ABSTRACT
A solution culture study was conducted to evaluate the effects of constant and variable salinity in spring wheat. In the variable salinity, plants were grown at 100 mol m-3 NaCl all the times after initial salt stress was introduced. In the variable salinity, a 12 days cycle was repeated. This involved changing the nutrient solution so that the plants were exposed to variable salinity but such that the average salinity experienced was 100 mol m-3 NaCl. Salinity significantly decreased number of tillers from 8 days after salt stress but leaf appearance stage was not significantly affected. Leaf extension growth was always higher at variable than at constant salinity in leaves 3, 4, 7 and the flag leaf but the difference was not always significant. In leaves 5 and 6 leaf extension growth was higher at constant than at variable salinity. Salinity significantly decreased leaf area, shoot and root dry weight per plant. These parameters were always higher at variable than at constant salinity but the difference was not significant.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.1112.1117
URL: https://scialert.net/abstract/?doi=ajps.2003.1112.1117
INTRODUCTION
Most salt tolerance data has been obtained from experiments where salinity was maintained essentially uniform throughout the root zone by irrigating the soil plot or sand culture with saline waters and using high leaching fractions (Maas and Hoffman, 1977). Except in controlled experiment conditions, soil salinity is seldom constant with time or uniform in space. In fact, salinisation often vary from concentration approximately that of the bottom of the root zone (Maas, 1986). Since soil salinity also increases between irrigations, as soil water is lost by evapotranspiration, plants must respond to a heterogeneous system, which is continuously changing. Solution culture is another method of propagation favoured by many workers to study the responses of plants to salt stress (for example, Kingsbury et al., 1984; Gorham et al., 1984; Rashid 1986; Shah 1987; Iqbal, 1988; Kemal-ur-Rahim, 1988; Wyn Jones and Gorham, 1989; Gorham, 1990). In this system the uniform application of salinity helps to minimize the problems encountered when interpreting results obtained from non-uniform salinity profiles in the fields. However, applying these data to field conditions where the distribution of salts is neither uniform in depth nor constant with time is difficult and requires knowledge of how plants respond to varying salinity (Maas and Hoffman, 1977). Previous experiments had shown that constant salinity decreases leaf extension rate (for example, Iqbal, 1988; Kemal-ur-Rahim, 1988). Therefore, this experiment was conducted to study the effect of constant and variable salinity on plant growth of spring wheat. In this experiment constant and variable salinity treatments had the same average salinity throughout but the variable salinity treatment had periods of low to high salinity. Therefore, this experiment would look at whether periods of low and high salinity have the same effect on leaf extension and growth as the constant salinity.
MATERIALS AND METHODS
Plant material and growth conditions: The experiment was initiated on 25 November and terminated on 18 January. The experiment was carried out in a growth room set at a temperature cycle of 24°C/16°C day and night and photoperiod of 16 h. Spring wheat variety ‘Wembley’ was used in this experiment. The seedlings received light from a bank of 125W fluorescent ‘warm white’ lights which provided between 200-300 ìmol m-2 s-1 photosynthetically active radiation (PAR) at initial plant level.
Experimental design and treatments: A randomized block design was used to minimize the effects of light reflection and air conditioning on the plants growing near the walls of the growth room. There were 4 blocks and 3 salinity treatments namely, control (0 mol m-3 NaCl), constant and variable salinity. In the constant salinity treatment plants were grown at 100 mol m-3 NaCl all the time after initial salt stress was introduced on 10 December. In the variable salinity treatment a 12 days cycle was repeated (Table 1).
| Table 1: | A twelve-day nutrient and salt solution cycle for variable salinity |
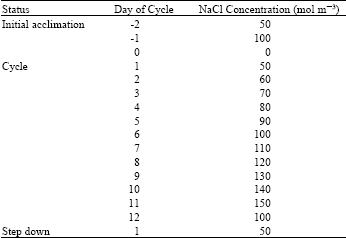 | |
This involved changing the nutrient solution so that the plants were exposed to variable salinity but such that the average salinity experienced was 100 mol m-3 NaCl. In both constant and variable salinity treatments, initial stress was imposed in 50 mol m-3 NaCl daily increments. At the start of the experiment the variable salinity treatment reached 100 mol m-3 NaCl on the second day of salt stress. On the next day (the first day of the cycle) salinity was decreased to 50 mol m-3 NaCl. On subsequent days it was increased in 10 mol m-3 NaCl daily increments, till it reached 150 mol m-3 NaCl on day 11 of the cycle. On day 12 it was decreased to 100 and then on day 13 back to 50 mol m-3 NaCl. The cycle was then repeated. The nutrient solutions in the control and constant salinity treatments were also changed on the day when the salinity was decreased to 100 mol m-3 NaCl in the variable salinity treatment (day 12 of the cycle). In constant and variable salinity treatments, NaCl was given in solution form from 4M stock solution, the necessary dilution factors being calculated.
Growth containers: Prior to seedling transplanting, the 10L plastic containers (Fig. 1) were panted black on the outside with bitminised paint to prevent light encouraging algal growth in the nutrient solution. The containers had a hole drilled in the side at the bottom, which was plugged with a 9 mm rubber bung. This was to facilitate easy changing of the nutrient solution. The containers were arranged along the sides of workbenches to facilitate easy access for maintenance and measurements. Each polystyrene lid was bored with 16 holes using a 9 mm heated cork borer. The holes were spaced to give a plant-to-plant distance of 50 mm and between row distances of 80 mm. This gave a planting density of approximately 250 plants m2. The polystyrene lids were also painted black on the upper surface and sides. Air to the containers was supplied via lengths of polyurethane tubing, cut into appropriate lengths and jointed together by 3-way air regulators.
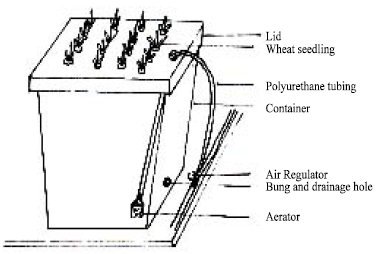 | |
| Fig. 1: | Growth container, showing aeration line, drainage hole and lid |
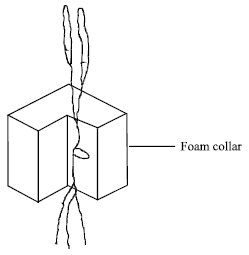 | |
| Fig. 2: | Expanded diagram of supportive foam collar around seedling and position of seed in collar |
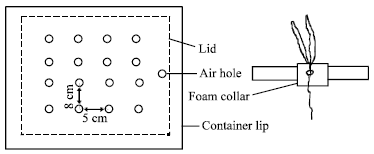 | |
| Fig. 3: | Polystyrene lid, showing dimensions of holes and spacing between them, plus sectional view showing foam collar seated in lid |
These regulators allowed the air to be restricted, to obtain uniform aeration between containers. A polyurethane tube led from each regulators into a container through a small 8 mm hole bored into the polystyrene lid and was terminated by an aerator. This uniform and fine bubbling facilitated oxygen absorption into the nutrient solution, to maintain healthy root growth. The whole network of tubing was tied onto the benches and connected to an air compressor, master pressure being regulated by a needle valve.
Germination and transplanting: Prior to seeding, approximately 500 seeds of wheat were soaked overnight in a muslin bag suspended under slow running tap water. Next morning the seeds were placed on capillary matting stretched over a plastic grid reinforced by wires and placed on top of plastic bowls (340 x 270 mm surface x130 mm deep). The bowls were filled with tap water and a supplement of 0.5 g L-1 of ‘Phostrogen’ (Phostrogen Ltd, Corwen, Clwyd, UK), a powdered plant food formulation was added to ensure an adequate nutrient supply to the germinating seeds. Wicks of capillary matting were always in contact with the water to ensure a continuously moistened newspaper for 2 days. When the radicles and coleoptiles were seen to be emerging, the newspapers were removed. On 7th December, at the 0+2 leaf appearance stage (0 fully emerged and 2 emerging leaves), seedlings of uniform height with well developed roots were teased out from the capillary matting, care being taken not to damage the roots which were important for ensuring immersion in the water. Each seedling was suspended in a collar of polyurethane foam (Fig. 2) and placed in an expanded polystyrene lid (Fig. 3). To ensure a uniform planting method, each plant was centered with its seed in the middle of the collar and the collar was pushed into the polystyrene so that the seed would be located approximately halfway to the depth of the lid. After the plats were inserted into the polystyrene lids and placed over the nutrient solution, they were allowed to adjust to their new environment for 3 days. If any plants were damaged in transplanting, it was usually noticeable after one day and they could be replaced.
Leaf extension growth: Leaf extension growth (LEG) of all leaves on the main stem was measured in mm with a ruler every day starting the day after initial salt stress. Leaf extension measurements started with leaf 3 because it was appearing at the time of initial salt stress. LEG measurements of leaf 3 were recorded from the ligule of leaf 1. LEG of subsequent leaves was recorded from the ligule of the leaf two positions down on the main stem.
Leaf appearance stage: After every second day the number of fully expanded and newly emerged leaves on the mainstem of the 4 central plants in each container were recorded to determine the leaf appearance stage (LAS i.e. the numbers of leaves appeared at a particular time).
Tiller appearance stage: The number of tillers on each of the 4 central plants in each container were recorded every second day staring from the day of stress till final harvest to determine the tiller appearance stage (TAS i.e. the number of tillers at a particular time).
Green leaf area: The final harvest was made on 15 January when the flag leaf was expanding. It was not possible to continue the experiment further due to limitation of vertical space for growing plants in the growth rooms. It was day 12 of the cycle in the variable salinity treatment. At the final harvest the 4 central plants from each of the replicate containers of each treatment were harvested. The leaves were detached from the stem area of stems and leaf area was determined using an automatic area meter (model AAM-7,Hayashi Denkoh Co., Tokyo, Japan).
Shoot and root dry weight: After recording leaf area, all the shoots parts (leaves plus stems) were rinsed twice with distilled water, blotted dry and then dried in an oven at 80 îC for 48 h. Similarly the roots of the same plants were also detached and rinsed first in tap water and then in distilled water to remove excessive salt present on them (after Rashid 1986). The roots were then blotted dry. The roots were later oven dried in the same way as the shoots and dry weight of shoot and root was recorded.
Statistical analyses: The data were subjected to analyses of variance (ANOVA) using the Genstat 5 statistical programme (Payne et al., 1987). When a significant F value was obtained for treatment effects, a least significant difference (LSD) test at 5 percent probability level was applied to the treatment means by using the following equation:
LSD = SEDM X T
Where LSD is least significant difference SEDM is standard error of difference between means T is value from distribution of t probability table at 5% level.
This test was used because as there were few treatment only a limited number of comparison could be made and therefore little risk of making type 1 errors (Gomez and Gomez 1984). All the graphs presented in this paper were prepared using the UNIRAS programme.
Experimental maintenance and observations: During the course of the experiment no insect or disease attack was observed. Plants growing near the walls of the growth room were found to be getting comparatively cooler air than plants in the center due to the air conditioning.
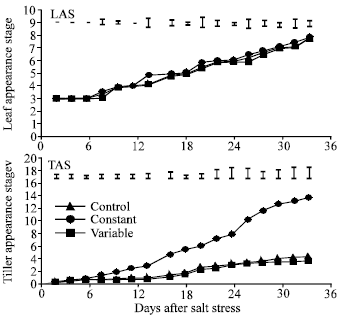 | |
| Fig. 4: | Effect of control (0 mole m-3 NaCl), constant (100 mol m-3 NaCl) and variable (50 to 150 mol m-3 NaCl) salinity on leaf appearance stage (LAS) and tiller appearance stage (TAS) of spring wheat. I= standard error of difference between means |
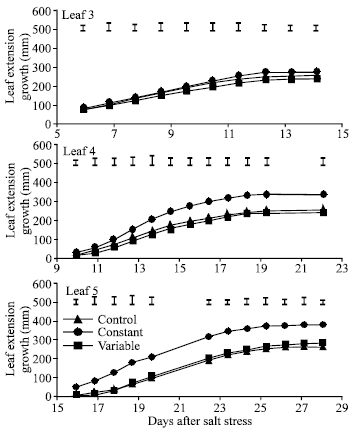 | |
| Fig. 5: | Effect of control (0 mole m-3 NaCl), constant (100 mol m-3 NaCl) and variable (50 to 150 mol m-3 NaCl) salinity on leaf extension growth of leaf 3, leaf 4 and leaf 5 of spring wheat. I= standard error of difference between means |
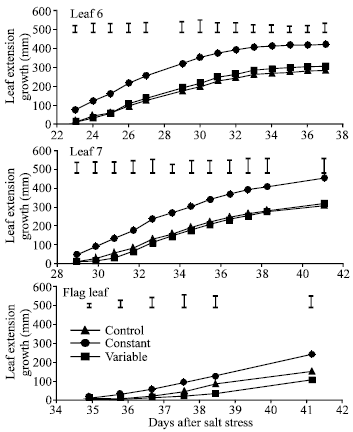 | |
| Fig. 6: | Effect of control (0 mole m-3 NaCl), constant (100 mol m-3 NaCl) and variable (50 to 150 mol m-3 NaCl) salinity on leaf extension growth of leaf 6, leaf 7 and the flag leaf of spring wheat. I= standard error of difference between means |
Therefore, the containers were re-randomized at weekly intervals. Sometimes the aerators (air stones) were found blocked. Hence, they were replaced regularly whenever required.
RESULTS
Effect of salinity on LAS, TAS and LEG: LAS was not significantly affected by salinity but was slightly higher in the control than in both salinity treatments from 14 days after salt stress (DAS) onward (Fig. 4). Salinity significantly decreased number of tiller from 8 days after salt stress. TAS was generally higher at variable than at constant salinity but the difference was not always significant. Salinity did not significantly affect LEG of leaf 3 (Fig. 5) and the flag leaf (Fig. 6) during the few days after their emergence (from 6 to 11 DAS and from 35 to 38 DAS, respectively). However, LEG was shorter in both salinity treatments than in the control. LEG was always higher at variable than at constant salinity in both of these leaves.
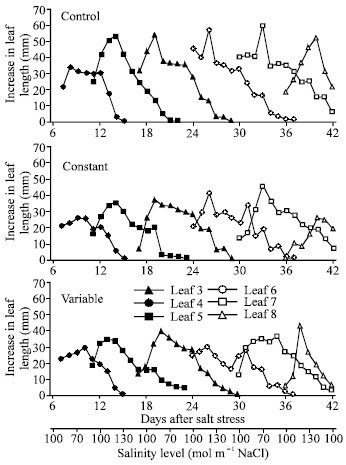 | |
| Fig. 7: | Effect of control (0 mole m-3 NaCl), constant (100 mol m-3 NaCl) and variable (50 to 150 mol m-3 NaCl) salinity on leaf extension growth of leaf 6, leaf 7 and the flag leaf of spring wheat. I= standard error of difference between means |
This trend was also observed in leaf 4 and leaf 7. Generally both salinity treatments significantly decreased LEG of these leaves. In leaves 3, 4, 7 and the flag leaf, LEG was generally higher at variable salinity than at constant salinity but the difference was not always significant. In leaves 5 and 6, LEG was generally higher at constant than at variable salinity.
DISCUSSION
Leaf extension growth was decreased markedly in later leaf insertions. This trend was also observed in an earlier experiment (Iqbal, 1988) where both NaCl and Na2SO4 salts decreased LEG in later leaf insertions of spring wheat. In the present study it was anticipated that at variable salinity daily increments in leaf length would decrease as salinity increased. Examination of Fig. 7 shows that this was not the case. Leaves 4, 5 and 7 had their main periods of extension growth during phases when salinity was increasing but the pattern was similar to constant salinity. It was also anticipated that at variable salinity daily increments of leaf length would be higher at variable than at constant salinity during periods when salinity was decreasing or less than at constant salinity. Leaf 6 had its main period of extension (first 5 days after appearance) when salinity was greater than at constant salinity. It was noticed that daily increments of leaf length for leaf 6 decreased with increase in salinity but increased when salinity was stepped down to 50 mol m-3 NaCl. In variable salinity there was some evidence that LEG increased when salinity decreased but a similar pattern was also observed in constant salinity, possibly indicating that this increase in leaf length could be attributed to solution changes. Munns et al. (1982) observed an increase in leaf elongation of barley plants grown for 7 days at 120 mol m-3 NaCl after plants were transferred to either 60 or 0.5 mol m-3 NaCl. They only examined leaf elongation for 4.5 h. Hence, the increase in leaf elongation that they observed could be also due to solution change.
REFERENCES
- Gorham, J., R.G. Wyn-Jones and A. Bristol, 1990. Partial characterization of trait for enhanced K+-Na+ discrimination in the D-genome of wheat. Planta, 180: 590-597.
CrossRefDirect Link - Kingsbury, R.W., E. Epstein and R.W. Pearcy, 1984. Physiological responses to salinity in selected lines of wheat. Plant Physiol., 74: 417-423.
Direct Link - Munns, R., H. Greenway, R. Delane and J. Gibbs, 1982. Ion concentration and carbohydrates status of the elongation leaf tissue of Hordeum vulgare growing at high external NaCl. II. Cause of the growth reduction. J. Exp. Bot., 33: 574-583.
Direct Link








