ABSTRACT
The seminal vesicle region of the hermaphrodite duct of the edible land snail, Limicolaria flammea (Müller) is described using light and electron microscopy. The lumen is lined with a single layer of epithelial cells surrounded by connective tissues and circularly arranged smooth muscle. Transmission electron microscopy reveals that there are two types of epithelial cells lining the lumen of the seminal vesicle duct. Type 1 epithelial cells are highly ciliated columnar with large irregular shaped basal nuclei. Type 2 epithelial cells are non-ciliated columnar with large irregularly shaped basal nuclei. The apical surface of the Type 2 cell bears numerous microvilli. Mitochondria, glycogen, lipids, rough and smooth endoplasmic reticulum, lysosomes, Golgi bodies and vacuoles are observed in the two types of epithelial cells and their lateral margins are joined apically by zonula adhaerens, below which are located septate junctions. The functional significance of the epithelial cells are discussed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.1835.1839
URL: https://scialert.net/abstract/?doi=pjbs.2007.1835.1839
INTRODUCTION
Terrestrial pulmonates are hermaphrodites, characterized by internal fertilization: which accounts for the complex reproductive system with varied functions (Gomez, 2001).
The hermaphrodite duct of pulmonates connects the ovotestis to the genital system (Hodgson, 1996). It is called a hermaphrodite duct because it conveys both ova and spermatozoa from the ovotestis. The hermaphrodite duct consists of three main regions, a proximal duct close to the ovotestis, the middle duct or seminal vesicle region and the distal duct close to the albumen gland and known as talon or receptaculum seminis (Bayne, 1973): the seminal vesicle is the largest of the 3 regions. In Succinea putris, in all mature and maturing snails prior to egg laying sperms are restricted to the gonads and seminal vesicle, but in snails raised in isolation, sperms are absent both prior to and after egg laying from the receptacula seminis (Bayne, 1973) but are present in the snails reared in communal containers, an indication that the receptaculum seminis is used for storage of foreign sperm received at copulation and the seminal vesicle for own sperm. However, many pulmonates lack receptaculum seminis as distinct organs (Bayne, 1973).
Limicolaria flammea (Müller, 1774), is an edible achatinid land snail endemic to West Africa (Bequaert, 1950; Crowley and Pain, 1970). The reproductive system of African Achatinidae was reviewed and classified to some systemic order by Bequaert (1950), Mead (1950) and Crowley and Pain (1970). Since this comprehensive description not much has been done in African Achatinidae. Although some aspects of the biology of Limicolaria have received some attention (Owen, 1966; Owiny, 1974; Egonmwan, 2004), no study has focused on the structure of the reproductive system of L. flammea at the electron microscopy level. Indeed, the structure of the hermaphrodite duct of pulmonates at light and electron microscopy level has been described only in few species (Tompa, 1984; Hodgson, 1996).
In this study, the structure of the seminal vesicle of the hermaphrodite duct of Limicolaria flammea is described.
MATERIALS AND METHODS
Collection of snails: Twenty specimens of L. flammea were collected by hand-picking from Iguobazuwa village, near Benin City in the rainforest area of southwest Nigeria (lat. 06° 191 N: long. 05° 361 E) between January and February, 2005 and the specimens were brought to the laboratory within 12 h after collection. The length of each snail was measured with a vernier caliper. To investigate the reproductive state of snails collected, the shell of each was broken open and the snail dissected under a binocular microscope. The entire reproductive system was removed and used for gross anatomical study. The hermaphrodite duct was removed from the reproductive system and the seminal vesicle which is the middle and major portion of the duct was dissected out and examined for presence of spermatozoa.
Light microscopy: Small pieces of tissues from different regions of the seminal vesicle were fixed in aqueous Bouin’s fluid. Fixed tissues were dehydrated in graded ethanol (70-100%) and embedded in paraffin wax through xylene. Transverse serial sections were cut at 6 μm, stained in haematoxylin and eosin observed under a light microscope.
Electron microscopy: Small pieces of tissues from the different regions of the seminal vesicle of the hermaphrodite duct of L. Flammea were fixed overnight in 2.5% glutaraldehyde in 0.2 M sodium cacodylate buffer with 5% sucrose (pH 7.8) at 4°C (Glauert, 1982; Reid, 1982). Fixed tissues were rinsed in several changes of sodium cacodylate buffer and post-fixed in 1.0% osmium tetroxide in cacodylate buffer for 2 h. The tissues were then dehydrated in graded ethanol and embedded in Araldite via propylene oxide. Ulthrathin sections were cut with glass knives using Reichert OM U3 ultramicrotome and picked on copper grids. Contrast was enhanced using uranyl acetate and Reynolds’s lead citrate (Reynolds, 1963) and sections were examined on a Philip 400T transmission electron microscope.
RESULTS
Gross anatomy of the seminal vesicle: The hermaphrodite duct in L. flammea connects the ovotestis to the genital system (Fig. 1). It originates as a slender tube where the collecting tubules from the ovotestis unite. The hermaphrodite duct is either dark brown or creamy white and it is highly folded upon itself and greatly distended. The hermaphrodite duct is swollen and sometimes very pronounced in L. flammea. The first region or proximal duct, close to the ovotestis is narrow, with highly ciliated epithelium (Fig. 2A) and does not store sperm. The second region or middle duct is dilated, bulbous, pigmented and contain spermatozoa ( Fig. 2B) and is referred to as seminal vesicle. When the seminal vesicle was opened, the seminal fluid contained in it was creamy white. In transverse section the seminal vesicle is highly folded and filled with spermatozoa (Fig. 2B). The seminal vesicle narrows distally near the albumen gland, then bends to form the third region of the hermaphrodite duct, receptaculum seminis or talon, which is a bulbous projection with a thin stalk in L. flammea. The hermaphrodite duct continues anteriorly towards the spermoviduct and the distal end of the third region within the albumen gland forms the carrefour or fertilization chamber with the ducts from the albumen gland, the male genital duct and the female genital duct.
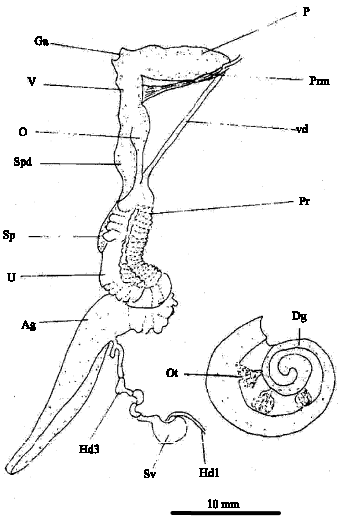 | |
| Fig. 1: | Diagrammatic reproductive system of Limicolaria flammea. Abbreviations: Ag, albumen gland; Dg, digestive gland; Ga, genital atrium; Hd 1, hermaphrodite duct (first region); Hd 3, hermaphrodite duct (third region); O, oviduct; Ot, ovotestis; P, penis; Prm, penis retractor muscle; Pr, prostate; Sp, spermatheca; Spd, spermatheca duct; Sv, seminal vesicle (second region of hermaphrodite duct); U, uterus; V, vagina; Vd, vas deferens |
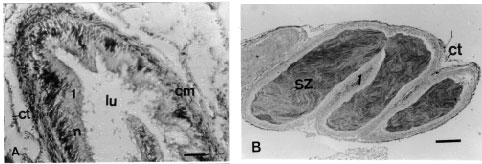 | |
| Fig. 2: | (A) Transverse section through the hermaphrodite duct (first region) of L. flammea showing the ciliated epithelium surrounded by circular muscle (cm) and connective tissue (ct). Section stained in haematoxylin and eosin. n, nucleus; lu, lumen. Scale bar = 200 μm. (B) Transverse section through the seminal vesicle (second region of the hermaphrodite duct) of L. flammea showing the ciliated cell (1) of the epithelium. Section stained in haematoxylin and eosin. ct, connective tissue; sz, spermatozoa. Scale bar = 200 μm |
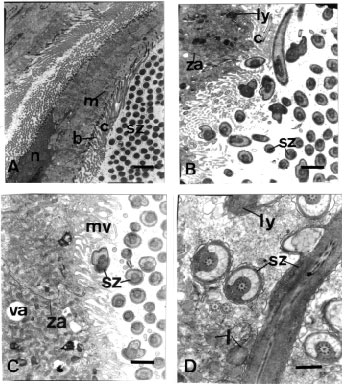 | |
| Fig. 3: | (A) Transmission electron micrograph through Type 1 (ciliated) epithelial cells from the seminal vesicle of L. flammea. Scale bar = 2μm. (B) Higher Magnification of the apical region of Type 1 cell of L. flammea. Scale bar = 1 μm. (C) Transmission electron micrograph through Type 2 (non-ciliated) epithelial cell from the seminal vesicle of L. flammea. Scale bar = 1μm. (D) Transmission electron micrograph through the cytoplasm of a type 2 cell of L. flammea in which spermatozoa are contained. Scale bar = 0.5μm. b, basal body; c, cilium; l, lipid; ly, lysosome; m, mitochondrion; mv, microvillus; n, nucleus; sz, spermatozoa; va, vacuole; za, zonula adhaerens |
The whole complex is held together by connective tissue.
Transmission electron microscopy shows that wall of the seminal vesicle of the hermaphrodite duct consists of a highly folded single layer of epithelial cells, a layer of connective tissue and a layer of circular muscle (Fig. 3). TEM reveals two types of epithelial cells. Type 1 (Fig. 3A, B) is highly ciliated columnar, with large irregular shaped basal nucleus. The cytoplasm contains numerous mitochondria, rough and smooth endoplasmic reticulum, lipid droplets, lysosome and Golgi bodies and irregularly shaped vacuoles. The lateral margins of adjacent ciliated cells are joined apically by zonula adhaerens, below which are located septate junctions (Fig. 3A). Many mitochondria are located close to the basal bodies and striated bodies of the cilia. Also found in the cytoplasm are putative glycogen granules, lipid droplets, few lysosomes and numerous cisterna of smooth endoplasmic reticulum. Type 2 cells (Fig. 3C) are also columnar with large irregularly shaped basal nucleus. The apical surface of the cell bears numerous microvilli. There are mitochondria, Golgi body, glycogen, few lipid droplet, lysosomes, and vacuoles. Some of these cells contain remnant of spermatozoa which may be undergoing the process of phagocytosis (Fig. 3D). The lateral margins of adjacent non ciliated cells are joined apically by zonula adhaerens, below which are located septate junctions.
DISCUSSION
The seminal vesicle region of the hermaphrodite duct is swollen and sometimes very pronounced in L. flammea as in Physa sp. (Duncan, 1958), Succinea putris (Rigby, 1965), Aplysia sp. (Thompson and Bebbington, 1969) and Bursatella leachi (Bebbington, 1969). It consists of a single layer epithelial layer lining the duct and a thin muscular and connective tissue layers. The seminal vesicle of L. flammea is highly folded and was greatly distended as a result of stored auto sperm. Mead (1950) reported that in Archachatina marginata ovum, the seminal vesicle is crammed with sperm, even in specimens that have copulated. The dark coloration of the hermaphrodite duct of L. flammea is probably due to melanin pigments. And at high egg production, almost the whole length of the hermaphrodite duct is creamy colour instead of dark brown or black. The results confirm observations of Tompa (1984) and Hodgson (1996) in pulmonates.
Two types of epithelial cells were identified in the seminal vesicle of L. flammea during this study. Two types of epithelial cells were also reported in the seminal vesicle of Helix pomatia (Breucker, 1964), Biomphalaria glabrata (de Jong-Brink, 1969), Achatina fulica (Hodgson, 1996) and Siphonaria capensis (Hodgson, 1996). These cells are named type 1 and type 2 (Hodgson and Shachak, 1991; Hodgson, 1996). Type I cells are ciliated while type 2 cells are non-ciliated / glandular or secretory (Hodgson, 1996). The two types of epithelial cells in the seminal vesicle of L. flammea contain glycogen and lipid which are storage compounds (Hodgson, 1996) which may play a role in sperm maintenance (Holm, 1946). The seminal vesicles of L. flammea examined during this study contain large number of sperms, which may confirm the suggestions that the area is used for sperm storage by the snail and also the maintenance of these sperm while within the seminal vesicle. The length of time the sperm remain in the seminal vesicle varies in different species hence maintenance of these sperm while in the seminal vesicle is very important. Although this was not investigated during this study, Hodgson and Shachak (1991) gave a period of about three months from the time sperm first appeared in the seminal vesicle of Sphincterochila zonata and S. prophetarum before copulation takes place.
The auto-sperm in the swollen seminal vesicle of L. flammea were observed to be inactive during this study. Similar observation was reported by Hodgson (1996). They are probably expelled by contraction of the muscular wall as in Succinea putris (Rigby, 1965) and pass along the hermaphrodite duct to the sperm channel by ciliary action of type 1 cells in its wall. The long cilia in the sperm channel assist their movement out of the seminal vesicle (Lusis, 1961) and this may prevent the sperm from entering the uterine lumen as in Arion ater (Lusis, 1961) and Helix pomatia (Breucker, 1964).
A number of functions have been suggested for the seminal vesicle of the hermaphrodite duct in pulmonates snails; such as sperm storage (Breucker, 1964; Buckland-Nicks and Chia, 1976; Garaerts and Joose, 1984; Tompa, 1984; Runham, 1988; Hodgson, 1990; Hodgson and Shachak, 1991; Hodgson, 1996), and sperm reabsorption and degeneration (Hodgson, 1996; Hodgson and Shachak, 1991), maintenance of auto spermatozoa by glycogen secretion from the ciliated cells during their storage (Kugler, 1965; Els, 1978; Hodgson and Shachak, 1991; Hodgson, 1996). Further maturation of the auto spermatozoa is also believed to occur in the seminal vesicle (Hodgson, 1996). It has been suggested that the ciliated cells may aid in the movement of gametes while the non-ciliated cells are probably involved in resorption and degeneration of spermatozoa in the seminal vesicle (Hodgson, 1996). The sperm are engulfed by the epithelial cells and degraded by lysosomal action on waste or excess spermatozoa (Nemeth and Kovaks, 1972; Buckland Nicks and Chia, 1976; Hodgson, 1996) or abnormal sperm (Tompa, 1984; Hodgson, 1996).
The functional differentiation of the seminal vesicle of L. flammea indicates that the region is an important region both for sperm maturation and resorption and degeneration.
REFERENCES
- Reynolds, E.S., 1963. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol., 17: 208-212.
CrossRefPubMedDirect Link








