ABSTRACT
Double infection by zucchini yellow mosaic virus (ZYMV) and cucumber mosaic virus (CMV) of the cucurbit host Cucurbita pepo (zucchini squash) cv. Eskandarani resulted in a synergistic effect where symptoms in doubly infected plants were more enhanced than plants singly infected by either virus. ELISA values revealed that the concentration of CMV in doubly infected zucchini squash plants is significantly higher than that of singly infected plants. However, ZYMV titer did not increase significantly in case of double infection compared to singly infected plants. Increase of CMV in doubly infected zucchini squash cv. Eskandarani was confirmed by local lesion assays and specific infectivity and it increased with increase in time post inoculation.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ppj.2003.85.90
URL: https://scialert.net/abstract/?doi=ppj.2003.85.90
INTRODUCTION
Infection of plants by multiple viruses is a common phenomenon (Falk and Bruening, 1994). Double or mixed infection of plants often results in intensified symptom severity and higher virus accumulation, a phenomenon referred to as synergy (Sano and Kojima, 1989; Pruss et al., 1997). In nature, mixed viral infections of many crops have had biological and epidemiological implications that have been realized and addressed in many studies (Kassanis, 1963; Rochow, 1972; Wang et al., 2002).
Zucchini yellow mosaic virus (ZYMV), like other species of the Potyviridae is transmitted by aphids in a non-persistent manner. The virus is also transmitted by mechanical inoculation but is not seed transmitted (Lisa and Lecoq, 1984). ZYMV frequently occurs with other viruses in cucurbits. It was first reported in Italy in 1981 (Lisa et al., 1981). The virus is now known to cause devastating epidemics in commercial cucurbits world wide (Lisa and lecoq, 1984; Said et al., 1999).
Cucumber mosaic virus (CMV) belongs to genus Cucumovirus of family Bromoviridae. Hosts of the virus belong to over 9 different plant families. The virus is transmitted by mechanical inoculation and by seed. Aphid vectors transmit the virus in a non-persistent manner (Francki, 1985). Several studies have reported double infections involving combination of potyviruses and viruses belonging to other genera (Rochow and Ross, 1955; Damirdagh and Ross, 1967; Sano and Kojima, 1989; Bourdin and Lecoq, 1994; Latham and Jones, 2001).
In this study, ELISA is used to monitor the virus titer in singly and doubly infected Cucurbita pepo (zucchini squash) plants grown under green house conditions. Zucchini squash plants were inoculated with ZYMV, CMV or both viruses. The relationship between virus concentration and symptom severity of doubly infected plants is discussed.
Materials and Methods
Viruses
A subculture of Zucchini yellow mosaic virus (ZYMV) (Said et al., 1999) was obtained through the ARC Giza, Egypt. The virus isolate was maintained and propagated in Cucurbita pepo (zucchini squash) cv. Escandarani. Seeds were germinated in 10 cm diameter pots (5 seeds pot-1). The plants were grown in an insect proof green house under day light for a 12-hr photoperiod. The temperature in the green house varied between 20-30°C. Treatments included inoculation with ZYMV, CMV or with a mixture of the two viruses.
An isolate of CMV was obtained from naturally infected cucumber (Cucumis sativus) plants in Nubaria, Alexandria, and verified by ELISA using CMV antisera. The CMV virus was inoculated 3 consecutive times in a local lesion host Phaseolus vulgaris. The virus was maintained and propagated in Nicotiana glutinosa under greenhouse conditions.
Serology
The double antibody form of ELISA was the main virus assay method used in this study (Clark and Adams, 1977). Commercial DAS-ELISA Kits specific for ZYMV and CMV (Agdia products) were used according to manufacturer’s instructions. For ELISA, samples of leaf extracts were tested at a dilution of (1:1000, w/v). A control of uninoculated plants was also included. Virus inocula were prepared by grinding equal fresh weights of young systemically infected leaves of source plants in a mortar and pestle with 0.05 M potassium phosphate buffer, pH 7.0 and mixing the extracts prior to inoculation. The source plants were C. pepo or N. glutinosa plants infected with either ZYMV or CMV virus respectively. All inoculations were made by rubbing carborundum-dusted leaves with each inoculum. Inocula of ZYMV, CMV, or a mixed inoculum of both viruses were applied to leaves of test plants 10 days after planting. In all experiments, six plants were used per treatment.
Specific infectivity of CMV
The effect of age of infection on CMV titer in singly and doubly infected C. pepo cv. Eskandarani plants was evaluated by ELISA.. Infectivity assay of CMV from singly or doubly infected C. pepo plants was performed on Phaseolus vulgaris L. Pinto, the local lesion host for CMV. For this purpose three leaf discs from the second leaf of singly or double infected C. pepo (one disc per leaf) were tested at different intervals after inoculation by either ELISA or infectivity test. Leaf extracts were prepared in PBS-PVP-T (1:20, w/v) and rubbed on eight half leaves of the assay plant. The presence of ZYMV in extracts from doubly infected C. pepo plants did not pose problems in scoring the results as P. vulgaris is not a local lesion host of ZYMV.
Results
Symptoms on singly and doubly infected plants
Either virus typically induced severe symptoms after 10 to 14 days post inoculation. Symptoms were chlorotic local lesions, yellow-green mosaic and leaf deformations. C. pepo cv. Eskandarani coinfected by ZYMV and C MV displayed strong synergistic responses in symptom expression compared to plants infected by either virus alone.
| Table 1: | Comparison of cucumber mosaic virus (CMV) titer in leaves of Cucurbita pepo cv. Eskandarani singly and doubly infected with CMV and zucchini yellow mosaic virus (ZYMV) as determined by enzyme-linked immunosorbent assay (ELISA) at various intervals following inoculation |
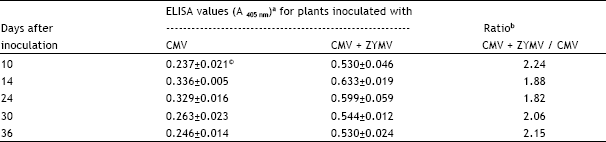 | |
| a Leaf extracts were prepared by homogenization with PBS-PVP-T and dilutions of 1:1000 (w/v) were tested by ELISA. Leaf extracts of comparable leaves from plants singly infected with ZYMV served as controls and gave negligible ELISA values (0.001-0.005). | |
| b Ratios for CMV titer in doubly: singly infected plants. | |
| c Values are means ± standard error for leaf extracts from six plants with each extract tested in duplicate wells, the plates were read with ELISA reader 20 min following addition of substrate. | |
| Table 2: | Comparison of Zucchini yellow mosaic virus (ZYMV) titer in leaves of Cucurbita pepo cv. Eskandarani singly and doubly infected with ZYMV and cucmber mosaic virus (CMV) as determined by enzyme-linked immunosorbent assay (ELISA) at various intervals following inoculation |
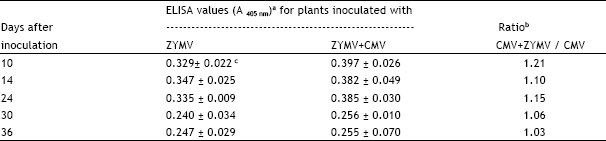 | |
| a Leaf extracts were prepared by homogenization with PBS-PVP-T and dilutions of 1:1000 (w/v) were tested by ELISA. Leaf extracts of comparable leaves from plants singly infected with CMV served as controls and gave negligible ELISA values (0.001-0.005). | |
| b Ratios for ZYMV titer in doubly: singly infected plants. | |
| c Values are means ± standard error for leaf extracts from six plants with each extract tested in duplicate wells, the plates were read with ELISA reader 20 min following addition of substrate. | |
Stunting and leaf deformations were much more pronounced in case of mixed infection of zucchini squash. Stunting of the doubly infected plants was a result of shorter internode lengths rather than slower development of the vegetative stages. Symptoms did not fade during the experimental period.
Comparative titer of CMV in singly and doubly infected plants
The CMV titer from singly and doubly infected C. pepo cv. Eskandarani plants varied during the experimental period (Table 1). The virus titer was highest 14 days post inoculation for either singly or doubly infected plants. The titer in the doubly infected plants had significantly higher CMV titer than did the corresponding singly infected plants (Table 1). The ratio of CMV titer in doubly: singly infected plants ranged from 1.82 to 2.24 with a mean value of 2.03 (Table 1).
| Table 3: | Effect of age of infection on cucumber mosaic virus (CMV) titer singly or doubly infected with CMV and zucchini yellow mosaic (ZYMV) as determined by local lesion infectivity assay and enzyme-linked immunosorbent assay (ELISA) |
 | |
| a Zucchini squash seedlings were inoculated at the primary leaf stage with CMV or with a mixed inoculum of CMV and ZYMV. | |
| bZucchini squash leaf extracts, prepared in PBS-PVP-T at a dilution of 1:20 (w/v), were rubbed on eight half-leaves of Phaseolus vulgaris ‘Pinto’ plants. | |
| cSubsamples of the same extracts used for the local lesion assay were tested by ELISA at dilutions of 1:1000 (w/v). | |
| dSpecific infectivity calculated by dividing mean local lesions per half-leaf by the corresponding ELISA absorbance value measured at 405 nm. | |
| eValues are mean local lesion numbers per half-leaf. | |
| fValues are averages for duplicate wells, plates reading was made 20 min after the addition of substrate. | |
Comparative titer of ZYMV in singly and doubly infected plants
No significant differences in ZYMV titer were detected between singly and doubly infected C. pepo cv. Eskandarani plants (Table 2). The ratios of ZYMV titer in doubly:singly infected plants ranged from 1.03 to 1.21 with a mean value of 1.11 (Table 2).
Effect of infection on CMV titer
The CMV titer as determined by ELISA in singly and doubly infected C. pepo cv. Eskandarani increased with age of infection (Table 3). The results of local lesion assays were highly correlated with those of ELISA made with samples of some extract at 10 and 24 days after inoculation, respectively. The specific infectivity (Local lesions / ELISA absorbance at 405 nm) of CMV increased by 9.7 and 11% in singly and doubly infected plants respectively (Table 3).
DISCUSSION
Infection of cucurbit species by potyviruses such as ZYMV, or by CMV are very common and cause considerable damage worldwide in severe epidemics in cucurbit fields either in single or mixed infections (Grafton-Cardwell et al., 1996; Luis-Arteaga et al., 1998). Double infection by CMV and the potyvirus ZYMV resulted in a synergistic effect on the cucurbit host zucchini squash cv. Eskandarani such response was obvious in terms of more severe symptom expression. A notable increase in host symptoms as a result of double infection were reported in several studies involving different combinations between potyviruses and viruses belonging to other genera. Examples of such synergistic effect was reported between Potato virus Y, Tobacco vein mottling virus or Tobacco etch virus together with the potexvirus Potato virus X in tobacco (Rochow and Ross, 1955; Damirdagh and Ross, 1967; Vance, 1991 and Vance et al., 1995); Zucchini yellow mosaic virus (ZYMV) and either Cucurbit aphid-borne yellow mosaic in muskmelon (Bourdin and Lacoq, 1994) or the cucumovirus Cucumber mosaic virus in cucumber (Poolpol and Inouye, 1986).
In most of such interactions, the accumulation of the potyvirus did not increase, but the accumulation of the other virus did increase (Anjos et al., 1992; Goldberg and Brakke, 1987; Pruss et al., 1997; Rochow and Ross, 1955 and Vance, 1991). However, mixed infection with a potyvirus which did not exhibit synergy was also reported of Peanut mottle virus with either tomato spotted wilt virus (Hoffmann et al., 1998) or Bean pod mottle virus (Anjos et al., 1992). Reports of increase or decrease in effect were also outlined (Karyeija et al., 2000).
Evidence is presented that supports the conclusion that the concentration of CMV in zucchini squash plants doubly infected with CMV and ZYMV is significantly higher than that in singly infected plants.
Much less is known about synergistic interaction occurring in cucurbit species doubly infected by CMV and potyviruses. Wang et al. (2002) reported strong synergistic pathological responses in mixed infection with ZYMV and CMV in zucchini squash. This was attributed to increase in the level of accumulation of CMV (+) RNA strand level. Comparing CMV titer in singly and doubly infected plants revealed that it was significantly higher in doubly infected zucchini squash.
The mean ratio of double to single infection is 2.03, this is in agreement with reports by Poolpol and Inouye (1986) of increase in CMV multiplication in cucumber in case of double infection by ZYMV. As for the titer of ZYMV, it did not show any significant change in single and double infections, the mean ratio of double to single infection being 1.11. Similar observations were reported by Wang et al. (2002). They report that ZYMV accumulates to similar levels in singly and mixed infected zucchini squash and melon. CMV specific infectivity increased slightly with age with 9.7 to 11% in singly and doubly infected zucchini squash respectively. Enhancement of CMV levels during synergy was due to both an increase in the number of CMV particles per cell, as well as the number of CMV-infected cells (Poolpol and Inouye, 1986). However, decline in the level of viruses with age was reported for several other viruses (Calvert and Ghabrial, 1983).
REFERENCES
- Anjos, J.R., U. Jarlfors and S.A. Ghabrial, 1992. Soybean mosaic potyvirus enhances the titer of two comoviruses in dually infected soybean plants. Phytopathology, 82: 1022-1027.
CrossRefDirect Link - Bourdin, D. and H. Lecoq, 1994. Increase in cucurbit aphid-borne yellow virus concentration by co-infection with sap-transmissible viruses does not increase its aphid transmissibility. Phytopathology, 141: 143-152.
CrossRefDirect Link - Calvert, L.A. and S.A. Ghabrial, 1983. Enhancement by soybean mosaic virus of bean mottle virus titer in doubly infected soybean. Phytopathology, 73: 992-997.
Direct Link - Clark, M.F. and A.N. Adams, 1977. Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol., 34: 475-483.
CrossRefPubMedDirect Link - Damirdagh, I.S. and A.F. Ross, 1967. A marked synergistic interaction of potato viruses X and Y in inoculated leaves of tobacco. Virology, 31: 296-307.
PubMedDirect Link - Falk, B.W. and G. Bruening, 1994. Will transgenic crops generate new viruses and new diseases. Science, 263: 1395-1396.
PubMedDirect Link - Goldberg, K.B. and M.K. Barakke, 1987. Concentration of maize chlorotic mosaic virus increased in mixed infections with maize dwarf mosaic virus strain. Phytopathology, 77: 162-167.
Direct Link - Grafton-Cardwell, E.E., R.F. Smith, J. Valencia and C.A. Farrar, 1996. Occurrence of mosaic viruses in melons in the central valley of California. Plant Dis., 80: 1092-1097.
Direct Link - Hoffmann, K., S.M. Geske and J.W. Moryer, 1998. Pathogenesis of tomato spotted wilt virus in peanut plants dually infected with peanut mottle virus. Plant Dis., 82: 610-614.
Direct Link - Karyeija, R.F., J.F. Kreuze, R.W. Gibson and J.P.T. Valkonen, 2000. Synergistic interactions of a potyvirus and a phloem-limited crinivirus in sweet potato plants. Virology, 269: 26-36.
CrossRefDirect Link - Latham, L.J. and R.A.C. Jones, 2001. Incidence of virus infection in experimental plots, commercial crops and stocks of cool season crop legumes. Aust. J. Agric. Res., 52: 414-424.
CrossRefDirect Link - Lisa, V., G. Baccardo, G. D'Agostino, G. Dellavalle and M. d'Aquilio, 1981. Characterization of a potyvirus that causes Zucchini Yellow Mosaic. Phytopathology, 71: 667-672.
CrossRefDirect Link - Luis-Arteage, M., J.M. Alvarez, J.L. Alonso-Prados, J.J. Bermal, F. Garcia-Arenal, A. Lavina and E. Moriones, 1998. Occurrence, distribution and relative incidence of mosaic viruses infecting field-grown melon in Spain. Plant Dis., 82: 979-987.
CrossRefDirect Link - Poolpol, P. and T. Inouye, 1986. Enhancement of cucumber mosaic virus multiplication by zucchini yellow mosaic virus in doubly infected cucumber plants. Ann. Phytopathol. Soc. Japan, 52: 22-30.
Direct Link - Pruss, G., X. Ge, X.M. Shi, J.C. Carrington and V.B. Vance, 1997. Plant viral synergism: The potyviral genome encodes a broad-range pathogenicity enhancer that transactivates replication of heterologous viruses. Plant Cell, 9: 859-868.
PubMedDirect Link - Rochow, W.F. and A.F. Ross, 1955. Virus multiplication in plants doubly infected by potato viruses X and Y. Virology, 1: 10-27.
PubMedDirect Link - Rochow, W.F., 1972. The role of mixed infections in the transmission of plant viruses by aphids. Ann. Rev. Phytopathol., 10: 101-125.
CrossRefDirect Link - Sano, Y. and M. Kojima, 1989. Increase in cucumber mosaic virus concentration in Japanese radish plants co-infected with turnip mosaic virus. Ann. Phytopathol. Soc. Japan, 55: 296-302.
Direct Link - Vance, V.B., 1991. Replication of potato virus X RNA is altered in co-infections with potato virus Y. Virology, 182: 486-494.
PubMedDirect Link - Vance, V.B., P.H. Berger, J.C. Carrington, A.G. Hunt and X.M. Shi, 1995. Proximal potyviral sequences mediate potato virus X/potyviral synergistic disease in transgenic tobacco. Virology, 206: 583-590.
PubMedDirect Link - Wang, Y., V. Gaba, J. Yang, P. Palukaitis and A. Gal-On, 2002. Characterization of synergy between cucumber mosaic virus and potyviruses in cucurbit hosts. Virology, 92: 51-58.
Direct Link








