Research Article
Ultrastructural Study of Muscles Fibers in Tick Hyalomma (Hyalomma) anatolicum anatolicum (Ixodoidea: Ixodidae)
University College, Makkah, Department of Biology, Umm Al-Qura University, Saudi Arabia
Ixodid ticks are highly specialised bloodsucking arthropods. The tick is known to be one of the important reservoir and vectors of microorganisms causing diseases to livestock and wild life (Hoogstraal et al., 1981). The ticks have the ability transmit certain pathogens to their progeny via transstadial and transovarial infection (Burgdorfer and Varma, 1967).
Specialized muscles are of the same origin as the somatic but are limited to a particular part of the body for a specific purpose. Somatic muscle terminology relates to the shape and the arrangement within the nematode. It may be platymyarian where the sarcoplasm is exposed on three sides; coelomyarian, where the sarcoplasm bulges in to the pseudocoel and the fibers extend up the sides; or circomyarian, where striated fibers surround the sarcoplasm (Smith, 1968, 1984).
Douglas (1943) and Balashov (1972) have described the musculature structure of the tick early. Balashov (1972) distinguished four groups of muscle according to their function and location; the capitulum musculature, leg muscles, skeletal muscles and visceral muscles. However, all that previous studies have been made using light microscope technique. In insects the relationship between muscles and the components of the integument in muscle insertions have been studied by Wong et al. (2005) with the electron microscope level in two insects, Calpodes ethius (Hesperiidae, lepidopetra) and Rhodnius prolixus (Reduviidae, hemiptera). The result shown that interdigitating processes whose membranes are joined by intermediate junctions increase the area of contact between the muscles and the epidermis. Also, Wong et al. (2005) studied the changes in interstitial cells of Cajal at the deep muscular plexus are associated with loss of distention-induced burst-type muscle activity in mice infected by Trichinell spiralis. The result shown the physiology and pathophysiology of the network of interstitial cells of Cajal associated with the deep muscular plexus (ICC-DMP) of the small intestine are still poorly understood. The author conclude that transient injury to ICC-DMP in response to T. spiralis infection is severe and associated with a complete lake of distention-induced burst-type muscle activity. Recent study shown that the neuromuscular system of Drosophila melanogaster has been studied for many years for its relative simplicity and because of the genetic and molecular versatilities. This study recorded three main types of striated muscles are present in this dipteran: fibrillar muscles, tubular muscles and super contractile muscle (Peron et al., 2009). Furthermore, the visceral muscles in adult flies and the body wall segmental muscles in embryos and larvae belong to the group of super contractile muscles (Peron et al., 2009). Larval body wall muscles have been the object of detailed studies as a model for neuromuscular junction function but have received much less attention with respect to their mechanical properties and to the control of contraction.
Contompasis et al. (2010) have described flightin is necessary for length determination, structural integrity and large bending stiffness of insect flight muscle thick filaments. In this study, they used atomic force microscopy to study the morphological and mechanical properties of fully hydrated native thick filaments isolated from Indirect Flight Muscle (IFM) of normal and mutant Drosophila lacking flightin (fln0). IFM thick filaments from newly enclosed (0-1 h old) wild-type flies have a mean length of 3.04±0.05 μm.
The aim of present study was to describes the ultrastructure and description of muscle of the of ixodid tick Hyalomma (Hyalomma) anatolicum anatolicum using the electron microscope technique.
Ticks Hyalomma (H) anatolicum anatolicum were obtained from a colony maintained at 28°C and 75% relative humidity in 2009 at the Department of biology, University College Umm Al-Qura University, Saudi Arabia. Unfed, nymph were excised transversely into halves just behind the fourth coxae and directly fixed in 3% glutaraldehyde in sodium cacodylate buffer (pH 7.2), post-fixed in OsO4, dehydrated in alcohol series and embedded in low viscosity resin (Spurr, 1969). For transmission electron microscopy (TEM) ultrathin sections were double-stained with uranyl acetate and lead citrate and examined under a Philips 400 TEM. Images were recorded on Ilford E.M cut film and printed on Ilfospeed multigrade paper (Bughdadi, 1989, 1999, 2008).
The results showed that muscles of the unfed ticks Hyalomma (Hyalomma) anatolicum anatolicum in longitudinal sections are spindle-shaped to cylindrical muscle fibers and have the known striated pattern of successive sarcomeres (Fig. 1). The muscles of the unfed ticks are distinguished according to structure, function position and into visceral and skeletal. The visceral muscles invest the gut, excretory system and other internal organs. The skeletal muscles are inserted either directly onto the body integument or onto inwardly directed apodermal extensions of the cuticle. These muscles include the capitulum, dorsoventral and leg oblique muscles. All muscle fibers are ensheathed in a sarcolemma (Fig. 3 and 4). Each sarcomere extends between adjacent Z-discs of the myofibril; the Z-disc is circumference by light I-bands and greater part of sarcomere is occupied by the denser A-band in relaxed muscles fibers (Fig. 1). The fibril sub-units, the myofilaments, from 4-5 nm diameter, correspond to the filament containing the protein actin, found in striated muscles of other arthropods, including vertebrates and other insects; these extend between the Z-discs. The thicker filaments about 15 nm diameter, extend through the A-bands and ending at the edge of the I-band regions (Fig. 1). These filaments corresponding to the filament containing the protein myosin. The light I-bands are greatly shortened in the case of contracted muscles (Fig. 2).
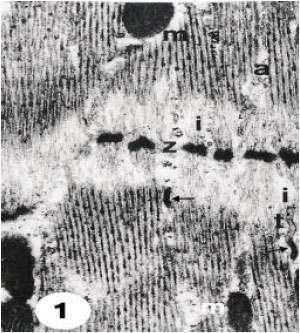 | |
| Fig. 1: | Ultrathin section of relaxed muscle fibres in dorsoventral skeletal muscles, showing successive sarcomeres. Light I-bands (i) occur consisting of actin filaments between the Z-discs (z) and the thick myosin filaments extended between the thin filaments in A-band (a). Note the arrows point showings the sarcoplasmic reticulum associated with T-system (t). Mitochondria (n) and nucleus (n) are observed. 10500 X |
 | |
| Fig. 2: | Ultrathin section of contracted muscle showing disappearance of I-bands seen in relaxed fibres which, seen in Fig. 1. 15000 X |
 | |
| Fig. 3: | Ultrathin section of parts of dorsoventral skeletal muscle fibres showing cell membrane invaginations (small arrow) extending into the sarcoplasm as tubular system (t) and closely associated to sarcoplasmic reticulum (large arrow). Note. Mitochondrion (m) and sarcoplasm (sl). 35000 X |
The Study showed that cell membrane invaginates into tiny tubules traversing the sarcoplasm (Fig. 3) which present closely associated to cisternae of sarcoplasmic reticulum (Fig. 1-4). The thick myosin filaments are present surrounded by 12 actin thin filaments (Fig. 4).
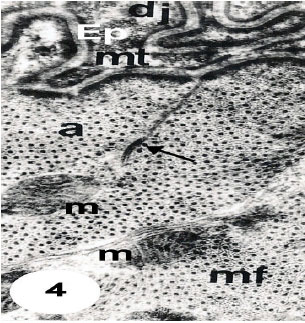 | |
| Fig. 4: | Ultrathin section in dorsoventral muscle fibres (mf) joining epidermal cells (Ep) by desmosome junction (dj). Note the numerous microtubules (mt) oriented parallel to the long axes of the adjacent muscle fibres. Note also that each thick myosin filament is surrounded by orbitals of 12 thin actin filaments. Note a-band (a), mitochondrion (m) and arrow points showing the association between T-system and sarcoplasmic reticulum. 15000 X |
The basal surface of the epidermal cell has a process that interdigitate with similar projections of the muscle cell. The apposed plasma membranes are linked by junctions and desmosome (Fig. 4). The cytoplasm of the epidermal cell appears largely devoted with complicated microtubules present in parallel with long axis of adjacent muscle fibers (Fig. 4).
In the sarcoplasm, the results recognized two types of muscle fibers according to four different measures, thickness and density and the mitochondrial size, distribution and population. The thin shape, 30-35 Fm in diameter, is first type, which present with electron-lucent interfilamentous space and their small, mitochondrial are round and have transverse cristae and clear intercisternal matrix. All these mitochondria appear accumulated in one side of the sarcoplasm (Fig. 5). the second type of muscle fibers are thicker than the first type about, 65-85 μm in diameter, with moderately dense interfilamentous space. their mitochondria are large oval to elongate and have irregularly arrayed or whorled cristae and dense intercisternal matrix (Fig. 5). Their mitochondria are present between the myofilaments (Fig. 5) or accumulated in one part of the cell (Fig. 6). Tracheoles are found in extracellular spaces, or invaginated into the depths of the cell (Fig. 5). The nuclei appear oval to elongate and are present beneath the cell membrane (Fig. 6).
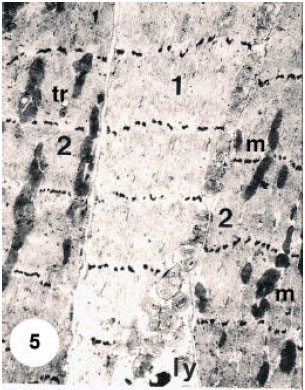 | |
| Fig. 5: | Ultrathin section in dorsoventral muscle fibres (mf) joining epidermal cells (Ep) by desmosome junction (dj). Note the numerous microtubules (mt) oriented parallel to the long axes of the adjacent muscle fibres. Note also that each thick myosin filament is surrounded by orbitals of 12 thin actin filaments. Note a-band (a), mitochondrion (m) and arrow points showing the association between T-system and sarcoplasmic reticulum. 15000 X |
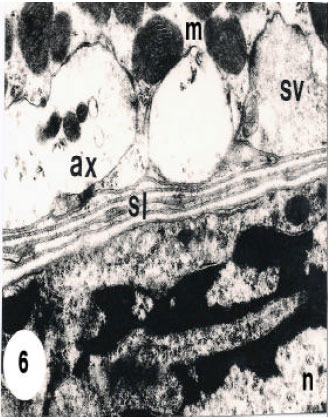 | |
| Fig. 6: | Ultrathin section of thick fibres from leg muscles showing accumulation of mitochondria (m) with irregularly arryed crista and dense intercisternal matrix. Note axons (ax) with synaptic vesicles (sv) embedded in the sarcoplasm. Note also nucleus (n) and sarcolemma (sl). 13500 X |
In the extracellular spaces, tracheole like shape is present and invaginated into the depths of the cell (Fig. 5). Axons are also found in the sarcoplasm and appear with synaptic vesicles (Fig. 6). The actin and myosin filaments, which present in visceral muscle fibers, appear occupying most of the cell volume extend through the cytoplasm (Fig. 1, 2 and 4).
The tick musculature has been earlier described by many authors in different levels of detail (Robinson and Davidson, 1913; Ruser, 1933; Douglas, 1943; Balashov, 1972). However, the structural design of the skeletal and visceral muscle fibers in the unfed nymph H. anatolicum is mainly similar to the striated muscles described for insects (Smith, 1968, 1984), vertebrates (Albert et al., 1983) and other arthropods (Hoyle and McNeill, 1968; Pringle, 1980). The attachment of skeletal muscles to the cuticular exoskeleton and to the abdomens is described and contrasted with the attachment of the muscular sheath surrounding the oesophagus of the tick, boophilus decoloratus (Beadle, 1973). However, Caperucci (2009) described that the digestive tube of the tick Amblyomma cajennense is responsible for the digestion during feeding on the host. Moreover, this study analyzed the midgut of unfed, partially engorged and fully engorged fed females as well as three infestations in rabbits. In A. cajennense, the midgut of unfed females possesses a pseudostratified epithelium composed of digestive and generative cells. However, perhaps the study of Caperucci is indirectly related in one way or another to study has been described by Beadle (1973) or probably extension to it, as they relate to the digestive system fiber.
Although the general similarity between the sarcomeres of H. anatolicum, insect and vertebrates, closer examination of longitudinal sections shows that, the arrangement of actin and myosin filaments within the A-band has a different shape. In H. anatolicum each actin filament in the A-band array lies midway between two myosin filaments. These results similar to that found in insects (Huxley and Hanson, 1957; Shafiq, 1963; Auber, 1965, 1967; Smith, 1965, 1966; Reger and Cooper, 1967), whereas in vertebrate skeletal muscle each actin filament is (shared) by three myosin filaments (Huxley and Hanson, 1957). Although, skeletal muscle fibers in H. anatolicum are similar to those of insects in having the same ratio of actin and myosin filaments; each myosin filament is surrounded by orbitals of up to 12 actin filaments, but 7 to 9 actin filaments and double hexagonal arrays of myofilaments were recorded in insect legs and flight muscles (Auber, 1967; Reger and Cooper, 1967). The increase in the actin filaments in number are similar to that shown by Smith (1968) who suggested that to be correlated with the slower or more infrequent activity. The invaginated tubules system (T-system) appear to be derived from the cell membrane and also appear closely associated the adjoining sarcoplasmic reticulum to probably form two-membranes (dyads) illustrated in the skeletal and visceral muscle fiber. This study is basically similar to skeletal muscle described for insect (Smith, 1984; Pringle, 1981).
Muscles, in vertebrate each transverse tubule usually lies between two (terminal cisternae) of the reticulum to constitute a three-membranes (triad). Moreover, in insect flight muscles, the transverse tubules are retained, but the sarcoplasmic reticulum is markedly reduced into small vesicles lying alongside the tubules (Ashhurst, 1967). These fibers certainly these fibers certainly working to move the wings at very high frequencies. However, the job carried out by the T and SR in muscle contraction has been variously studied (Smith, 1968, 1984). When the motor nerve broadcast a signal in the muscle-cell plasma lemma, this electrical excitation spreads rapidly through the T-system and subsequently into the SR. The latter releases into the cytosol a large amount of Ca++ stored in its lumen. The resulting fortuitous rise n free Ca++ concentration initiates myofibril contraction.
The thickness and density and mitochondrial of the two types of skeletal muscle fibers are varying in thickness and density. Furthermore, distribution and population of all these organelles seen in the present study, probably represent phases of physiological operations of the same cell type. In the present study, obvious increasing in number, content and population of the mitochondria are seems to provide the required energy through ATP synthesis was occurred in insect muscles, but the variable n within the sarcoplasm poses a problem concerning the morphogenetic relationship between the developing contractile material and the surrounding cytoplasm and its contents (Brosemer et al., 1963; Smith, 1984). Peron et al. (2009) established that whether this system has the requisites to be considered a good model in which to perform a functional characterization of Drosophila genes, with a known muscular expression, as well as Drosophila homologs of human genes, the dysfunction of which, is known to be associated with human hereditary muscle pathologies.
The visceral muscles fibers appear in the present study are similar in the structure to those of insects and are considered to achieve slow contractions similar to those of vertebrate smooth muscles; these lack the T- system and SR. The physiology and biochemistry of these muscle cells investing the tick and insect internal organs are insufficiently known and need for require further studies. In addition to studies that rely on high-technology laboratory tests such as histochemistry and biochemical analysis. General plans for the future of the research work we would like to perform determination depend on molecular characterization of the ticks organs.
ACKNOWLEDGMENT
Author express his appreciation to Professor Dr. El-Sayed Fahim, Department of Biology, University College, Makkah, for his support and encouragement.