ABSTRACT
The impact of the combined stress factors of heavy metals (cadmium and nickel) and UVA+B radiation in Pisum sativum was investigated. Seeds were grown in sandy loam for 30 days and then randomly divided into equal two groups. The first group was exposed for 5 h day-1 to supplemental 6.4 KJ. m-2.day-1 dose UVA+B radiation beside white light (1100 Lux), while the second one was exposed to white light only giving light intensity (1100 Lux). Plants from each group irrigated with nutrient solution having 0.0, 5, 10 and 20 μmol Cd or Ni for two weeks and then harvested. The results showed that, Cd or Ni treatments induced significant decreased the length, fresh weight, dry weight and density of both root and shoot. UVA+B resulted in synergistic inhibitory effects on all measured growth parameters. Total chlorophyll, carotenoid, total carbohydrates and proteins showed a similar pattern to that of growth parameters. The amount of UV-screening pigments and anthocyanin was highest under the supplemental UVA+B radiation, while the combination of Cd or Ni and UVA+B decreased them. From this result, it showed that the combined treatment of UVA+B and Cd or Ni showed an antagonistic effects on UV-screening pigments. The combined stress of UVA+B radiation and Cd or Ni also resulted in a reduction of peroxidase and catalase activity, while increasing of H2O2 level in Pisum sativun plant. The data showed that in some measured parameters was a UVA+B and Cd or Ni synergistic effect, while with other parameters an antagonistic action was observed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2007.139.154
URL: https://scialert.net/abstract/?doi=ajpp.2007.139.154
INTRODUCTION
Solar radiation is one of the most essential factors for life on Earth. The solar radiation is not only beneficial, but may also be harmful, especially in the ultraviolet (UV) region. Most of the harmful UV radiation is absorbed by a protective layer in the atmosphere. This important layer, the ozone layer, has decreased during the last decades due to man-made air pollution. The concentration of ozone has decreased mainly due to the release of anthropogenic pollutants such as chlorofluorocarbons (Pyle, 1996). Consequently, a larger proportion of the UV-B spectrum reaches the Earth’s surface with serious implications for all living organisms (Xiong and Day, 2001; Caldwell et al., 2003). Not only is the air polluted, but also the soil; more and more heavy metals, e.g., cadmium (Cd) and nickel (Ni), are accumulating in the ground. Thus, enhanced UV radiation and heavy metal concentrations are new man-made stress factors to which the plants have to adapt anew. Elevated UV radiation has pleiotropic effects on plant development, morphology and physiology. The morphological consequences of UV supplemented white-light treatment include reduced growth, thickening of leaves and of cuticular wax layers. UV-B radiation reduced plant height and leaf area in sensitive species of cucumber (Tevini and Teramura, 1989) and needle length (Sullivan et al., 1996). Rice (Oryza sativa L.) showed a significant decrease in total biomass with increased UV-B radiation (Teramura et al., 1991). Moreover, Donald et al. (1997) noticed ’new red fire’ lettuce plants grown in the absence of solar UV-B or UV-A radiation had greater fresh and dry weight than those grown under ambient UV-B and UV-A and reduced the concentration of anthocyanin in the leaves which was reflected in a loss of red coloration. The height, biomass and leaves area of silver birch saplings tended to be reduced after long-term exposure to UV-B (Tegelberg et al., 2001; Tegelberg, 2002). Chlorophyll a and b contents of Phaseolus vulgaris leaves dropped after UV-B stress (Michaela et al., 2000). Under UV-B exclusion, chlorophyll content of Fagus sylvestris leaves was higher, the chlorophyll a/b ratio and the carotenoids content was lower than in leaves under the ambient level of UV-B radiation (Laposi et al., 2002). Anthocyanin accumulates in young, expanding foliage of various plant species in response to ultraviolet (UV) radiation exposure (Close and Beadle, 2003). Increased level of UV absorbing compounds, also protein damage was indicated by strong effect of photosynthetically active radiations PAR/UV light. Tegelberg (2002) noticed that symptoms of UV-B stress of three northern deciduous woody species included changes in the metabolism of carbohydrates and phenolic compounds. In a long term study, elevated UV-B radiations decrease significantly the ratio of storage starch to chloroplast area in field-grown silver birch leaves. There are many protective mechanisms that plants use to both tolerate and avoid UV-damage. The accumulation of UV-absorbing compounds can aid in avoidance of UV penetration to sensitive layers within the leaf. UV-screening compounds are thought to constitute phenyl-propanoid including flavones, flavonols, cinnamoyl esters and anthocyanin that provide a UV-A and UV-B screens (Cockell and Knowland, 1999).
A common plant response to different environmental stress (such as ozone, UV-light or extreme temperatures), is the accelerated generation or/and accumulation of reactive oxygen species, including hydrogen peroxide (H2O2), superoxide anion and hydroxyl radicals (Karpinski et al., 1999). Excess H2O2 can be converted to water and molecular oxygen spontaneously or by catalase, or detoxified through action of various peroxidases or ascorbate-glutathione pathway (Podila et al., 2001). Catalase is found mainly in peroxisomes and glyoxysomes which therefore play a significant role in defense against ozone stress (del Rio et al., 2002). Glutathione is a potent cellular reductant with a broad redox potential. It acts as a scavenger of peroxides and serves as a storage and transport form of reduced sulphur (May et al., 1998). Laakso et al. (2001) noticed UV-B treated Scots pines needles showed lower total glutathione concentrations after the vegetation. Glutathione content variations are thought to be an early protective response mechanism to UV-B radiation, before flavonoid shielding is achieved. Manninen et al. (2000) found that UV-B radiation induce cumulative oxidative stress in conifers. The annual variation in needle glutathione has been found to be much wider than corresponding ascorbate variation. This suggests that glutathione responds more sensitively to changes in ultraviolet radiations.
Cd is a highly toxic and persistent environmental poison for plants and animals (di Toppi and Gabbrielli, 1999). Cd interferes with many cellular functions mainly by complex formation with side groups of organic compounds such as proteins resulting in inhibition of essential activities. Although the mechanisms of cytoplasmic toxicity are identical in all organisms, different plant species and varieties show a wide range of plasticity in Cd tolerance, reaching from the high degree of sensitivity of most plants on the one hand to the hyperaccumulating phenotype of some tolerant higher plants on the other hand (McGrath et al., 2001). Cd induces genetic and biochemical changes in plant metabolisms that are related to general and Cd-specific stress responses (Blinda et al., 1997). Cadmium is a potent inhibitor in the biosynthesis of chlorophyll (Stobart et al., 1985). Nickel has been defined as essential micronutrient, because of its involvement in enzymatic activity in legumes (Welch, 1995). In most plants it is found at the level of 0.1-5 ppm (on a dry weight basis), with a wide threshold range of 40-246 ppm for toxicity symptoms, dependent upon the plant species (Kabata-Pendias and Pendias, 1992). The most common symptoms are chlorosis and inhibited photosynthesis and respiration (Foy et al., 1978). The mechanisms governing Ni2+ toxicity are not well understood. The dry matter of wheat was not affected by Ni treatment (Zeller and Feller, 1999). Nickel prevented any growth of red beet plant (Kukier and Chaney, 2001). The higher concentrations of heavy metal elements (Cd, Zn, Mn and Cu) absorbed would be toxic to tobacco plant and the amount of reducing sugars decreased (Wen, 1983). The activation of the cellular antioxidant metabolism belongs to the general stress responses induced by heavy metals (Dietz et al., 1999). Although an active antioxidative metabolism does not represent a Cd tolerance mechanism in a strict sense, it is beneficial for plant performance under heavy metal stress. Inadequate activities of antioxidant defense systems cause oxidative damage, lipid peroxidation and membrane leakage in plants exposed to Cu, Fe and also to Cd.
The objective of this work was to know how Pisum sativum plants respond to the combination of UV radiation and heavy metals. Do they have a protective mechanism against the increased UV radiation and is this mechanism affected by heavy metals?
MATERIALS AND METHODS
Plant Material and Plantation
The experimental plant used in this experiment was pure strain of Pisum sativum. Seeds were grown in plastic pots (15 cm) were filled with pre-sieved garden soil (sandy loam). About 6.0 cm deep, healthy Pisum sativum seeds were soaked. All pots were gently watered up to saturationand then kept in the open air and irrigated regularly every other day until treatments (for one month). Each pot contains four plants.
Cadmium, Nickel and Ultraviolet Treatments
After 30 days from seed soaking, the planted pots were irrigated every two days with one-fifth Long Ashton solution containing 0.0, 5, 10 and 20 μM Cd or Ni (added individually as chloride). The pots were randomly divided into two equal groups. One group from Cd and Ni treatments group exposed to white light only using fluorescent lamp OSRAM L. 18W/12 giving light intensity (1100 Lux), (0.0 μM Cd or Ni plants = Negative control) ; while the other was received UVA+B for 5 h day-1 (6.4 KJ. m-2.day-1 dose) beside white light (1100 Lux) (0.0 μM Cd or Ni plants = Positive control). Treatments were replicated with 4 pots.
UV A+B Setup
UV irradiation treatments were applied using OSRAM SYLVANIA ULTRAVIOLET 350 BL F40 lamp. Timers were set to automatically turn irradiation lamps on at midday time. Radiation doses and radiation power emitted were calculated according distance between lamp axis and Pisum sativum as presented in the lamp instruction manual (Gilbert, 1996). Distance between lamp and upper leaves of Pisum sativum were set to 40 cm and were periodically monitored and reset as Pisum sativum grows. Plant pots were kept in a growth chamber (1x1 m) around 27±2°C, 60-70% relative humidity, (14:10) light: dark periods and irrigated regularly for two weeks. An electric fan was operated during the light period to avoid the increase in temperature and to keep the growth chamber’s temperature around 27±2°C. Then, the harvested plants were prepared to the different measurements.
Growth Parameters
Length, fresh weight, dry weight and density of shoot and root were measured.
Estimation of Shoot and Root Density
According to Arduini et al. (1994) roots and shoots were separated and oven dried at 70°C until constant weight was obtained. The root and shoot density was estimated by dividing the average dry weight by the average root length (weight/unit length) measured at the end of treatment.
Estimation of Chlorophyll and Carotenoids Content
Total chlorophyll and carotenoid were estimated in the fresh plant leaves according to the procedure of Lichtenthaler (1987).
Anthocyanin Extraction and Quantification
Fresh l eaves tissue was extracted in 3 mL of acidified (1% HCl, v/v) methanol for 24 h at °C. 2 mL of the extract, 1.5 mL of water and 2.5 mL of chloroform were added and centrifuged for 20 min at 4°C. The anthocyanin content in the supernatant was estimated by measuring absorbance at 535 nm (Adamse, 1988).
Total Carbohydrates
The total available carbohydrate content were extracted according to Smith et al. (1964) and estimated colorimetrically by the Phenol-Sulphuric acid method as described by Dubois et al. (1951).
Total Protein Content
Total protein content was extracted with trichloroacetic acid and NaOH and estimated spectrophotometrically according to Lawery et al. (1951).
Determination of UV-absorbing Pigments
The extraction was done in methanol: water: HCl (79:20:1 by vol.) according to (Flower-Ellis and Olsson, 1993).
Assay of H2O2
H2O2 concentration in the incubation medium of treated leaf explants was measured by the FOX method (Jiang et al., 1990; Wolff, 1994) based on the peroxide-mediated oxidation of Fe2+ with xylenol orange.
Antioxidant Enzymes
Enzymes extracts were prepared by homogenizing Pisum sativum plant in a prechilled morter in 20 mL chilled extraction buffer (pH 7.5). Extracts were then centrifuged at 6000 rpm for 20 min at 5°C. Enzyme assays were conducted immediately following extraction.
Determination of Catalase Activity
Catalase activity was assayed in a reaction mixture containing 100 mM potassium phosphate (pH 6.5), 1.0 mM EDTA, 60.0 mM H2O2 and approximately 30 μg extract protein in a method following Aebi (1983). Activity was determined by following the decomposition of H2O2 at 240 nm.
Determination of Peroxidase Activity
Peroxidase activity was determined by following the dehydrogenation of guaicol at 436 nm (Malik and Singh, 1980).
Determination of Glutathione (GSH) Content
Glutathione content was determined spectrophotometrically following the method described by Griffith (1980).
Statistical Analysis
Analysis of variance (ANOVA), correlation tests and differences between control and treated plants (significance) were analyzed using Post hoc test on all data using SPSS program. The data considered significant at a level of p<0.05.
RESULTS
Growth Parameters
In comparison with the negative control (0.0 μM Cd or Ni), it was found that Cd and Ni significantly affected the growth parameters of Pisum sativum such as length and fresh weight of shoot and root (Table 1), dry weight and density of both root and shoot (Table 2) and show a remarkable reduction in all of these parameters of the negative control value. The degree of reduction in all previous parameters was greater in plants irradiated with UVA+B.
Total Chlorophyll and Carotenoids
Total chlorophyll content of Pisum sativum plant decreased by 27.23 and 40% in leaves of plants receiving 20 μM Cd and Ni, respectively to negative control (Fig. 1A). It was noticed that, supplementation of UVA+B reduced total chlorophyll. The percentage of reduction was 46.58 and 50.48% in plants irrigated with 20 μM Cd and Ni and received UVA+B radiation, respectively compared to negative control.
| Table 1: | Shoot length (cm), root length (cm), shoot fresh weight (g) and root fresh weight (g) of Pisum sativum plant (45 day old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and irrigated with UVA+B (6.4 KJ.m-2.day-1 dose) radiation |
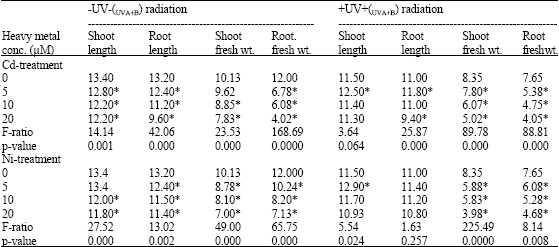 | |
| *Plants were harvested 15 days post-heavy metals and UV treatments. Each values is a mean of three replicates (n = 3). Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability | |
| Table 2: | Shoot dry weight (g), root dry weight (g), shoot density and root density (g/cm) of Pisum sativum plant (45 days old) irrigated having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with (6.4 KJ.m-2.day-1 dose) radiation |
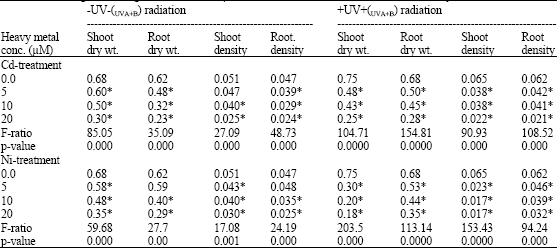 | |
| *Plants were harvested 15 days post-heavy metals and UV treatments. Each values is a mean of three replicates (n = 3). Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability | |
The results showed that, the combined treatment of UV and Cd or Ni showed a synergistic reaction of the two stress factors. Carotenoids showed a similar pattern to that of the total chlorophyll in that there was a significant reduction of this pigment with all Cd and Ni concentration (Fig. 1B). This reduction in carotenoids was increased after exposure to UV radiation.
Total Carbohydrates
It was noticed that from the results illustrated in Fig. 2, exposure of Pisum sativum seedlings to Cd (F = 75.40, p = 0.000) and Ni (F = 255.07, p = 0.000) decrease significantly the total carbohydrates content. The percentage reduction being 37.15 and 49.6% in plants irrigated with 20 μM Cd and Ni, respectively and not irradiated with UVA+B compared to negative control. Enhanced UVA+B radiation resulted in a significant reduction of total carbohydrates. The highest reduction in total carbohydrates was in plants irrigated with 20 μM Cd and Ni and irradiated with UVA+B. The percentage of reduction was 52.45 and 61.63% in seedlings exposed to UV and 20 μM Cd and ni, respectively compared to negative control. These results show that, the combined treatment of UVA+B and Cd or Ni caused synergistic reaction of the two stress factors.
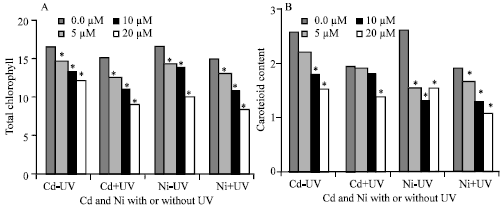 | |
| Fig. 1: | Total chlorophylls(A) and carotenoids content (B) (μg g-1 fresh wt.) of Pisum sativum plant (45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Each value is a mean of three replicates (n = 3). Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
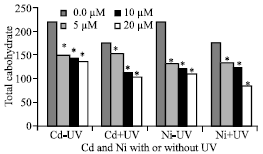 | |
| Fig. 2: | Total carbohydrate expressed as mg 100 g-1 dry weight of Pisum sativum plant. (45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
Total Protein
Total protein content was significantly negative correlated with increasing Cd (r = -0.931**, p = 0.000) or Ni (r = -0.892**, p = 0.000) concentrations (Fig. 3). The protein content was significantly reduced after supplemental of UVA+B. The highest reduction in total protein was in plants irrigated with Ni and irradiated with UVA+B.
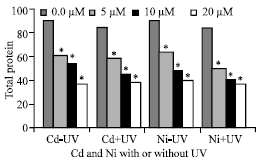 | |
| Fig. 3: | Total protein expressed as mg 100 g-1 fresh weight of Pisum sativum plant.(45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
Anthocyanin
The results showed in Fig. 4 showed, Cd (r = +0.936**, p = 0.000) and Ni (r = +0.954**, p = 0.000) induced anthocyanin production. It was also found that, UVA+B irradiated plants had more anthocyanin than other plants not exposed to UVA+B radiation, which may indicate a greater stress.
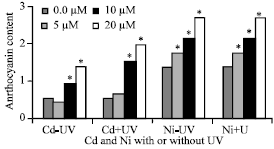 | |
| Fig. 4: | Anthocyanin content expressed as anthocyanin/g of Pisum sativum plant.(45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test). |
UV-absorbing Compounds
UVA+B radiation significantly increased these pigments. The production of UV-absorbing compounds was highest under the supplemental UV-radiation, while Cd and Ni decreased them. The combined treatment of UV and Cd or Ni showed an antagonistic reaction of the two stress factors. (Fig. 5) Also, it was noticed that, high concentrations of Cd and Ni decreasing the level of UV-absorbing compounds.
H2O2 Level
In general, H2O2 level in Pisum sativum leaves was positive correlated with increasing Cd (r = +0.815**, p = 0.000) or Ni (r = +0.867**, p = 0.000) concentration. These results are true for both plants exposed or not exposed to UVA+B radiation (Fig. 6). But, the production of H2O2 was higher in plants irradiated with UVA+B. Also, H2O2 level was higher in plants irradiated with UVA+B and treated with Ni (r = 0.909**, p = 0.000) than those treated with Cd (r = 0.688**, p = 0.000). It was also noticed that, the increasing in H2O2 reached its highest value at 20 μM Ni.
Catalase and Peroxidase Activity
The results showed in Fig. 7 showed that, the activity of catalase and peroxidase was depressed with increasing Cd and Ni concentration compared with negative control plants. The activity of these two enzymes was reduced under the supplemental UVA+B radiation. It was shown that, the reduction in catalase activity was higher in plants irrigated with Ni than Cd at all concentrations used. Exposure to UVA+B radiation and 20 μM Cd and Ni caused a decrease in catalase activity by 56.8 and 70%, respectively compared with negative control.
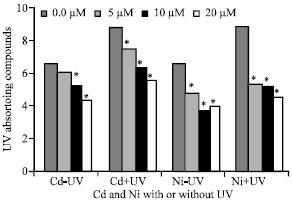 | |
| Fig. 5: | UV-absorbing compounds expressed as absorbance/g fresh weight by measuring total absorbance from A280 to A350 nm of Pisum sativum plant.(45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
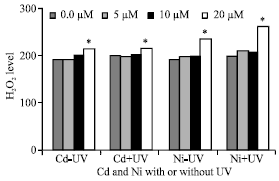 | |
| Fig. 6: | H2O2 level expressed as μM/g fresh weight of Pisum sativum plant. (45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
Glutathione Content
Glutathione content in Pisum sativum leaves was negative correlated with increasing Cd (r = -0.621*, p = 0.031) or Ni (r = -0.371, p = 0.236) concentrations compared with control (Fig. 8).
This reduction in GSH was increased after supplemental of UVA+B and exposure to Cd (r = -0.329, p = 0.310) and Ni (r = -0.600*, p = 0.039). The results showed that, glutathione activity did not show statistically significant changes with Cd or Ni concentrations.
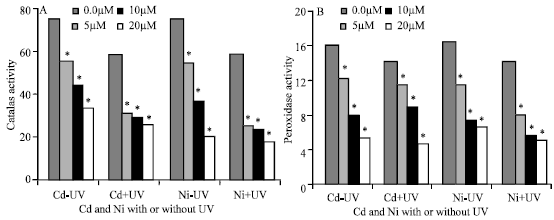 | |
| Fig. 7: | Catalase (A) and peroxidase activity (B) expresed as unit/g fresh weight of Pisum sativum plant. (45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. Astrickes indicate significant differences among Cd or Ni concentrations with or without UVA+B radiation at 0.05 level of probability (Post hoc test) |
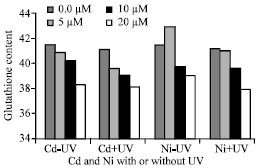 | |
| Fig. 8: | Effect of different Cd and Ni concentrations (μM) and UVA+B radiation on glutathione level expressed as μg g-1 fresh weight of Pisum sativum plant. Astrickes indicate significant differences among Cd or Ni concentrations with or without (45 days old) irrigated with nutrient solution having 0.0, 5, 10 and 20 μM Cd or Ni and Irradiated with UVA+B (6.4 KJ.m-2.d-1 dose) radiation. Plants were harvested 15 days post-heavy metals and UV treatments. UVA+B radiation at 0.05 level of probability |
DISCUSSION
Root length and elongation are essential for plants exploring for water and mineral nutrients. A decrease in root elongation due to heavy metals in nutrient solution has been reported for Cd in Fagus sylvatica seedlings (Breckel, 1991). The results presented show that a supply of 5, 10 and 20 μM Cd and Ni separately depressed the shoot and root length of Pisum sativum plant (Table 1). The same trend also obtained in fresh, dry weight, root and shoot density. The decrease of plant growth with increasing Cd concentration in the nutrient solution can be due to a direct toxic effect on plant metabolism or to an indirect effect of Cd by lowering availability of nutritional elements (Foy et al., 1978). In roots and leaves of pea plants Cd produced a significant inhibition of growth as well as a reduction in the transpiration and photosynthesis rate, chlorophyll content of leaves and an alteration in the nutrient status in both roots and leaves (Sandalio et al., 2001). After irradiated with UV, the percentage of inhibition of all morphological parameters measured was increased (Table 1 and 2). Vascular plants exposed to UV-B levels led to appreciable reductions in biomass production and leaf area. There was a significant reduction in biomass and plant height of various plant species with increasing UV-B radiation (Donald, 2004). A reduction in whole-plant photosynthetic surface area might ultimately explain the reductions in growth and biomass reductions (Xiong and Day, 2001). Reduced growth may result from direct photochemical damage to key macromolecules such as proteins and nucleic acids, or as an indirect consequence of the increased production of reactive oxygen species in plants exposed to UV-B (Jansen et al., 1998).
Cd and Ni decreased the content of both total chlorophylls and carotenoids (Fig. 1A and B). Cd exposure significantly decreased total chlorophyll and carotenoids in Brassica napus and pea plant (Larsson et al., 1998; Sandalio et al., 2001). Dubé and Bornman (1992) also observed a decrease in chlorophyll content in spruce when Cd was present. However, Cd can induce oxidative stress indirectly by producing disturbances in chloroplasts. Thus, Cd produces degradation of chlorophyll and carotenoids as well as an inhibition of their biosynthesis (Bazzaz et al., 1992) which can produce disturbances in the electron transport rates of PSI and PSII leading to the generation of oxygen free radicals. According to Stobart et al. (1985) the decrease in chlorophyll is due to inhibition of protochlorophyllide reduction and inhibition of aminolevulinic acid synthesis. Kupper et al. (1996) reported that substitution of the central atom of chlorophyll, magnesium, by heavy metals is an important damage mechanism in stressed plants. This substitution prevents photosynthetic light-harvesting in the effected chlorophyll molecules, resulting in a breakdown of photosynthesis. In the present study, enhanced UVA+B resulted in a significant reduction in total chlorophyll and carotenoid was increased (Fig. 1A and B). It is often assumed that UV-B radiation may affect the photosynthetic pigments, either because of inhibition of their synthesis or increased destruction. In this connection, Strid (1993) suggested that UV-B radiation has no specific effects on the enzymes of the chlorophyll biosynthetic pathway but, rather, influences the genetic regulation of chlorophyll binding protein, leading to destruction of chlorophyll. It has been reported that UV-B radiation resulted in greater reduction in the amount of Chl.b as opposed to Chl.a and may point a more selective destruction of Chl-b biosynthesis or degradation of precursors (Marwood and Greeberg, 1996). In addition, Ultraviolet-B radiations have an indirect damaging effect on plant cells. UV-B radiation may cause oxidative damage in chlorophylls and polyunsaturated lipids by increasing the formation of free radicals and peroxides (Jordan, 1996). UVA+B influence on the amount of carotenoids, where it was decreased with UV radiation (Fig. 1B). Carotenoids have been shown to increase (Middleton and Teramura, 1993) or decrease (Tevini and Teramura, 1989) depending on species, growth conditions, etc. Furthermore, since carotenoids are reputed to protect photosynthetic membranes against internally and externally generated photooxidative products, a reduction in carotenoids could thus lead to a reduction in the photosynthetic process.
In the present study, the results showed that, Cd and Ni decreased total carbohydrate in Pisum sativum plant (Fig. 2). In sunflower, Pankovi et al. (2000) reported on decreased photosynthetic rates at high Cd concentrations. Cd produced a significant reduction in photosynthesis rate in pea plant (Sandalio et al., 2001).They stated photosynthesis is sensitive to disturbances in gas exchange through the stomata, Cd stimulates the stomata to close and thus CO2 assimilation will be decreased. Some enzymes of photosynthetic carbon reduction cycle are sensitive to Cd and could also be responsible for the productivity decrease (De Filipps and Ziegler, 1993). UV radiation induced a highly significant reduction in total carbohydrate contents in Pisum sativum plant irrigated with Cd or Ni (Fig. 2). The reduction in carbohydrate contents in response to UV radiation could be attributed to the destructive damage of photosystems induced by UV radiation which led to the decrease in photosynthesis efficiency. It is evident that UV-B can potentially impair the performance of all three main component processes of photosynthesis, the photophosphorylation reactions of the thylakoid membrane, the CO2-fixation reactions of the Calvin cycle and stomatal control of CO2 supply (Allen et al., 1998).
By increasing Cd or Ni concentrations in irrigation solution led to decreased in total protein content in Pisum sativum plant (Fig. 3). Sasadhar (1987) found that a decrease in protein and chlorophyll content of Cascuta reflexa irrigated with 5 and 10 μg mL-1 lead. He suggested that the chlorophyll synthesizing system and chlorophyllase activity significantly affected by lead. The decrease in protein content revealed that soluble protein may have leaked or diffused out of the plant material, or possibly the catabolic enzymes were induced and destroyed the proteins. After irradiated with UVA+B, the total protein content was decreased in Pisum sativum plant treated with two heavy metals (Fig. 3). In general, UV radiation damages lipids, nucleic acids and proteins in leaves of higher plants (Jordan, 1996; Vass, 1997).
Cd and Ni induced anthocyanin production. But, UVA+B irradiated plants had more anthocyanin (Fig. 4). UV-B-induced anthocyanin pigmentation in rice seedlings may have a role in minimizing the UV-B damage (Sharma, 2001). Moreover, the irradiation of seedlings with artificial UV-B light also elicits strong induction of anthocyanin similar to the sunlight (Singh et al., 1999). The accumulation of anthocyanin may be of advantage to plants as anthocyanin can protect the growing meristematic zones from likely genetic damage caused by UV- radiation (Stapleton and Walbot, 1994). Several studies have indicated that in vitro anthocyanin could act as effective antioxidants (Sarma et al., 1997). It is known that anthocyanin can make complexes with other molecules- copigment (Brouillard, 1983) and such complexation can protect partner compounds against oxidative damages.
The amount of UV-absorbing compounds increased after UV-radiation (Fig. 5). Accumulation of the UV-B-absorbing pigments is one of the ways by which plants protect them from the harmful effect of UV radiation (Caldwell et al., 1983). Protective responses are stimulated mainly by UV-B radiation, including increased production of UV-B-absorbing compounds (e.g. flavonoids), secondary compounds ubiquitous in higher plants (Tevini et al., 1991). Flavonoids are thought to protect photosynthetic tissues by acting as screening pigments, absorbing UV-B radiation (Middleton and Teramura, 1993). (UV-absorbing compounds in Pisum sativum plant were decreased when Cd and Ni was present. Charles and Caldwell (2001) found that, copper altered the levels of UV-absorbing compounds of cucumber leaf and cotyledon tissues. Cadmium decreased the level of UV-absorbing compounds of Brassica napus when exposed to UV-B radiation (Larsson et al., 1998). According to Larsson et al. (1998), the decrease in UV-absorbing compounds is due to the synthesis of phytochelatins (induced by heavy metals). Glutathione (GSH) is thought to act as a signal transductor of the UV-B stimuli in the induction of UV-absorbing compounds. When Kalbin et al. (1997) exposed pea plants to UV-B radiation the content of GSH increased 4.5-fold. They also observed that UV-B stimulated mRNA encoding for chalcone synthase and phenylalanine ammonia lyase, which are key enzymes in UV-absorbing compounds synthesis. GSH is also a precursor of phytochelatine, induced by Cd and thus possible competition for GSH may lead to a decreased induction of UV-absorbing compounds or a reduced ability to chelate cadmium ions.
In the present study, the results showed that, H2O2 level in Pisum sativum leaves was increased with increasing Cd and Ni concentration especially at 20 μM in comparison with control plants (Fig. 6). These results are in agreement with Cho and Kim (2003) and Chen et al. (2000), they found that, H2O2 content increased in tomato seedlings and rice plant exposure to Cd and Cu. Heavy metals are known to be involved in many ways in the production of reactive oxygen species (ROS) (Halliwell and Gutterridge, 1984) and the H2O2 accumulation caused by Cd exposure may occur in a manner similar to that in plants stressed (Prasad et al., 1994). Sandalio et al. (2001) concluded that, accumulation of H2O2 is a result of the inhibition of antioxidative enzymes catalase and peroxidase. The production of H2O2 in Pisum sativum plant was higher in plants irradiated with UV than exposed to white light (Fig. 6). The level of oxygen free radicals in cells can also be enhanced by a decrease of the enzymatic antioxidants involved in their detoxification such as superoxide dismutase, catalase or peroxidase (Sandalio et al., 2001). Catalase and peroxidase activity of Pisum sativum plant was reduced by increasing concentrations of Cd and Ni in the nutrient solutions (Fig. 7A and B). The decline of catalase activity has been associated with Cd toxicity in pea plant (Sandalio et al., 2001), Phaseolus vulgaris (Chaoui et al., 1997) and Helianthus annuus (Gallego et al., 1996). The reduction of catalase activity was due to a decrease in the protein content (Sandalio et al., 2001). Cd uptake inhibited the system involved in H2O2 removal (glutathione/glutathione reductase, catalase and ascorbate peroxidase in scots pine seedlings (Schützendübel et al., 2001). This causes an accumulation of H2O2 and induces the synthesis of ascorbate and glutathione. Analysis of catalase and peroxidase in the present study revealed that elevated UV radiation induced a significant reduction in all the previous parameters (Fig. 7).
Glutathione in Pisum sativum plant was decreased when Cd and Ni was present (Fig. 8). Schützendübel et al. (2001) found that, Cd inhibited the systems involved in H2O2 removal (glutathione, catalase and ascorbate peroxidase) and caused H2O2 accumulation in Scots pine seedlings. The depletion of glutathione in Pisum sativum plant irrigated with Cd or Ni caused by an increased consumption of glutathione for phytochelatine production (Xiang and Oliver, 1998). Because the synthesis of glutathione is demand driven, the low glutathione concentration might have triggered increased sulfur uptake and its own synthesis (May et al., 1998). Gallego et al. (1996) concluded that, the depletion of GSH, by the formation of Cd-phytochelatine complex, could also contribute to oxidative stress by depressing the antioxidative response. Another reason for an overall reduction in the endogenous level of GSH might be due to its utilization as a reducing substrate in the synthesis of ascorbate. GSH is also consumed and degraded in order to protect cellular membranes from lipid peroxidation. The level of GSH is reduced during chilling-induced process of photoxidation (Wise and Naylor, 1987) that also involves lipid peroxidation.
In conclusion, Cd and Ni decreased growth of Pisum sativum plant. After exposure to UVA+B radiation, this reduction was future increased. The protective mechanisms in plants irradiated with UVA+B were reduced after exposure to Cd or Ni. The production of H2O2 was higher in plants irradiated with UV then those expose to white light, while catalase and peroxidase activity was lower.
REFERENCES
- Allen, D.J., S. Nogues and N.R. Baker, 1998. Ozone depletion and increased UV-B radiation: Is there a real threat to photosynthesis. J. Exp. Bot., 49: 1775-1788.
Direct Link - Arduini, I., D.L. Godbold and A. Onnis, 1994. Cadmium and copper change root growth and morphology of Pinus pinea and Pinus pinaster seedlings. Physiol. Plant., 92: 675-680.
CrossRefDirect Link - Bazzaz, F.A., G.L. Rolfe and R.W. Carlson, 1992. Effect of Cd on photosynthesis and transpiration of excised leaves of corn and sunflower. Physiol. Plant, 32: 373-377.
CrossRefDirect Link - Blinda, A., B. Koch, S. Ramanjulu and K.J. Dietz, 1997. De novo synthesis and accumulation of apoplast proteins in leaves of heavy metal exposed barley seedlings. Plant Cell Environ., 20: 969-981.
Direct Link - Brouillard, R., 1983. The in vivo expression of anthocyanin colour in plants. Phytochemistry, 22: 1311-1323.
CrossRefDirect Link - Caldwell, M.M., W.G. Gold, G. Harris and C.W. Ashurst, 1983. A modulated lamp system for solar UV-B (280-320 nm). supplementation studies in the field. Photochem. Photobiol., 37: 479-485.
CrossRefDirect Link - Caldwell, M.M., C.L. Ballare, J.F. Bornman, S.D. Flint and L.O. Bjorn et al., 2003. Terrestrial ecosystems, increased solar ultraviolet radiation and interactions with other climatic change factors. Photochem. Photobiol. Sci., 2: 29-38.
Direct Link - Chaoui, A., S. Mazhoudi, M. Ghorbal and E. El-Fergani, 1997. Cadmium and zinc induction of lipid peroxidation and effects on antioxidant enzymes activities in bean (Phaseolus vulgaris L.). Plant Sci., 127: 139-147.
Direct Link - Charles, R. and A. Caldwell, 2001. Effect of elevated copper on the ultraviolet light-absorbing compounds of cucumber cotyledon and leaf tissues. J. Plant Nutr., 24: 283-285.
Direct Link - Chen, L.M., C.C. Lin and C.H. Kao, 2000. Copper toxicity in rice seedlings: Changes in antioxidative enzyme activities, H2O2 level and cell wall peroxidase activity in roots. Bot. Bull. Acad. Sin., 41: 99-103.
Direct Link - Cho, U. and I. Kim, 2003. Effect of cadmium on oxidative stress and activities of antioxidant enzymes in tomato seedlings. Korean J. Ecol., 26: 115-121.
CrossRefDirect Link - Close, D.C. and C.L. Beadle, 2003. The Ecophysiology of foliar anthocyanin. Botanical Rev., 69: 149-161.
CrossRefDirect Link - Cockell, C.S. and J. Knowland, 1999. Ultraviolet radiation screening compounds. cambridge philosophical society. Biol. Rev., 74: 311-345.
Direct Link - De Filippis, L. and H. Ziegler, 1993. Effect of sublethal concentrations of zinc, cadmium and mercury on the photosynthetic carbon reduction cycle of Euglena. J. Plant Physiol., 142: 167-172.
CrossRefDirect Link - Del Rio, L.A., J. Corpas, L.M. Sandalio, J.M. Palma, M. Gomez and J.B. Barroso, 2002. Reactive oxygen species, antioxidant systems and nitric oxide in peroxisomes. J. Exp. Bot., 53: 1255-1272.
CrossRefDirect Link - Di Toppi, L.S. and R. Gabbrielli, 1999. Response to cadmium in higher plants. Environ. Exp. Bot., 41: 105-130.
Direct Link - Dietz, K.J., U. Kramer and M. Baier, 1999. Free Radicals and Reactive Oxygen Species as Mediators of Heavy Metal Toxicity. In: Heavy Metal Stress in Plants: From Molecules to Ecosystems, Prasad, M.N.V. and J. Hagemeyer (Eds.). Springer-Verlag, Berlin, pp: 73-97.
Direct Link - Donald, K.T., 2004. Influence of PAR and UV-A in determining plant sensitivity and photomorphogenic responses to UV-B radiation. Photochem. Photobiol., 79: 307-315.
Direct Link - Dube, S. and J. Bornman, 1992. Response of spruce seedlings to simultaneous exposure to UV-B radiation and cadmium. Plant Physiol. Biochem., 30: 761-767.
Direct Link - Dubois, M., K. Gilles, J.K. Hamilton, P.A. Rebers and F. Smith, 1951. A colorimetric method for the determination of sugars. Nature, 168: 167-167.
CrossRefPubMedDirect Link - Foy, C.D., R.L. Chaney and M.C. White, 1978. The physiology of metal toxicity in plants. Ann. Rev. Plant Physiol., 29: 511-566.
CrossRefDirect Link - Gallego, S.M., M.P. Benavides and M.L. Tomaro, 1996. Effect of heavy metal ion excess on sunflower leaves: Evidence for involvement of oxidative stress. Plant Sci., 121: 151-159.
CrossRefDirect Link - Griffith, O.W. and A. Meister, 1979. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (s-n butyl homocysteine sulfoximine). J. Biol. Chem., 254: 7558-7560.
PubMedDirect Link - Halliwell, B. and J.M. Gutteridge, 1984. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem. J., 219: 1-14.
PubMed - Jansen, M.A.K., V. Gaba and B.M. Greenberg, 1998. Higher plants and UV-B radiation: balancing damage, repair and acclimation. Trends Plant Sci., 3: 131-135.
CrossRefDirect Link - Jiang, Z.Y., A.C.S. Woollard and S.P. Wolff, 1990. Hydrogen peroxide production during experimental protein glycation. FEBS Lett., 268: 69-71.
Direct Link - Kabata-Pendias, A. and H. Pendias, 1992. Trace Elements in Soils ands Plants. 2nd Edn., CRC Press, Boca Raton, Fla, ISBN-10: 0849366437.
Direct Link - Kalbin, G., A.B. Ohlsson, T. Berglund, J. Rydstrom and A. Strid, 1997. Ultraviolet-B-radiation-induced changes in nicotinamide and glutathione metabolism and gene expression in plants. Eur. J. Biochem., 249: 465-472.
CrossRefDirect Link - Karpinski, S., H. Reynolds, B. Karpinska, G. Wingsle, G. Creissen and P. Mullineaux, 1999. Systemic signaling and acclimation in response to excess excitation energy in Arabidopsis. Science, 284: 654-657.
CrossRefDirect Link - Kukier, U. and R. Chaney, 2001. Amelioration of nickel phytotoxicity in Muck and mineral soils. J. Environ. Qual., 30: 1949-1960.
Direct Link - Kupper, H., F. Kupper and M. Spiller, 1996. Environmental relevance of heavy metal-substituted chlorophylls using example of water plants. J. Exp. Bot., 47: 259-266.
Direct Link - Laakso, K., H. Kinnunen and S. Huttunen, 2001. The glutathione status of mature Scots pines during the third season of UV-B radiation exposure. Environ. Pollut., 111: 349-354.
CrossRef - Laposi, R., O. Veres Sz. Mile and I. Meszaros, 2002. Photosynthesis-ecophysiological properties of beech (Fagus sylvestris L.) under the exclusion of ambient UV-B radiation. Acta Biologica Szegediensis, 46: 243-245.
Direct Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Lichtenthaler, H.K., 1987. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol., 148: 350-382.
CrossRefDirect Link - Marwood, C.A. and B.M. Greenberg, 1996. Effect of supplementary UV-B radiation on chlorophyll synthesis and accumulation of photosystems during chloroplast development in Spirodela Oligorrhiza. Photochem. Photobiol., 64: 664-670.
Direct Link - May, M., T. Vernoux, C. Leaver, M. van Montagu and D. Inze, 1998. Glutathione homeostasis in plants: Implications for environmental sensing and plant development. J. Exp. Bot., 49: 649-667.
CrossRefDirect Link - McGrath, S., E. Lombi and F.J. Zhao, 2001. Whats new about cadmium hyperaccumulation? New Phytol., 149: 2-3.
Direct Link - Michaela, A., K. Norbert and N. George, 2000. Effect of cold and UV-B stress on scavenging systems of Phaseolus vulgaris leaves. Poster, Am. Society Plant Biol.
Direct Link - Middleton, E.M. and A.H. Teramura, 1993. The role of flavonol glycosides and carotenoids in protecting soybean from ultraviolet-B damage. Plant. Physiol., 103: 741-752.
CrossRefDirect Link - Pankovi, D., M. Plesniar, N. Petrovi and Z. Saka, 2000. Effects of nitrogen nutrition on photosynthesis in Cd-treated sunflower plants. Ann. Bot., 86: 841-847.
Direct Link - Podila, G., A. Paolacci and M. Badiani, 2001. The Impact of Greenhouse Gases on Antioxidants and Foliar Defence Compounds. In: The Impact of Carbon Dioxide and Other Greenhouse Gases on Forest Ecosystems, Karnosky, D.F., R. Ceulemans, G.E. Scarascia-Mugnozza and J.L. Innes (Eds.). CABI Publishing, Vienna, Austria, pp: 57-125.
- Prasad, T., M. Anderson, B. Martin and C. Stewart, 1994. Evidence for chilling induced oxidative stress in maize seedlings and a regulatory role for hydrogen peroxide. Plant Cell, 6: 65-74.
Direct Link - Sandalio, L.M., H.C. Dalurzo, M. Gomez, M.C. Romero-Puertas and L.A. del Rio, 2001. Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J. Exp. Bot., 52: 2115-2126.
Direct Link - Sarma, D., Y. Sreelakhsmi and R. Sharma, 1997. Antioxidant ability of anthocyanins against ascorbic acid oxidation. Phytochemestry, 45: 671-674.
CrossRef - Jana, S., T. Dalal and B. Barua, 1987. Effects and relative toxicity of heavy metals on Cascuta reflexa. Water Air Soil Pollut., 33: 23-27.
CrossRefDirect Link - Schutzendubel, A., P. Schwanz, T. Teichmann, K. Gross, R. Langenfeld-Heyser, D.L. Godbold and A. Polle, 2001. Cadmium-induced changes in antioxidative systems, hydrogen peroxide content and differentiation in scots pine roots. Plant Physiol., 127: 887-898.
CrossRefDirect Link - Singh, A., T. Seli and R. Sharma, 1999. Sunlight-induced anthocyanin pigmentation in maize vegetative tissues. J. Exp. Bot., 50: 1619-1625.
Direct Link - Smith, D., G.M. Poulsen and C.A. Roguse, 1964. Extraction of total available carbohydrates from grass and legume tissues. Plant Physiol., 39: 960-962.
Direct Link - Stapleton, A.E. and V. Walbot, 1994. Flavonoids can protect maize DNA from the induction of ultraviolet radiation damage. Plant Physiol., 105: 881-889.
Direct Link - Stobart, A.K., W.T. Griffiths, I. Ameen-Bukhari and R.P. Sherwood, 1985. The effect of Cd2+ on the biosynthesis of chlorophyll in leaves of barley. Physiol. Plant, 63: 293-298.
CrossRefDirect Link - Strid, A., 1993. Alteration in expression of defence genes in Pisum sativum after exposure to supplementary ultraviolet-B radiation. Plant Cell Environ., 34: 949-953.
Direct Link - Sullivan, J.H., B.W. Howells, C.T. Ruhlandand and T.A. Day, 1996. Changes in leaf expansion and epidermal screening effectiveness in Liquidambar sturaciflua and Pinus taeda in response to UV-B radiation. Physiol. Plant, 98: 349-357.
Direct Link - Tegelberg, R., R. Julkunen-Tiittoand and P.J. Aphalo, 2001. The effects of long-term elevated UV-B on the growth and phenolics of field-grown silver birch Betula pendula. Global Change Biol., 7: 839-848.
Direct Link - Teramura, A.H., L.H. Ziska and A.E. Sztein, 1991. Changes in growth and photosynthetic capacity of rice with increased UV-B radiation. Physiol. Plant., 83: 373-380.
CrossRefDirect Link - Tevini, M. and A.H. Teramura, 1989. UV-B effects on terrestrial plants. Photochem. Photobiol., 50: 479-487.
CrossRefDirect Link - Tevini, M., J. Braun and G. Fieser, 1991. The protective function of the epidermal layer of rye seedlings against ultraviolet-B radiation. Photochem. Photobiol., 53: 329-333.
CrossRefDirect Link - Welch, R.M. and L. Shuman, 1995. Micronutrient nutrition of plants. Crit. Rev. Plant Sci., 14: 49-82.
CrossRefDirect Link - Wise, R.R. and A.W. Naylor, 1987. Chilling-enhanced peroxidation: The peroxidative destruction of lipids during chilling injury to photosynthesis and ultrastructure. Plant Physiol., 83: 272-277.
Direct Link - Wolff, S.P., 1994. Ferrous ion oxidation in presence of ferric ion indicator xylenol orange for measurement of hydroperoxides. Methods Enzymol., 233: 182-189.
CrossRefDirect Link - Xiang, C. and D.J. Oliver, 1998. Glutathione metabolic genes coordinately respond to heavy metals and jasmonic acid in Arabidopsis. Plant Cell, 10: 1539-1550.
CrossRefPubMedDirect Link - Xiong, S.F. and A.T. Day, 2001. Effect of Solar Ultraviolet-B Radiation during springtime ozone depletion on photosynthesis and biomass production of antarctic vascular plants. Plant Physiol., 125: 738-751.
PubMedDirect Link - Zeller, S. and U. Feller, 1999. Long-distance transport of cobalt and nickel in maturing wheat. Eur. J. Agron., 10: 91-98.
Direct Link - Larsson, E., J. Borman and H. Asp, 1998. Influence of UV radiation and Cd2+ on chlorophyll florescence, growth and nutrient content in Brassica napus. J. Exp. Bot., 49: 1031-1039.
CrossRef








