ABSTRACT
High production of cell biomass and somatic embryos could be established in the suspension cultures of Cyperus aromaticus. The callus derived from root pieces was used for preparation of the cell suspension cultures. The cells pretreated with 2,4-Dichlorophenoxyacetic acid (2,4-D), α-Naphthaleneacetic Acid (NAA) and picloram in the suspension cultures produced somatic embryos after one week of transferring onto Murashige and Skoog (MS) solid medium without Plant Growth Regulators (PGR). With a small amount of cell inoculum (0.12 g) cultured in liquid MS medium supplemented with 2.0 mg L-1 NAA for three weeks followed by one week culture on solid MS basic medium produced an average of 10.5 g fresh embryogenic cell biomass of C. aromaticus. The embryogenic cells differentiated to produce primodia leaves after transferred to MS medium supplemented with 1.0 mg L-1 NAA.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/biotech.2006.75.78
URL: https://scialert.net/abstract/?doi=biotech.2006.75.78
INTRODUCTION
Cyperus aromaticus (Ridley) Mattf and Kukenth, a parennial herb belonging to the Cyperaceae family, is grown widely in temperate and tropical region including Malaysia. It is a monocotyledonous grass-like herb with solid triquetrous culms (Nather Khan, 1990). It contains Juvenile Hormone III (JH III) with the phytochemical structure related to the JH III found in the insects (Toong et al., 1988). According to Schwartz et al. (1998), the simple processing of the leaves of this plant may offer an effective, inexpensive and environmentally safe method of mosquito control.
Plant cell culture has been pursued for more than twenty years as a tool for the elucidation of secondary metabolites biosynthesis and as a potential means for their biotechnological production to rival extraction process based on whole plant material (Hamill and Rhodes, 1993). Somatic embryogenesis is one of the in vitro culture techniques by which somatic cells differentiate into somatic embryos through an orderly series of characteristic morphological stages (Vasic et al., 2001; Von Arnold et al., 2002). According to Martin (2003), suspension cultures induced the development of higher number of embryogenic cells. In this study, we investigate the effect of growth regulators supplemented into the culture medium on the biomass production of the embryogenic suspension cultures of Cyperus aromaticus to be used as the material source for the production of JH III in the future.
MATERIALS AND METHODS
Cell suspension culture of C. aromaticus was initiated by transferring one gram of the friable callus derived from root pieces into 100 mL Erlenmeyer flask containing 30 mL MS (Murashige and Skoog, 1962) liquid medium supplemented with 1.5 mg L-1 2, 4-Dichlorophenoxyacetic acid (2, 4-D) and 2.5 mg L-1 α-Naphthaleneacetic acid (NAA), the best medium determined for callus induction. The pH of the medium was adjusted to 5.7-5.8 before autoclaving at 121°C for 13 min under a pressure of 1.05 kg cm-2. All the cultures were placed on a rotary shaker at 100 rpm and incubated at 25±2°C under continuous light intensity of 32.5 μE m-2 s-1. The cell suspension cultures were subcultured at 4-weeks interval. The established cell suspension cultures of C. aromaticus were used as the material source for subsequent studies.
Fresh weight 0.12 g of the C. aromaticus cells harvested from 3 weeks old cell suspension cultures were inoculated into 100 mL Erlenmeyer flask containing 30 mL MS liquid medium supplemented with one of the following Plant Growth Regulators (PGR): 2, 4-D, NAA or picloram at 0, 2.0, 4.0, 6.0, 8.0 and 10.0 mg L-1. The cultures were maintained on a gyratory shaker at 100 rpm. The flasks were placed under continuous light provided by cool white florescent lights with an intensity of 32.5 μE m-2 s-1. The fresh weight of the cell biomass was determined from each flask. Two culture flasks were taken randomly every week over a 4 weeks period to determine the cell biomass and the cell growth pattern. Each of the experiments was repeated three times.
The three weeks old cells cultured in MS liquid medium supplemented with 2 mg L-1 NAA, MS + 2 mg L-1 picloram and MS + 6 mg L-1 2, 4-D were transferred to PGR free MS solid medium for one week for the induction of embryogenic cells. These three media, MS + 2 mg L-1 NAA, MS + 2 mg L-1 picloram and MS + 6 mg L-1 2, 4-D, each was chosen as the best proliferation medium for C. aromaticus cells from each type of growth regulator. The one-week old embryogenic cells formed on the basic MS solid medium were then transferred to MS solid medium supplemented with 1 mg L-1 NAA for embryos development. The biomass of the embryogenic cells were determined after one week cultured in MS basic medium. Some of the cells were removed from the embryogenic induction cultures (basic MS medium) and from the embryos development cultures (MS + 1 mg L-1 NAA medium) for histological studies to determine the formation and development of the somatic embryos.
Cells for histological observation were fixed in FAA (formaldehyde: acetic acid: ethanol, 5: 5: 90) for 24 h. After dehydration process through a gradual ethanol series, these samples were embedded in xylene wax and cut into 8 μm thick slices. The samples were stained with safranin and fast green. Cultures were examined and photographed with a stereo zoom microscope (SZH, Olympus).
RESULTS AND DISCUSSION
Cell suspension cultures of Cyperus aromaticus were prepared by culturing the friable type of callus in MS liquid medium supplemented with three different plant growth regulators namely 2, 4-D, NAA and picloram. The cells could be maintained and showed good growth in MS liquid medium supplemented with 2.0 - 10.0 mg L-1 of 2, 4-D, NAA or picloram. Basic MS medium without the addition of any growth regulators supported very low cell growth within the 4 weeks culture duration. Different type and different concentration of PGRs in the culture medium gave different effect on the production of embryogenic cell biomass of C. aromaticus. Maximum biomass was obtained when the cells were cultured in MS liquid medium supplemented with 2.0 mg L-1 NAA (Fig. 1). When 2,4-D was supplemented into the liquid MS medium, MS + 6.0 mg L-1 2, 4-D induced the maximum cell biomass (Fig. 2). While MS liquid medium with the addition of 2.0 mg L-1 picloram produced the best cell biomass among other tested concentrations of picloram (Fig. 3).
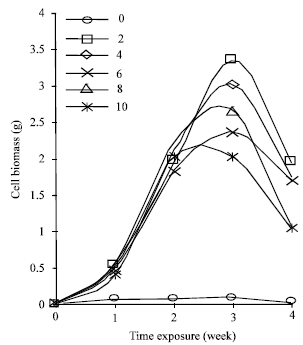 | |
| Fig. 1: | Growth dynamic of Cyperus aromaticus suspension cultures in MS + (0-10 mg L-1) NAA (n = 6) |
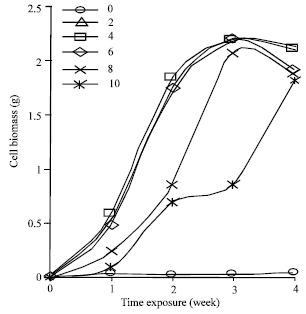 | |
| Fig. 2: | Growth dynamic of Cyperus aromaticus suspension cultures in MS + (0-10 mg L-1) 2, 4-D (n = 6) |
The cell biomass decreased on the 4th week for all the cells cultures growing in MS supplemented with 2, 4-D or NAA. The cells continued to grow in MS liquid medium supplemented with picloram even after 4 weeks of culture. Cells cultured in MS basic medium turned brown after one week of cultures and did not support any cell growth subsequently (Fig. 1-3).
| Table 1: | Formation and proliferation rate of embryonegic cells from three weeks old cells harvested from three different culture medium after transferred for one week on MS solid medium without any PGRs |
 | |
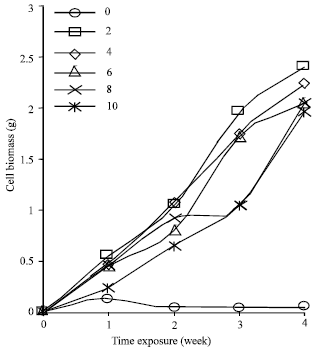 | |
| Fig. 3: | Growth dynamic of Cyperus aromaticus suspension cultures in MS + (0-10 mg L-1) picloram (n = 6) |
This indicated that the cell suspension culture of C. aromaticus could not be maintained in the MS medium without PGRs. It was reported by Karam et al. (2003) that depletion of nutrient, accumulation of toxic products and absent of PGRs were other limiting factors that might have led to cell deaths and eventually to a decline in cell growth.
The cells harvested from the three weeks old cultures in MS + 2.0 mg L-1 NAA, MS + 6.0 mg L-1 2, 4-D and MS + 2.0 mg L-1 picloram, formed embryogenic cells after the cells were transferred onto basic MS solid medium for one week with a proliferation rate of 2.3, 2.7 and 3.0, respectively (Table 1). This showed that pretreatment of cells with different type of PGRs could affect the proliferation and formation of the somatic embryos. Vasil (1984) reported that the cultures must be plated on agar medium to obtain further development of mature somatic embryos. Tawfik and Noga (2002) and Vasic et al. (2001) reported that the embryogenic calli of many plant species were developed when the calli were transferred to PGR free medium. While Ruffoni and Massabü (1996) found that the embryonic cells of Lisianthus russellianus were differentiated into globular, heart and torpedo-shaped structures after 30 days in a medium without PGRs.
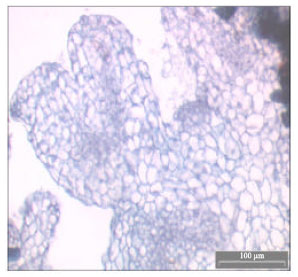 | |
| Fig. 4: | Historiculture study showing the embryos of C. aromaticus differentiated into young primodia leaves after transferred to solid MS + 1.0 mg L-1 NAA |
Present study showed that by culturing the C. aromaticus cells in a liquid medium as cell suspension cultures for three weeks then transferred to a solid MS medium without any growth regulators could be a very effective method for producing high quantity of cell biomass and embryogenic cells of C. aromaticus. With a small amount of cell inoculum (0.12 g) cultured in liquid MS medium supplemented with 2.0 mg L-1 NAA for three weeks followed by one week culture on solid MS basic medium produced an average of 10.5 g fresh cell biomass of C. aromaticus. The pretreatment of cells with NAA was more effective compared to 2, 4-D and picloram.
Histological examination of C. aromaticus cells revealed that somatic embryos formed after they were transferred to PGR free solid MS medium. The embryogenesis process could be detected within the first week after cultured on PGR free MS medium. This was characterized by cells with prominent nucleus, dense cytoplasm and thicker cell wall than the normal callus cells. It was relatively isodiametric and small with respect to neighboring cells. The embryogenic cells that were transferred to 1.0 mg L-1 NAA turned from light yellow to green in colour with the formation of primodia leaves (Fig. 4). The same characteristics were reported by Quiroz-Figueroa et al. (2002) in their histological studies on the developmental stages and differentiation of two different somatic embryogenesis systems of Coffea arabica.
ACKNOWLEDGMENTS
We thank the Malaysian Government for the IRPA Research Grant and Universiti Sains Malaysia, Penang, Malaysia for research facilities.
REFERENCES
- Hamill, J.D. and M.J.C. Rhodes, 1993. Manipulating Secondary Metabolism in Culture. In: Plant Biotechnology Volume III: Biosynthesis and Manipulation of Plant Products Blackie Academic and Professional. Grierson, D. (Ed.). Chapman and Hall, London, Glasgow, New York, Tokyo, Melbourne, Madras, pp: 178-209.
- Karam, N.S., F.M. Jawad, N.A. Arikat and R.A. Shibl, 2003. Growth and rosmarinic acid accumulation in callus, cell suspension and root cultures of wild Salvia fruticosa. Plant Cell, Tissue Org. Cult., 73: 117-121.
Direct Link - Martin, K.P., 2003. Plant regeneration through somatic embryogenesis of Holostema ada-kodien, a rare medicinal plant. Plant Cell Tissue Org. Cult., 72: 79-82.
CrossRefDirect Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Quiroz-Figueroa, F.R, C.F.J. Fuentes-Cerda, R. Rojas-Herrera and V.M. Loyolo-Vargas, 2002. Histological studies on the developmental stages and differentiation of two different somatic embryogenesis systems of Coffea Arabica. Plant Cell Rep., 20: 1141-1149.
CrossRefDirect Link - Schwartz, A.M., S.M. Paskewitz, A.P. Orth, M.J. Tesch, Y.C. Toong and W.G. Goodman, 1998. The lethal effects of Cyperus iria on Aedes aegypt. J. Am. Mosquito Control Assoc., 14: 78-82.
Direct Link - Tawfik, A.A. and G. Noga, 2002. Cumin regeneration from seedling derived embryogenic callus in response to amended Kinetin. Plant Cell Tissue Org. Cult., 69: 35-40.
CrossRefDirect Link - Toong, Y.C., D.A. Schooley and F.C. Baker, 1988. Isolation of insect juvenile hormone III from a plant. Nature, 333: 170-171.
CrossRefDirect Link - Vasic, D., G. Alibert and D. Skoric, 2001. Protocol for efficient repetitive and secondary somatic embryogenesis in Helianthus maximiliani (Schrader). Plant Cell Rep., 20: 121-125.
Direct Link - Von Arnold, S., I. Sabala, P. Bozhkov, J. Dyachok and L. Filonova, 2002. Developmental pathways of somatic embryogenesis. Plant Cell. Tissue Organ Cult., 69: 233-249.
CrossRefDirect Link








