ABSTRACT
Tyrosinase (EC 1.14.18.1) was extracted from potato (Somanum tuberosum) and four edible fungi such as Agaricus bisporus (Ab), Lentinus edodes (Le), Voluariella voluacea (Vv) and Pleurotus eryngii (Pe). The activity, kinetic parameters (Km, Vmax), optimum pH and temperature, activation energy and stability of the enzyme from different sources were determined. Comparatively, tyrosinase from Ab presented the highest activity and stability. The activity order was related to the intrinsic specific activity of the enzyme, the extraction efficiency and the assay conditions.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/biotech.2006.344.348
URL: https://scialert.net/abstract/?doi=biotech.2006.344.348
INTRODUCTION
Tyrosinase, which is also called polyphenol oxidase (EC 1.14.18.1), is a copper-containing enzyme widely distributed in animals, plants and microorganisms (Robb, 1984; Sánchez-Ferrer et al., 1995; Seo et al., 2003; Kim and Uyama, 2005). In the presence of oxygen, this enzyme catalyzes the hydroxylation of monophenols to o-diphenols and their subsequent dehydrogenation to o-quinones. The latter products are unstable in aqueous solution, further polymerizing to undesirable brown, red, or black pigments. Therefore, tyrosinase is the primary enzyme responsible for melanization in animals and browning in plants. Besides, tyrosinase has a wide variety of useful applications, such as in detection of phenols in aqueous and organic media (Zhang et al., 2001) and in removal of toxic chemicals including phenols and aromatic amines from industrial waste water and drinking water (Wada et al., 1995). The potential of using this enzyme in clinical trials has also been recognized (Baharav et al., 1996). Furthermore, tyrosinase is of great interest in organic synthesis because of its specific regioselectivity (Yang and Robb, 1991; Müller et al., 1998; Valero et al., 2003) and its use in organic media has been evaluated (Yang and Robb, 1993, 2005).
Tyrosinase from various fruits and vegetables has been studied, but comparison of the catalytic properties of the enzyme from different microorganisms has been rarely reported. In this study, the enzyme was extracted from potato (Somanum tuberosum) and four edible fungi such as Agaricus bisporus (Ab), Lentinus edodes (Le), Voluariella voluacea (Vv) and Pleurotus eryngii (Pe), and the catalytic properties of the enzyme, such as the kinetic parameters (Km, Vmax), optimal conditions of pH and temperature, activation energy, and enzyme stability, were determined and investigated. To our knowledge, this study was the first one to report the activity of tyrosinase from edible mushrooms Le, Vv and Pe.
MATERIALS AND METHODS
Materials: Potato (Somanum tuberosum) and four edible mushrooms (Ab, Le, Vv and Pe) were used as source materials of tyrosinase and purchased from a local supermarket in Shenzhen, China. L-dopa (3,4-dihydroxy-L-phenylalanine) was obtained from Sigma Chemical Co.
Enzyme preparation: The enzyme was obtained as the supernatant after centrifugation of an extract of a source material (50 g) in 100 mL phosphate buffer (50 mM, pH 6.0) (Yang and Robb, 2005).
Enzyme activity assay: The activity of the enzyme solution was determined by following the formation of dopachrome spectrophotometrically at 30°C (Yang and Robb, 1993). After addition of 50 μL enzyme extract to a cuvette containing 1.2 mL phosphate buffer (50 mM, pH 6.0) and 0.8 mL 10 mM L-dopa, the solution was immediately mixed and the increase in absorbance at 475 nm (indicating the formation of dopachrome) was recorded with the Pharmacia Biotech Ultraspec 2000 UV/Vis spectrophotometer. The initial rate was calculated from the linear part of the progress curve (i.e., over the first 2-3 min). One unit of tyrosinase was defined as the amount of the enzyme catalyzing the oxidation of 1 μmol L-dopa to dopachrome per minute at 30°C. The extinction coefficient of dopachrome at 475 nm was 3600 M-1cm-1 (Duckworth and Coleman, 1970).
Determination of Km, Vmax: The reaction was carried out in a cuvette where 50 μL of enzyme solution was added to 2 mL 50 mM phosphate buffer containing various concentrations of the substrate L-dopa (0-5 mM). The Km and Vmax values of tyrosinase from different sources were obtained from the Lineweaver-Burk plots.
Optimum pH and temperature: Optimum pH was determined by measuring tyrosinase activity at 30°C over a pH range of 5.0-8.0 in 50 mM phosphate buffer. Optimum temperature was obtained by measuring the activity of tyrosinase in 50 mM phosphate buffer (pH 6.0) at various reaction temperatures (20-80°C), which were controlled by the electronic temperature controller attached to the spectrophotometer.
Storage lifetime: Preparations of tyrosinase from different sources were stored at 4°C and assayed for activity at 30°C periodically as noted above.
RESULTS AND DISCUSSION
Kinetic parameters: Tyrosinase was extracted from five different sources into phosphate buffer and the crude enzyme extracts were used for all the following studies. The progress curves of the L-dopa oxidation reactions catalyzed by tyrosinase are shown in Fig. 1. Regardless of the sources from which the enzyme was extracted, the initial rate of the enzyme-catalyzed reactions responded proportionally to the enzyme concentration (data not shown), suggesting that the reactions were not interfered by the endogeneous phenolic substances present in the crude enzyme extracts (Soysal and Söylemez, 2004).
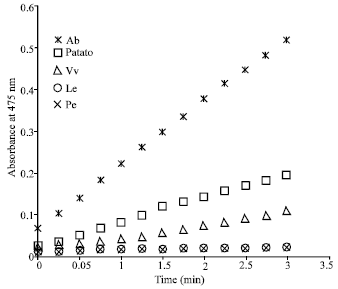 | |
| Fig. 1: | Progress curves of the L-dopa oxidation reactions catalyzed by tyrosinase obtained from different sources |
| Table 1: | Comparison of catalytic properties of tyrosinase preparations obtained from different sources |
 | |
The kinetic parameters of tyrosinase were determined using its extracts from the five sources and the results are listed in the third entry of Table 1. In all the five cases, the enzyme followed the Michaelis-Menten kinetics, with the lowest Km (0.60 mM) presented by the mushroom (Ab) tyrosinase and the highest Km (12.6 mM) by the potato tyrosinase. Previous study (Duckworth and Coleman, 1970) reported a Km value of 0.26 mM for mushroom tyrosinase at pH 7.0 when the same substrate, L-dopa, was used. Although the Km value reported in our study for the same enzyme from mushroom Ab is comparable, the difference between the two reported data may be related to the difference in the pH that was used for the enzyme assay and the source from which the enzyme was obtained. The relatively higher Km values for the enzymes from the other 4 sources suggest that under the assay conditions (pH 6.0) these enzymes possess lower affinity to the substrate, which, however, may be to some degrees dependent on the pH used for the assay (Duckworth and Coleman, 1970). As shown in the next section, all these 5 enzymes presented different optimum pH. On the other hand, the Vmax and Vmax/Km values reported in Table 1 confirmed that the enzyme extract obtained from Ab showed the highest activity whereas the one from Pe presented the lowest activity. However, it is unknown whether this activity order reflects either the order of the specific activity or the order of the total activity units of the enzyme extracted from different sources or both.
Study on the effect of substrate concentration has also revealed that all the enzymes, except the one from Le, catalyzed the L-dopa oxidation reactions with their initial rates increasing with the increase in the substrate concentrations over a tested range of 0-5.0 mM. A higher substrate concentration was not employed because of the limited solubility of the substrate. The Le enzyme, on the other hand, seemed to be subjected to substrate inhibition when the substrate concentration was higher than 3.4 mM (Fig. 2).
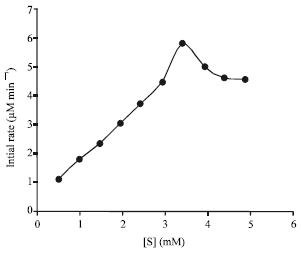 | |
| Fig. 2: | Variation of the activity of tyrosinase from Le upon the change in substrate concentration |
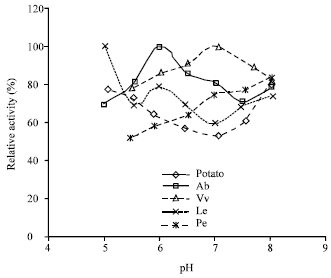 | |
| Fig. 3: | Dependence of tyrosinase activity on pH |
Optimum pH: The effect of pH on enzyme activity was investigated by measuring tyrosinase activity at 30°C over a pH range of 5.0-8.0 in 50 mM phosphate buffer and the results were plotted in Fig. 3. The enzymes from Ab and Vv had their pH optima at 6.0 and 7.0, respectively. The activity of the Pe enzyme increased with the increase in pH within the tested range, suggesting that this enzyme was more active under alkaline conditions with presumably an optimum pH higher than 8.0. Similarly, the enzyme from Le may have its optimum pH equals to or lower than 5.0. For potato tyrosinase, a V-shaped relation between enzyme activity and pH was observed within the tested pH range. The abnormal pH activity profiles might be due to the presence of multiple forms of mushroom tyrosinase (Jolley et al., 1969). Therefore, present results have substantiated the previous reports that the pH optimum of tyrosinase is highly dependent on the enzyme source and the nature of the substrate used.
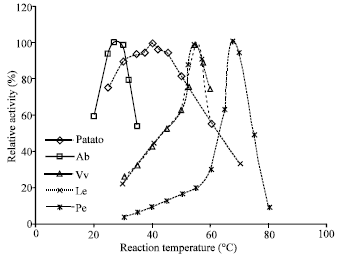 | |
| Fig. 4: | Effect of reaction temperature on the activity of tyrosinase from different sources |
For instance, the pH optimum was 4.8 for tomato tyrosinase using 3,4-dihydroxyphenylacetic acid as the substrate (Spagna et al., 2005), 6.5 for banana tyrosinase using dopamine as the substrate (Yang et al., 2000), and 8.0 for artichoke tyrosinase using pyrogallol as the substrate (Doğan et al., 2005). Two isoforms of tyrosinase from hybrid poplar showed different pH optima (6.0 and 7.0) when using catechol as the substrate (Wang and Constabel, 2003).
Optimum temperature and activation energy: The dependence of enzyme activity on the reaction temperature for the five enzyme preparations is shown in Fig. 4. The optimum temperature for tyrosinase was 27, 40, 55, 55 and 67°C for the enzyme extracted from Ab, potato, Le, Vv and Pe, respectively. Among the five enzyme preparations, potato tyrosinase retained most of its activity (75% of the maximum) over a broad temperature range (25-50°C) and the Pe enzyme was highly active at high reaction temperature, obtaining a 25 fold increase in activity when the reaction temperature was raised from 30°C to its maximum temperature of 67°C. The same enzyme from other sources has been reported to have different optimum temperatures in the literature. For example, tyrosinase from banana and tomato had an optimum temperature of 30°C (Yang et al., 2000) and 40°C (Spagna et al., 2005), respectively. Our experiments have provided some new sources, such as Le, Vv and Pe, from which a more thermophilic enzyme can be prepared.
According to the Arrhenius equation, activation energies of the reactions catalyzed by the five different tyrosinase preparations can be calculated from the slope when plotting the natural logarithm of the initial reaction rates against 1/T, where T is the absolute reaction temperature.
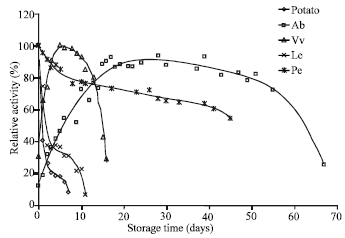 | |
| Fig. 5: | Stability of tyrosinase from different sources at 4°C |
Reactions were carried out under the same conditions as for determination of the optimal reaction temperature, and the temperature ranges used were 25-40°C for potato, 20-27°C for Ab, 30-55°C for Vv and Le and 30-60°C for Pe. The results are listed in the last entry of Table 1. The activation energy obtained in this study for tyrosinase from Ab (56.4 kJ mol-1) was comparable with the result reported in (Yang and Robb, 1993) for the same enzyme source when using 4-methylcatechol as the substrate (57.3 kJ mol-1). The data for all the four edible mushrooms, which were broadly similar in the range of 42.5-56.4 kJ mol-1, were obviously higher than the one for potato (13.6 kJ mol-1), suggesting that the enzyme from potato should have a relatively higher kcat. However, the variation of the activation energies obtained in this study did not seem to be related to the activities presented by the enzyme prepared from different sources.
Stability: To investigate the stability of tyrosinase during storage, five enzyme preparations were kept at 4°C and their activities were measured at 30°C periodically. As shown in Fig. 5, the activities of the enzyme samples obtained from potato, Le and Pe decreased with the increase in the storage time: The activities of the former two enzyme preparations dropped steeply to 8.8 and 30.8% of their original activities, respectively, after 7 days of storage at 4°C; while the latter sample obtained from Pe showed a much longer storage lifetime, remaining 54.5% of its original activity after being kept at 4°C for 45 days. Interestingly, the activity of the tyrosinase extract from Ab increased with storage time during the initial 15 days and then decreased gradually for about 35 days before declining abruptly. Similar phenomenon occurred to the extract from Vv. Comparatively, Ab was the source that was able to generate the enzyme preparation which remains highly active during the longest storage time. After 67 days of storage at 4oC, the enzyme prepared from this source still presented 25.2% of its maximal activity and twice as its initial activity when the enzyme was freshly prepared. The high lifetime stability and especially the activation effect of this enzyme during storage, is worth further investigation.
Activity comparison: The activities of the enzyme extracts freshly prepared from the five different sources can be compared in the second entry of Table 1. Among the five sources, Ab was the source from which the highest enzyme activity was obtained, followed by potato and the other 3 edible mushrooms (Le, Vv, Pe). This was confirmed by the progress curves of the enzymatic reactions shown in Fig. 1 and the Vmax data listed in the third entry of Table 1.
However, the activity order shown in the second entry of Table 1 did not seem to be in agreement with the other kinetic data, such as optimum pH and temperature and activation energy, listed in Table 1. In fact, the rather high optimum pH (≥8.0) and high optimum temperature (67°C) of tyrosinase from Pe may be used to partially explain the extremely low activity presented by such enzyme in the activity assay: The optimal reaction conditions for this enzyme (pH≥8.0, 67°C) were far apart from the conditions used in the activity assay (pH 6.0, 30°C). As a result, it could be expected that if assayed under their optimal conditions, the enzymes from Le and Pe, both of which presented minimal activities in Table 1, would achieve their maximal activities. Therefore, the activity order shown in Table 1 may be related to factors such as the intrinsic specific activity of the enzyme from different sources, the efficiency of enzyme extraction, and the assay conditions used to determine the enzyme activity.
CONCLUSIONS
In this study, the catalytic properties of tyrosinase obtained from potato and four edible mushrooms (Ab, Le, Vv and Pe) have been determined and investigated. Comparatively, the crude enzyme preparation extracted from Ab presented the highest activity and the highest storage lifetime. To our knowledge, this study gave the first report about the catalytic properties of tyrosinase from edible mushrooms Le, Vv and Pe. The results reported in this study would be beneficial to the use of tyrosinase in its various applications, especially when one considers the selection of the source from which an enzyme with high activity and stability can be prepared.
REFERENCES
- Baharav, E., O. Merimski, Y. Shoenfeld, R. Zigelman and B. Gilbrud et al., 1996. Tyrosinase as an aotoantigen in patients with vitiligo. Clin. Exp. Immunol., 105: 84-88.
Direct Link - Duckworth, H.W. and J.E. Coleman, 1970. Physicochemical and kinetic properties of mushroom tyrosinase. J. Biol. Chem., 245: 1613-1625.
Direct Link - Doan, S., Y. Turan, H. Erturk and O. Arslan, 2005. Characterization and purification of polyphenol oxidase from artichoke (Cynara scolymus L.). J. Agric. Food Chem., 53: 776-785.
Direct Link - Jolley, R.L., R.M. Nelson and D.A. Robb, 1969. The multiple forms of mushroom tyrosinase. J. Biol. Chem., 244: 3251-3257.
Direct Link - Kim, Y.J. and H. Uyama, 2005. Tyrosinase inhibitors from natural and synthetic sources: Structure, inhibition mechanism and perspective for the future. Cell. Mol. Life Sci., 62: 1707-1723.
Direct Link - Muller, G.H., A. Lang, D.R. Seithel and H. Waldmann, 1998. An enzyme-initiated hydroxylation-oxidation carbo Diels-Alder domino reaction. Chem. Eur. J., 4: 2513-2522.
Direct Link - Sanchez-Ferrer, A., J.N. Rodriguez-Lopez, F. Garcia-Canovas and F. Garcia-Carmona, 1995. Tyrosinase: A comprehensive review of its mechanism. Biochim. Biophys. Acta, 1247: 1-11.
PubMed - Seo, S.Y., V.K. Sharma and N. Sharma, 2003. Mushroom tyrosinase: Recent prospects. J. Agric. Food Chem., 51: 2837-2853.
Direct Link - Soysal, C. and Z. Soylemez, 2004. Properties of wheat bran polyphenol oxidase. Nahrung, 48: 5-8.
Direct Link - Spagna, G., R.N. Barbagallo, M. Chisari and F. Branca, 2005. Characterization of a tomato polyphenol oxidase and its role in browning and lycopene content. J. Agric. Food Chem., 53: 2032-2038.
Direct Link - Valero, E., M.I. Lozano, R. Varon and F. Garcia-Carmona, 2003. Enzymatic synthesis of 3'-hydroxyacetaminophen catalyzed by tyrosinase. Biotechnol. Prog., 19: 1632-1638.
Direct Link - Wada, S., H. Ichikawa and K. Tatsumi, 1995. Removal of phenols and aromatic amines from wastewater by a combination treatment with tyrosinase and a coagulant. Biotechnol. Bioeng., 45: 304-309.
Direct Link - Wang, J. and C.P. Constabel, 2003. Biochemical characterization of two differentially expressed polyphenol oxidases from hybrid poplar. Phytochemistry, 64: 115-121.
Direct Link - Yang, Z. and D.A. Robb, 1993. Comparison of tyrosinase activity and stability in aqueous and nearly nonaqueous environments. Enzyme Microb. Technol., 15: 1030-1036.
Direct Link - Yang, C., S. Fujita, M.D. Ashrafuzzaman, N. Nakamura and N. Hayashi, 2000. Purification and characterization of polyphenol oxidase from Banana (Musa sapientum L.) pulp. J. Agric. Food Chem., 48: 2732-2735.
Direct Link - Yang, Z. and D.A. Robb, 2005. Tyrosinase activity in reversed micelles. Biocatal. Biotransform., 23: 423-430.
Direct Link - Zhang, S., H. Zhao and R. John, 2001. A dual-phase biosensing system for the determination of phenols in both aqueous and organic media. Anal. Chim. Acta, 441: 95-105.
Direct Link








