 4: | SEM photos during growth of the nocardioform actinomycete Nocardia sp. strain-MBR on NR-latex gloves (upper; A and B, test while C control) and NR granules (lower; A and B, test while C control) |
For this purpose, known amount of each of the following synthetic rubber materials; polybutadiene rubber, styrene rubber or synthetic polyisoprene rubber was dissolved in organic solvent and left in 250 mL Erlenmeyer flask till evaporation and 50 mL MSM were added and autoclaved. The flasks were inoculated with 2 mL of a 6 days old preculture of Nocardia sp. strain-MBR cells that were washed with sterile saline solution 0.9% (w/v). At the end of 6 weeks incubation period the % reduction in weight of the synthetic rubber material was determined. Results indicated that the synthetic cis-1-4-polyisoprene rubber was the most susceptible to degradation by the nocardioform actinomycete, while polybutadiene rubber was the most resistant and the loss in weight of the rubber substrates were, 2 and 11% for polybutadiene and synthetic polysioprene, respectively. It was recognized that The styrene rubber was moderately affected by the degradative capability of the bacterium (weight loss, 6%), the reason may be due to the presence of styrene ring could hinder rubber degradation process in the Nocardia sp strain-MBR.
Degradation of structurally analogous acyclic isoprenoid compounds: In order to study the potency of the rubber-degrading isolate, Nocardia sp. strain-MBR to degrade structurally analogous acyclic isoprenoid compounds and other related compounds, their utilization was investigated. The results indicated that the bacterial candidate was able to utilize branched hydrocarbon oligomers. It was recognized that the cells of Nocardia sp. strain-MBR showed very good growth on saturated-branched hydrocarbons (squalane and pristane) and moderate growth on unsaturated-branched acyclic isoprenoids phytol and squalene. On the other hand, no growth was recognized when other acyclic isoprenoid alcohols, aldehydes, keto or acidic forms was used as energy source. Utilization of similar compounds by rubber-degrading bacterium Mycobacterium fotuitum NF4 was also recorded by Berekaa and Steinbuchel (2000). These results collectively indicated that the bacterium has the metabolic capability to utilize some isoprenoid compounds as carbon and energy sources.
REFERENCES
- Arenskotter, M., D. Baumeister, M.M. Berekaa, G. Potter, R.M. Kroppenstedt, A. Linos and A. Steinbuchel, 2001. Taxonomic characterization of two rubber degrading bacteria belonging to the species Gordonia polyisoprenivorans and analysis of hyper variable regions of 16S rDNA sequences. FEMS Microbiol. Lett., 205: 277-282.
Direct Link - Arenskotter, M., D. Broeker and A. Steinbuchel, 2004. Biology of metabolically diverse genus Gordonia. Appliied Environ. Microbiol., 70: 3195-3204.
Direct Link - Berekaa, M.M. and A. Steinbuchel, 2000. Microbial degradation of the multiply branched alkane 2,6,10,15,19,23-Hexamethyltetracosane (Squalane) by Mycobacterium fortuitum and Mycobacterium ratisbonense. Applied Environ. Microbiol., 66: 4462-4467.
Direct Link - Berekaa, M.M., A. Linos, R. Reichelt, U. Keller and A. Steinbuchel, 2000. Effect of pretreatment of rubber material on its biodegradability by various rubber degrading bacteria. FEMS Microbiol. Lett., 184: 199-206.
Direct Link - Berekaa, M.M., A. Barakaat, S.M. El-Sayed and S.A. El-Aassar, 2005. Degradation of natural rubber by Achromobacter sp. NRB and evaluation of culture conditions. Polish J. Microbiol., 54: 55-62.
Direct Link - Bode, H., K. Kerkhoff and D. Jendrossek, 2001. Bacterial degradation of natural and synthetic rubber. Biomacromolecules, 2: 295-303.
Direct Link - Braaz, R., W. Armbruster and D. Jendrossek, 2005. Heme-dependent rubber oxygenase RoxA of Xanthomonas sp. cleaves the carbon backbone of poly (cis-1,4-isoprene) by a dioxygenase mechanism. Applied Environ. Microbiol., 71: 2473-2478.
Direct Link - Ehrlich, G., H.E. Taylor and H. Waelsch, 1948. The effect of surface-active substances on the fuchsin reaction of higher fatty aldehydes. J. Biol. Chem., 173: 547-551.
Direct Link - Heisey, R.M. and S. Papadatos, 1995. Isolation of microorganisms able to metabolize purified natural rubber. Applied Environ. Microbiol., 61: 3092-3097.
Direct Link - Jendrossek, D., G. Tomasi and R.M. Kroppenstedt, 1997. Bacterial degradation of natural rubber: A privilege of actinomycetes? FEMS Microbiol. Lett., 150: 179-188.
Direct Link - Kajikawa, S., A. Tsuchii and K. Takeda, 1991. Treatment of natural rubber waste by microorganisms. J. Agric. Chem. Soc. Japan, 65: 981-986.
Direct Link - Linos, A. and A. Steinbuchel, 1998. Microbial degradation of natural and synthetic rubbers by novel bacteria belonging to the genus Gordona. Kautschuk Gummi Kunststoffe, 51: 496-499.
Direct Link - Linos, A., A. Steinbuchel, C. Sproer and R.M. Kroppenstedt, 1999. Gordonia polyisoprenivorans sp. Nov., a rubber degrading actionmycete isolated from automobile tire. Int. J. Syst. Bacteriol., 49: 1785-1791.
Direct Link - Linos, A., M.M. Berekaa, R. Reichelt, U. Keller and J. Schmitt et al., 2000. Biodegradation of cis-1,4-polyisoprene rubbers by distinct actinomycetes: Microbial strategies and detailed surface analysis. Applied Environ. Microbiol., 66: 1639-1645.
Direct Link - Linos, A., M.M. Berekaa, A. Steinbuchel, K.K. Kim, C. Sproer and R.M. Kroppenstedt, 2002. Gordonia westfalica sp. nov. a novel rubber-degrading actinomycete. Int. J. Syst. Evol. Microbiol., 52: 1133-1139.
Direct Link - Rook, J.J., 1955. Micobiological deterioration of vulcanized ruubber. Applied Microbiol., 3: 302-309.
Direct Link - Rose, K. and A. Steinbuchel, 2005. Biodegradation of natural rubber and related compounds: Recent insights into a hardly understood catabolic capability of microorganisms. Applied Environ. Microbiol., 71: 2803-2812.
Direct Link - Seubert, W., 1960. Degradation of isoprenoid compounds by microorganisms. J. Bacteriol., 79: 426-434.
Direct Link - Subramananiam, A., 1995. The chemistry of natural ruber latex. Immunol. Allergy Clin., 15: 1-20.
Direct Link - Tsuchii, A., T. Suzuki and K. Takeda, 1985. Microbial degradation of natural rubber vulcanizates. Applied Environ. Microbiol., 50: 965-970.
Direct Link - Tsuchii, A. and K. Takeda, 1990. Rubber-degrading enzyme from bacterial culture. Applied Environ. Microbiol., 56: 269-274.
Direct Link - Tsuchii, A., K. Takeda, T. Suzuki and Y. Tokiwa, 1996. Colonization and degradation of rubber pieces by Nocardia sp. Biodegradation, 7: 41-48.
Direct Link - Yamada, Y., H. Moto, S. Kinoshita, N. Takeda and H. Okada, 1975. Oxidative degradation of squalene by Arthrobacter species. Applied Microbiol., 29: 400-404.
Direct Link - Yamada, Y., I. Naoki, N. Takuya and K. Atsuko, 1988. Oxidative pathway from squalene to geranylacetone in Arthrobacter sp. strain Y-11. Applied Environ. Microbiol., 54: 381-385.
Direct Link - Tsuchii, A., K. Takeda and Y. Tokiwa, 1997. Degradation of the rubber in truck tires by a strain of Nocardia. Biodegradation, 7: 405-413.
Direct Link








