Research Article
Induction of Somatic Embryogenic Callus From the Leaves of Pereskia grandifolia
Sains University Malaysia, Malaysia
Chan Lai Keng
Sains University Malaysia, Malaysia
Pereskia grandifolia is a type of cactus plant belonging to the Cactaceae family and Pereskioideae subfamily. It has a woody stem with large fascicles of spines and leaves that are different from the other cacti species. P. grandifolia also known as ‘Rose of Burma’ and has been used by the Malaysian Chinese as traditional remedy for the prevention and treatment of uterus cancer and fibroids. The leaves extract was found to inhibit the cell growth of NCI-II23 (human non-small lung adenocarcinoma) and Caov-3 (human ovarian carcinoma) cell lines (Ooi et al., 2003).
The conventional propagation method of Pereskia grandifolia is beset with problems of scanty and delayed rooting of seedlings and vegetative cuttings. This approach can hardly fulfill the increasing demand of the local market. In vitro culture technique could be the alternative method for the production of plant material as future commercial supplies of P. grandifolia for the preparation of herbal products. The production of somatic embryogenic calluses could be used as the material source and was fundamentally more efficient than root and shoots induction. The process is usually less elaborate and less diversity of media are required (Endress, 1994). Until today, the induction of somatic embryogenic callus has not been reported for any of the Pereskia sp. This study hence reports the induction of embryogenic calluses from the different parts of the P. grandifolia leaves.
Establishment of aseptic plant materials: P. grandifolia plants were collected from the herbal garden of School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia. The young leaves were used as explants for the induction of embryogenic callus. The surface of the leaves was wiped with 35% ethanol followed by washing with 50% Clorox® (v/v) solution with the addition six drops of Teepol. They were then rinsed with running tap water for 30 min. The explants were then surface-sterilized with 150 mg L-1 Sodium dichloroisocyanurate (NaDCC) for 15 min. They were then rinsed three times with sterile distilled water. The mid-vein section of the leaf (1x1 cm) and the leaf blade (1x1 cm) were cut from the aseptic explants before inoculated onto solid Murashige and Skoog (1962) (MS) medium without any plant growth regulator. The pH of the medium was adjusted to 5.7-5.8 followed by the addition of 7.5 g L-1 agar (Chile Algas) before the medium was autoclaved using Elite Sterilizer EAC-4000C at 1.05 kg cm-2 and 121°C for 13 min. The cultures were placed in the culture room maintained at 25±2°C under continuous cool white fluorescent lights with a photon flux density of 150-200 lux. The aseptic leave explants were then used for the induction of embryogenic callus.
Induction of embryogenic callus: The aseptic mid-vein section of leaf (1x1 cm) and the leaf blade (1x1 cm) were cultured on Gamborg B5 medium (Gamborg et al., 1968) supplemented with 0, 2, 4, 6, 8 and 10 mg L-1 2,4-dichlorophenoxyacetic acid (2,4-D). Six experimental units were used for each medium and each type of leaf explant. The experiment was carried out using the complete randomized block design. Each experimental unit consisted of three explants. Biomass of callus induced from each type of explant in each medium was determined after 8 weeks of culture. The data were analyzed using two-way analysis of variance (ANOVA) and the best callus induction medium was selected after comparison of mean using Tukey test at p = 0.05.
Effect of different part of leaf Mid-vein section on induction of embryogenic callus: The leaf mid-vein was cut into two sections: From middle of the leaf to the basal end near the petiole (MP) and from the middle part of the leaf towards the leaf apex (MA). These explants were cultured on B5 medium supplemented with 6 mg L-1 2,4-D, the optimum callus induction medium determined in the previous section. Twenty explants were used for each type of explant. The biomass of callus induced from the two different sections of leaf mid-vein was recorded after 8 weeks of culture. The data was analyzed using student t-test at p = 0.05.
Optimization of culture medium for the induction of embryogenic callus: The MP leaf explants were cultured on B5 medium supplemented with 4.0, 4.5, 5.0, 5.5, 6.0, 6.5, 7.0, 7.5 and 8.0 mg L-1 2,4-D. Six experimental units with three explants per unit were used for each medium using complete randomized block design. Biomass of callus that was induced from each of the different medium was recorded after 8 weeks of culture. The data was analyzed using two-way Analysis of Variance (ANOVA) and the best medium was selected after comparison of mean values by Tukey test at p = 0.05.
Histological study of embryogenic callus: To study the developmental stages of the somatic embryogenic callus, the callus tissues were fixed in formaldehyde, glacial acetic acid and absolute alcohol in the ratio of 1:1:18 for 48 h. The fixed tissues were dehydrated in a series of tertiary butyl alcohol (TBA) (50, 70, 85, 98, 100% (v/v), absolute TBA) followed by submerging in xylene for 1-2 h. The samples were embedded in Paraplast Tissue Embedding wax overnight. The wax blocks were sliced into 10 μm thickness sections which were then double stained with Safranin and Fast Green. The prepared slides were observed using the Olympus SZ-PT Phaco light microscope.
Healthy and aseptic leaf explants were used for the induction of embryogenic callus. The amount of calluses initiated from solid B5 medium supplemented with different concentration of 2,4-D (0, 2, 4, 6, 8 and 10 mg L-1) was significantly different when using the mid-vein section leaf explants but not for the leaf blade explants. More calluses were induced from the mid-vein section of the leaf cultured on B5 medium supplemented with 6 mg L-1 2,4-D (0.478 g) and B5 medium supplemented with 8 mg L-1 2,4-D (0.461 g). B5 medium supplemented with less than 6 mg L-1 or more than 8 mg L-1 2,4-D induced less than 0.25 g of embryogenic callus from the mid vein section of P. grandifolia leaves after 8 weeks of culture. Only 0.1-0.22 g of callus was induced from the leaf blade explants that do not contain any leaf veins when they were cultured on B5 medium supplemented with 2-10 mg L-1 of 2,4-D. Callus was not induced either from the mid-vein section of the leaves or the leaf blade of P. grandifolia when they were cultured on B5 medium without 2,4-D (Table 1).
| Table 1: | Production of embryogenic callus from the leaf explants of P. grandifolia on solid B5 medium supplemented with different concentration of 2,4-D after 8 weeks of culture |
 | |
| Mean values within columns followed by same alphabet were not significantly different based on Tukey test at p = 0.05 | |
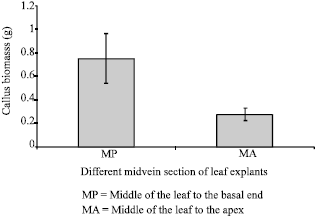 | |
| Fig. 1: | Effect of different part of leaf mid-vein section on the induction of Embryogenic callus of P. grandifolia |
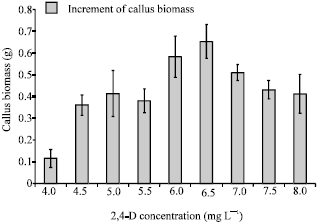 | |
| Fig. 2: | Effect of 2,4-D supplemented into B5 medium on Induction of Embryogenic callus from the mid-vein of Pereskia grandifolia leaves |
The amount of calluses produced from different section of the leaf mid-vein was found to be significantly different. The MP section leaf explants produced the most callus (0.752±0.212 g) compared to the MA section (0.278±0.053 g) on B5 medium supplemented with 6 mg L-1 2,4-D (Fig. 1). Chen et al. (1999) had used different part of in vitro leaf explants of Oncidium Gower Ramsey to induce direct somatic embryos. Their research showed that the formation of the foliar embryos was affected by the maturity state of the leaves and more foliar embryos were formed on the older leaves. In induction of somatic embryogenic callus of P. grandifolia, various part of leaf did play a role in the formation of embryogenic callus beside the effect of plant growth regulator, 2,4-D. This was because the mid-vein section of leaf explants consisted of more vascular tissue as compared to the tissue at the leaf blade.
 | |
| Fig. 3: | A-C :Embryogenic callus formed from the leaf explants of P. grandifolia when cultured on B5 medium supplemented with 2,4-D. (A) Nodular callus from leaf mid-vein; (B) Fine roots produced from embryogenic callus after 8 weeks of culture; (C) Histological section of embryogenic callus induced from leaf explants (L) showing active cell division (C1) and meristematic zone (MZ) (C2) |
In the optimization of medium study using the MP leave as explants, B5 medium supplemented with 6.5 mg L-1 2,4-D was found to induce a maximum biomass (0.652±0.078 g) of embryogenic callus within 8 weeks of culture and this was found to be not significantly different to the amount of embryogenic callus induced on B5 medium supplemented with 6 or 7 mg L-1 2,4-D (Fig. 2). Bhojwani and Razdan (1996) reported that 2,4-D (0.5-1 mg L-1) was generally used for the induction of embryogenic callus. However, Yemets et al. (2003) reported that 8 mg L-1 2,4-D was needed to be added into the MS basic medium together with 1 μM copper sulphate for the high-frequency production of somatic embryos of cassava for cryopreservation purpose. Chengalrayan and Gallo-Meagher (2001) also found that 13.6 μM 2,4-D was supplemented into MS basic medium to induce embryogenic calluses from the sugarcane young leaves (Saccharum sp. Hybrib cv. CP 84-1198). The amount of 2,4-D that was needed for induction of embryogenic calluses was totally depended on the sensitivity of the plant cell. As suggested by Arnold et al. (2002), the competence of embryogenic callus induction of each plant species was due to the sensitivity of the explant cells towards the various type of auxin. Kim et al. (2004) observed that embryogenic callus could be obtained from the hypocotyl segments of Medicago sativa L. using MS medium supplemented with 0.02 mg IAA and 1.0 mg L-1 zeatin after 45 days of culture. While Kim et al. (2003) found that embryogenic callus could be propagated from the leaf explants of Rosa hybrida L. on MS medium with the presence of NAA, Zeatin and GA3. However, our experimental result indicated that for P. grandifolia, 2 mg L-1 2,4-D incorporated into the MS medium was sufficient to induce embryogenic callus from the leaf explants but 6.5 mg L-1 of 2,4-D was needed to induce higher biomass of embryogenic calluses.
Histological study showed that no callus or nodular embryos were formed after one week of culture from the leaf blade explants that were cultured on B5 medium supplemented with 6.5 mg L-1 2,4-D but the tissues were swollen at the cutting edges. However, the embryogenic calluses were formed from the leaf mid-vein explants. The embryogenic calluses were light green in color and nodular in shape (Fig. 3A). Small fine roots were formed from the embryogenic callus surface after 5 weeks of culture in all B5 medium supplemented with 2,4-D (Fig. 3B). These nodular calluses were composed of non-vacuolated cells with central nuclei undergoing a rapid mitotic division (Fig. 3C1) and forming meristematic cells zone (Fig. 3C2). These results supported our findings that mid-vein sections (MP) of P. grandifolia leaves were easier to form embryogenic calluses when they were cultured on B5 medium supplemented with 6.5 mg L-1 2,4-D compared to the leaf blade explants.
The authors wish to express their gratitude to School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia for research facilities.