Research Article
Pollen Grains of Lagos Lagoon Swamp and Hinterland Vegetation-I
Department of Botany and Microbiology, University of Lagos, Lagos, Nigeria
O. T. Ogundipe
Department of Botany and Microbiology, University of Lagos, Lagos, Nigeria
Over the years, pollen grains and spores have been preserved in various geological deposits as a result of sedimentation. These pollen grains and spores can be retrieved, identified and interpreted through various techniques in the field and laboratory. By utilizing the variation of palynomorphs distribution in the air, soil sediments and various other sources, palynological studies have been a vital tool to scientists in fields such as biostratigraphy, climatology, medicine-alleviation of pollinosis (hay fever-allergenic disease) forensic studies, mellisopalynology, plant evolution, taxonomy and environmental restoration activities (Bryant et al., 1990; Perveen et al., 2004; Mbagwu and Edeoga, 2006; Adekanmbi and Sowunmi, 2006, 2007; Palazzesi et al., 2007; Ige, 2009).
Due to high species diversity however, there is the limitation of identifying a number of pollen grains recovered from sediments, air and other product and substances e.g., honey necessitating the need for a reference pollen atlas. Lieux (1983) compiled an atlas of pollen of trees, shrubs and woody vines of Louisiana and other Southeastern States, Willard et al. (2004) put together an atlas of pollen and spores of the Florida everglades. However, apart from the work of Sowunmi (1973, 1995) on the compilation of pollen grains of Nigerian woody plants; the works of Takahashi and Ulrich (1989), Chene et al. (1978), Salard-Chebodaeff and Dejax (1991) and Biffi and Grignani (1983) which are published, other existing atlases, few although are in-house work of oil companies and are considered strictly confidential and therefore are not available for public use.
The polliniferous material (some flowers, buds, or even single stamens) was treated with hot water in order to make it soft. Anthers were picked out under a dissecting microscope and placed in a centrifuge tube. Sample preparation was carried out at the Palynological Unit of Botany Department, University of Lagos; some at the Jodrel Laboratory, Micromorphology Section, Royal Botanic Garden, Kew and the Oxford University Centre for Environment (OUCE), of the University of Oxford, UK following standard sample preparation method as outlined by Erdtman (1969) and Faegri and Iversen (1989). Slides were made.
Photomicrography was carried out under Olympus light microscope with Motic MC2000 (2.0 Megapixel) camera. Specimens are illustrated at either 1000x or 400x. Scanning Electron Micrographs were taken using Hitachi S-4700 Scanning Electron Microscope (SEM) from prepared stubs coated with gold in the Emitech Sputter Coater K550. Pollen grains were described based on their morphological characteristics. Amb or overall grain shape is first defined, followed by shape classes as defined by Erdtman (1969). The shape classes are based on measurements of the polar axis (P) and equatorial diameter (E) and the resulting P/E ratio (see glossary). Mean dimensions are provided, followed by minima and maxima in parentheses. The diameters (polar and equatorial axis) of 10 non-folded pollen grains were measured in equatorial view at X400 magnification. Aperture and exine thickness were also measured. The measured values were directly rounded to the nearest micrometer unit (Moore and Webb, 1983; Moore et al., 1991).
Pollen morphological characters of the species studied are presented by family. The list of species included in the study and collection information is shown in Table 1. The descriptive photomicrographs of studied pollen is compiled in Fig. 1-6.
Characteristic of pollen grains:
Acanthaceae
Asystasia gagentica: (Fig. 1)
| • | Shape: Triangular amb; radially symmetrical; Prolate grain; P/E (1.63) |
| • | Size: Polar axis 19.5 (15.7-22.7) μm; Equatorial axis 12.0 (9.6-14.0) μm |
| • | Aperture: Tricolporate, pore round with lolongate annulus; colpi long, conspicuously extending through the entire length of the grain |
| • | Exine: Coarsely reticulate, areolate, heterobrochate, with a single row of bacule; sexine thicker than nexine, exine thicker at the equatorial area, exine surface psilate |
Asystasia schimperi: (Fig. 1)
| • | Shape: Triangular amb; radially symmetrical; Prolate grain; P/E (1.5) |
| • | Size: Polar axis 18.29 (14.0-21.0) μm; Equatorial axis 12.2 (8.75-14) μm |
| • | Aperture: Tricolporate, pore round, colpi long and wide, as long as the length of the grain |
| • | Exine: Coarsely reticulate, heterobrochate, with a single row of bacule; sexine thicker than nexine, exine thicker at the equatorial area |
Asystasia vogeliana: (Fig. 2)
| • | Shape: Tri angular amb; radially symmetrical, Prolate grain P/E (1.71) |
| • | Size: Prolate grain; polar axis 17.7 (14.0-21.0) μm; equatorial axis 10.4 (8.7-12.2) μm |
| • | Aperture: Tricolporate, pore round, with lolongate annulus; colpi long, conspicuously extending through the entire length of the grain |
| • | Exine: Coarsely reticulate; sexine as thick as nexine, baculate. Exine surface psilate |
Justicia elegantusa: (Fig. 2)
| • | Shape: Rounded triangular amb; bilaterally symmetrical; prolate grain; P/E: 1.49 (1.40-1.63) μm |
| Table 1: | Collection information and common names of taxa included in pollen atlas |
 | |
 | |
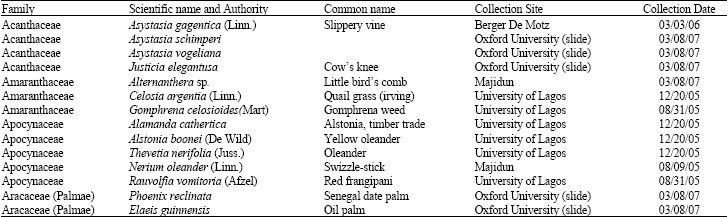
| Fig. 1: | (a-f) Asystasia gagentica. a and b: Equatorial view, c and d: Polar view, a and d: Light micrographs x 1000, e and f: SEM micrographs, e: x 3000, f: x 1000, (g-l): Asystasia schimperi, Lmx 1000, show aperture, sculpture and shape at equational view and k and l: Same grain at polar view |
| • | Size: polar axis: 38.6 (36.1-42.4) μm; equatorial axis 25.9 (23.4-28.4) μm |
| • | Aperture: Diporate, pore lalongate, pore height 3.8 (2.1-4.7) μm; pore width 5.5 (3.2-7.1) μm |
| • | Exine: Reticulate, with rounded areoles arranged in two ranks (4-6 each) on either side of the pore; areole diameter 3.4 (2.9-3.9) μm; sexine thicker than nexine, exine thicker around the equatorial region of the grain |
Amarantaceae:
Alternanthera sp.: (Fig. 3)
| • | Shape: Circular amb; spherical grain; P/E (1.11) |
| • | Size: Polar axis 15.31 (14.0-17.5) μm; Equatorial axis 13.8 (12.2-15.7) μm |
| • | Aperture: Inaperturate |
| • | Exine: Lophate, with round lacunae |
Celosia argentia: (Fig. 3)
| • | Shape: Circular amb; spherical grain |
| • | Size: Maximum dimension 27.6 (22.7-33.2) μm |
| • | Aperture: Periporate, oval pore |
| • | Exine: Pitted; exine heterobrochate, tectate |
 | |
| Fig. 2: | (a-f): Asystasia vogeliana, LM x 1000, a and b is showing the structure and aperture of the grain at equatorial view, c: Subequatiorial view, (d-f): Polar view, (g-i): Justicia elegantusa LM x 1000, g and h: equatorial view and I: Polar view |
Gomphrena celosioides: (Fig. 4)
| • | Shape: Circular amb; spherical grain; P/E (1.07) |
| • | Size: Polar axis 11.5 (10.5-13.1) μm; Equatorial axis 10.7 (8.7-12.2) μm |
| • | Aperture: Inaperturate |
| • | Exine: Coarsely reticulate, intectate |
Apocynaceae:
Alamanda cathertica: (Fig. 4)
| • | Shape: Rounded triangular amb, sides convex; circular to elliptical in equatorial view; pollen grain isopolar, radially symmetrical Prolate grain spheroidal grain P/E (1.16) |
| • | Size: Polar axis 29.4 (28.0-32.3) μm; Equatorial axis 25.3 (22.7-28.0) μm |
| • | Aperture: Tricolporate; colpi long, wide, pore round |
| • | Exine: Exine microreticulate, pertectate |
Alstonia booeni: De willd. (Fig. 5)
| • | Shape: Rounded triangular amb, sides convex; circular to elliptical in equatorial view; pollen grain isopolar, radially symmetrical Prolate grain spheroidal grain P/E (1.14) |
| • | Size: Polar axis 22.0 (17.5-28.0) μm; Equatorial axis 19.3 (14.0-24.5) μm |
| • | Aperture: Tricolporate; colpi long, narrow. Pore nearly circular or lalongate, each bordered by an annulus |
| • | Exine: Exine 1.4+/-0.1 μm thick in the inter-apertural regions, considerably thicker at the apertures, stratification indistinct, exine punctate |
 | |
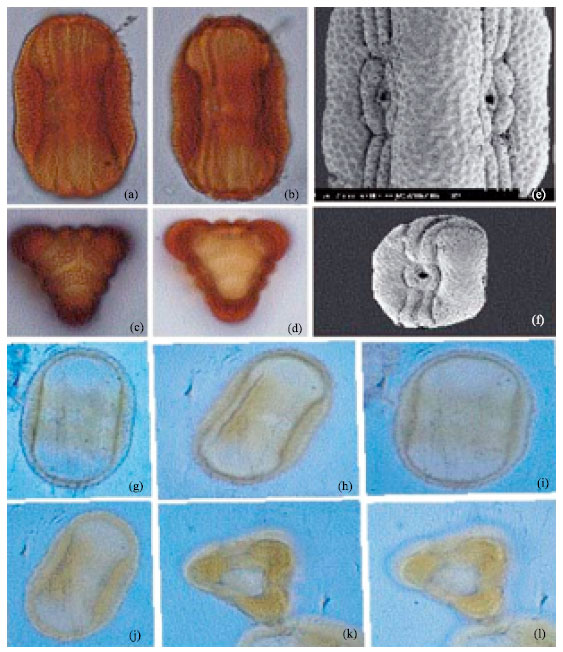
| Fig. 3: | (a-l): Showing lacunae and ridges in Alternanthera sp., LM x 1000 Celosia argentia. (m-r): showing spherical shape, circular aperture and pitted exine in pollen of Celosia argentia (Linn), LM x 1000 |
Thevetia nerifolia: (Fig. 5)
| • | Shape: Pollen grains isopolar, radially symmetrical; goniotreme, sides convex. P/E (1.11) |
| • | Size: Polar axis 30.8 (26.2-36.7) μm; Equatorial axis 27.7 (22.7-33.2) μm |
| • | Aperture: Tricolporate, colpi long and wide, pore oval, lolongate |
| • | Exine: Exine reticulate, tectate |
Nerium oleander: (Fig. 6)
| • | Shape: Spherical to triangular amb, radially symmetrical |
| • | Size: Polar axis 24.5 (19.2-29.7) μm; Equatorial axis 24.2 μm |
| • | Aperture: Triporate to tetraporate, pore nearly circular, lolongate, each bordered by an annulus |
| • | Exine: Exine thick, considerably thicker at the apertures, stratification indistinct |
Rauvolfia vomitoria: (Fig. 6)
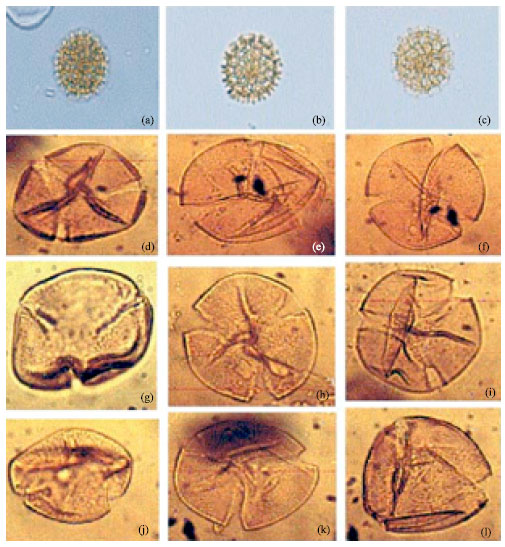 | |
| Fig. 4: | (a-c): Showing coarse reticulation of Gomphrena celosioides (Mart) LM x 1000, (d-l) Alamanda cathertica, LM x 1000. Note lolongate colpi and psilate structure of the exine |
| • | Shape: Amb angular, isopolar, radially symmetrical; sides convex to spherical; Prolate grain P/E (1.9) |
| • | Size: Polar axis 24.9 (20.1-33.2) μm; Equatorial axis 13.1 (14.0-17.0) μm |
| • | Aperture: 3-colporate, occasionally 4-colporate, syncolpate; Colpi united at the poles in most grains, tapering towards the poles. Ora large, longitudinally elongated, ca. 8.8 μm long, 2.0 μm wide each with a very narrow annulus |
| • | Exine: Reticulate, subtectate. Exine thin in interapertural regions (1.4+-0.1 μm thick), thicker at aperture, 2.4+/-0.4 μm thick |
Aracaceae:
Phoenix reclinata: (Fig. 6)
| • | Shape: Circular amb, radially symmetrical grain |
| • | Size: 21.3 (19.5-22.5) μm; Equatorial axis 22.0 (21.0-23.0) μm |
| • | Aperture: Monosulcate, sulcus long and wide |
| • | Exine: Reticulate, psilate |
Elaeis guinnensis: (Fig. 6)
| • | Shape: Oblate to angular amb |
| • | Size: 28.7 (27.5-30.0) μm; Equatorial axis 28.0 (27.0-29.0) μm |
| • | Aperture: Monosulcate to trichotomosulcate |
| • | Exine: Psilate, stratification indistinct |
Pollen in Acanthaceae family are relatively eurypalynous with wide pollen morphology and generally very beautiful. Acanthaceae are mostly prolate in equatorial view and trigonal to circular in polar view. Apertures are usually tricolporate, could be diporate as in Justicia elegantusa, which agrees with J. americana studied by Willard et al. (2004) sometimes heterocolpate as in Brillantasia lamium and tricolpate as found in Nelsonia canescens (Amule, 1999). In biostratigraphic application, the counterpart fossil for the species Asystasia gangetica is Areolites formosus. Polar view has never been identified as far as the author is aware.
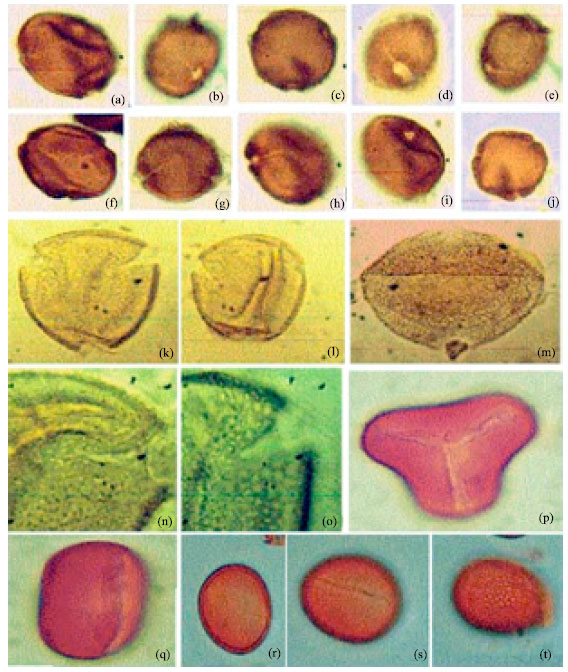 | |
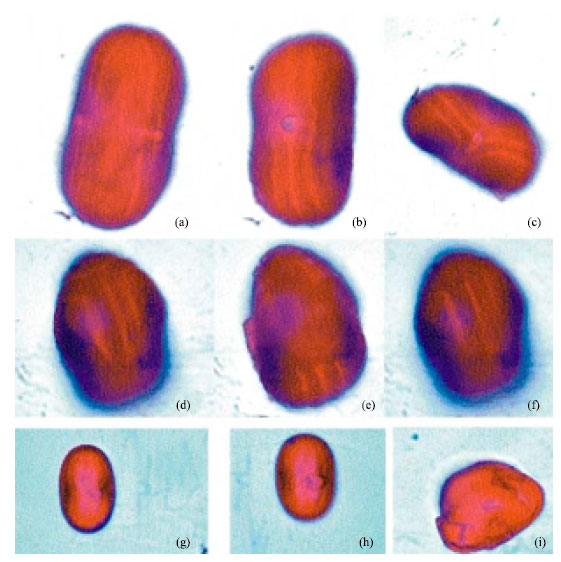
| Fig. 5: | (a-j): Alstonia boonei (De Wild), LM x 1000, (k-o): Showing punctuate structure in Thevetia nerifolia (juss.), LM, k-m x 400, n-o x 1000. (p-q): Elaeis guinnensis, LM x 1000. (r-t): Phoenix reclinata, LM x 1000 |
Amaranthaceae is also eurypalynous comprising of different morphological types of pollen, ranging from inaperturate to polyporate. The different species studied Gomphrena celosioides, Celosia argentia, Alternanthera sp., have one thing in common and that is the spherical amb (shape). The polyporate type pollen exhibited in some genera e.g., Celosia sp., are very similar to those in the Chenopodiaceae and that is why they are usually put together as Chenopod/Amaranth during routine analysis.
Genera in Apocynaceae exhibit palynological extremes. This is indicated by variety in the shape of the pollen grains, aperture, size and ornamentation of the studied species. Pollen grains in Aracaceae are usually monocolpate as found in Phoenix reclinata except E. guinnensis which exhibit variations ranging from monocolpate to trichotomosulcate aperture.
This study compliments the few existing atlases of pollen grains from tropical areas. It has provided additional characters for use in plant taxonomy, as well as serving as reference material for routine pollen analytical research.
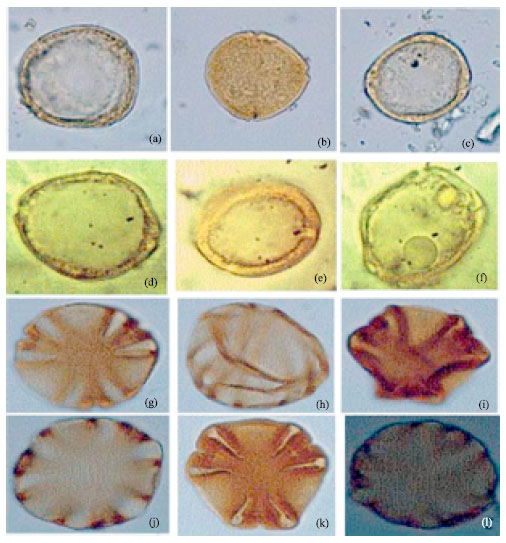 | |
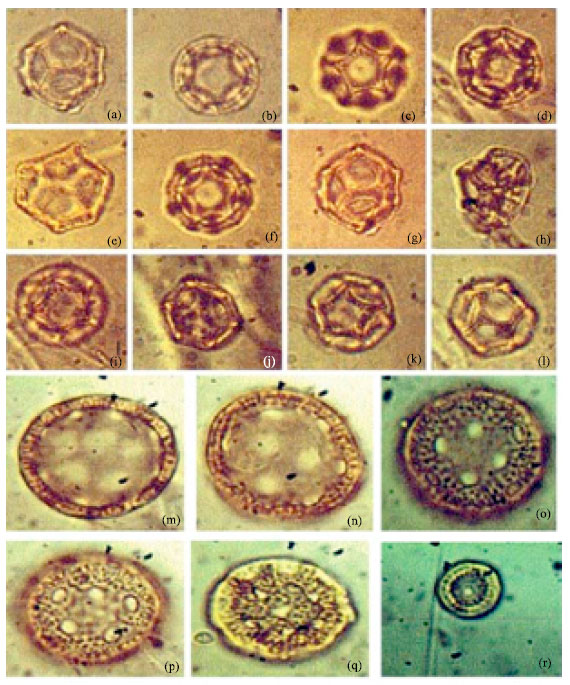
| Fig. 6: | (a-f): Showing polymorphism in the pollen of Nerium oleander (Linn.), LM x 1000, (g-l): Showing well developed margo in the pollen of Rauvolfia vomitoria (Afzel), LM x 1000 |
Special thanks to the University of Lagos for the approval of research leave during this study and also for funding. A deep sense of gratitude also goes to the Keeper, Professor Mark Chase of Jodrell Laboratory and Dr. Carol Furness (my host) of The Royal Botanic Gardens, Kew, London for the provision of bench space and equipment.