ABSTRACT
The influence of NaCl (25, 50, 75 mM) on selected physiological responses of kudzu (Pueraria lobata) was evaluated. Kudzu was grown for 14 days at various NaCl concentrations under greenhouse conditions at 550 μmol m-2 s-1 photon flux density, 45-50% RH and air temperature of 25±5°C. Kudzu growth was significantly reduced with an increase in NaCl concentration. Photosynthesis and stomatal conductance was significantly reduced as NaCl increased from above 25 mM. The reduction in stomatal conductance coincided with a similar reduction in transpiration. However, the decline in photosynthesis was independent of internal CO2 concentrations. Chlorophyll a and b were each sustained similar concentrations with the increase in NaCl up to 50 mM, which was followed by a significant decline in the concentration of both chlorophylls. Leaf water potential decreased with the increase in cellular solute accumulation as NaCl concentration increased in the growth medium. Carbohydrate accumulation in kudzu leaves increased significantly with an increase in salt concentration. In general, Na accumulation by kudzu increased as NaCl increased in the growth medium. Higher accumulation of Na occurred in roots compared with the shoots at 25 mM of NaCl. However, Na accumulation was similar in both roots and shoots at higher NaCl concentrations.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.114.119
URL: https://scialert.net/abstract/?doi=ajps.2004.114.119
INTRODUCTION
Adverse effects on crops resulting from increased salt concentrations in soil have become an unavoidable problem in arid and semiarid regions[1,2]. This increase in salinity has ultimately led to a reduction in crop growing areas[3]. Salt buildup at the soil surface occurs especially in areas of low precipitation and high temperature, due to the high rate of evaporation that leaves the minerals contained in the water behind in the soil[4]. Furthermore, irrigation with water high in salt content aggravates the problem, especially in areas poorly drained[5].
Reductions in plant growth due to salinity stress have often been attributed to three physiological mechanisms encompassing reduction in cell turgor pressure and photosynthetic activity and negative effects of salt ions on critical metabolic pathways[5].
Expansion of meristematic cells in the roots and shoots, which is ultimately responsible for plant growth, can be inhibited by increased salt concentrations[5]. Increased salt concentrations on agricultural lands can continue to present problems even some time after restoration processes are undertaken. The evaluation of species with relatively high salt tolerances as alternative crops is becoming a common practice as part of the restoration process. Kudzu (Pueraria lobata) is a leguminous, weedy vine with pubescent stems, trifoliate leaves and a perennial deep root system that is commonly found in the southeastern United States[6]. Kudzu is a hardy plant with high growth potential, even under adverse conditions such as on eroded soil with low fertility, low pH and poor water holding capacity[7]. Moreover, kudzu can control soil erosion and enhance soil fertility through nitrogen fixation[8]. These characteristics of kudzu make it an ideal candidate to be evaluated as an alternative species for use in the restoration of high-salinity soils. However, there is very little information available on the effects of salt on kudzu. Therefore, the objective of this study was to determine the effects of NaCl at soil concentrations of 0, 25, 50 and 75 mM on selected physiological responses of kudzu, these include plant growth, leaf gas exchange, pigment concentration, osmotic and water potentials and carbohydrate accumulation. In addition, plant uptake of sodium at the various NaCl concentrations was evaluated.
MATERIALS AND METHODS
Plant material: Kudzu seeds were scarified in concentrated sulfuric acid for 30 min[9]. The seeds were germinated in plastic pots containing a half-and-half mixture of vermiculite and potting soil. The plants were placed in a greenhouse and allowed to grow at 550 μmol m-2 s-1 photon flux density, 45-50% RH and air temperature of 25±5°C. Four weeks after emergence, uniform plants were individually transplanted into 900 ml glass containers, which contained half-strength Hoagland's solution[10] at a pH of 6.5. Each container was wrapped with aluminum foil to prevent light penetration to prohibit algae growth. One week later, the plants were randomly assigned to NaCl treatments 0, 25, 50 and 75 mM. A continuous airflow through the nutrient solution was achieved through a connection of glass containers to air pumps. Distilled water was added as needed to compensate for water loss due to evapotranspiration. The plants were allowed to grow under the experimental conditions for fourteen days.
Growth determination: The leaf area of seven plants from each treatment was determined with Li-Cor-3100 leaf area meter (Lincoln, NE, USA). Each plant sample was then oven dried at 85°C for 48 h and the shoot and root dry weights were recorded.
Gas exchange measurement: Simultaneous measurement of CO2 assimilation, stomatal conductance, transpiration and internal CO2 concentration of intact second, most mature leaves of seven plants from each treatment were made. The measurements were taken under control environment four hours after the onset of the light period at day fourteen of the treatment application. The selected leaf of each sample was enclosed in a flow-through plexiglas assimilation chamber (4.5x11.8x7.3 cm) of a Li-Cor 6200 photosynthesis system (Lincoln, NE, USA) as described by McDermitt et al.[11]. Standard measurement conditions of the growth chamber were 360 μmol m-2 s-1 photon flux density, 20-25% RH and 29°C and a leaf to air vapor pressure difference of 1.0 kPa.
Water potential measurement: Leaf water potential of seven samples of each treatment was measured as described by Al-Hamdani et al.[12]. Leaf discs (0.24 cm2) were removed from each plant using a leaf cutter psychrometer. The discs were sealed in the psychrometer chamber, which was then allowed to equilibrate (temperature and vapor) in a 25°C water bath. Using a thermocouple psychrometer meter (model 85, Logan, Utah), the wet bulb depression was determined. Using the resulting readings, water potential was calculated for the individually calibrated psychrometers. The same number of samples of the same leaves was used for osmotic potential determination. Cell sap, 100 μL, was extracted from each of the selected leaves with a French Press, loaded on a paper disc and measured with a vapor pressure osmometer (model 5520, Wescor, Logan, UT).
Chlorophyll determination: A sample of four leaf discs, with a total area of 7.5 cm2, were placed in a vial containing 5 ml DMF (N, N-Dimethylformamide) and incubated in the dark for 36 h at 4°C to extract chlorophyll. Chlorophyll a and chlorophyll b concentrations of seven samples from each treatment were determined spectrophotometrically by the method of Inskeep and Bloom[13].
Carbohydrate determination: Carbohydrate analysis was conducted following a procedure slightly modified from Chatterton et al.[14]. The samples were ground into a fine powder and a 100-500 mg portion was placed in a sealed vial and used for the determination of soluble sugars, starch and fructan as reported in detail by Wilson and Al-Hamdani[15].
Sodium uptake: Seven sample aliquots (0.1 -0.7 g) of dried root and shoot from each treatment were refluxed for 15 min in 10 ml 6N HNO3. To the samples, 5 ml concentrated HNO3 was added and the reflux was continued until approximately 5 ml of solution was left. After the solution had cooled, 2 ml of water and 5 ml of 30% hydrogen peroxide was added. The samples were heated slowly and hydrogen peroxide was added to the solution, one-ml at a time, until effervescence ceased. The samples were allowed to cool and HCl was added in a ratio of 1:2 (v/v). After all the plant tissues were digested the solution was then brought up to 75 ml with distilled H2O. The metal content the acid extracted was determined with a Buck Model 210 VGP atomic absorption spectrophotometer at 357.9 nm.
Data analysis: The experiment was repeated twice and the data combined. All variables were statistically analyzed as a randomized complete block design with each replicate as a block. Mean separations for the variables, which showed significant F values, at 5% level of probability, of the ANOVA analysis was based on the least significant difference (LSD) test[16].
RESULTS AND DISCUSSION
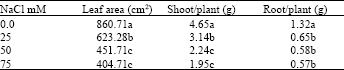
Kudzu growth represented by leaf area was significantly reduced by the presence of the salt in the growth medium (Table 1). There was a reduction of 27.59, 47.52 and 52.98% in leaf area as a result of an increase of salt concentration to 25, 50 and 75 mM, respectively. However, the reduction in leaf area was not significantly different between the NaCl concentrations of 50 and 75 mM. Similarly, the shoot growth significantly declined as impacted by the presence of the salt. On the other hand, root growth was reduced by approximately 56% at 50 and 75 mM in contrast to 50% reduction for those grown at 25 mM.
| Table 1: | Influence of different concentrations of NaCl on kudzu growth |
 | |
| Table 2: | Influence of different NaCl concentrations on gas exchange characteristics of kudzu |
 | |
| Table 3: | Influence of different concentrations of NaCl on kudzu chlorophyll a and b in kudzu |
 | |
| Table 4: | Influence of different NaCl concentrations on osmotic and water potentials status in kudzu leaf |
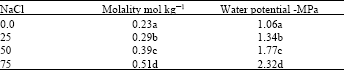 | |
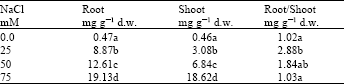
| Table 5: | Sodium uptake by the root and shoot of Kudzu as influenced by the different NaCl concentrations in the growth media |
 | |
| Means followed by the same lowercase letter in each column are not significantly different based on the LSD test (P=0.05) | |
Nevertheless, the decline in root growth was not statistically significant among the different NaCl concentrations. Increase in salt concentration was reported to inhibit the expansion of meristematic tissues, which is ultimately responsible for the growth of the plant[5]. In a study on Atriplex prostrata, an herbaceous halophyte, it was shown that plant growth was dramatically inhibited by salinity[17]. Furthermore, a continuous significant reduction in shoot and root growth of pigeon pea (Cajanus cajan) was a result of increased salt concentrations from 40 to 80 mol m-3[18]. Detrimental effects of salinity on plant growth mostly relate to the toxic consequence of Na and Cl and to the impact of these ions on water and nutrient uptake. In addition, reduction in kudzu growth could be due to influence of salt stress o photosynthetic activities. Salinity induced reduction in kiwifruit (Actinidia sinensis) growth, was, in part, due to a decline in leaf development and photosynthetic activity[19].
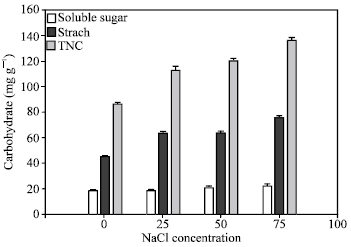 | |
| Fig. 1: | Influence of different NaCl concentrations on Carbohydrate accumulation in Kudzu root. Each point represents the mean of two experiments (n=14)±SE |
Carbon dioxide assimilation was significantly reduced at both salt concentrations of 50 and 75 mM (Table 2). However, this reduction was not significantly different at 25 mM NaCl in comparison to the control and to 50 mM NaCl. Stomatal conductance was gradually reduced as the salt concentration increased in the growth medium (Table 2). The reduction in stomatal conductance coincided with a similar reduction in leaf transpiration, which was significantly reduced at both 50 and 75 mM NaCl in comparison to the control plants. Internal carbon dioxide concentration was not influenced by the different NaCl concentrations with the exception of those grown at 75 mM, which were significantly increased. Similar findings by Wang et al.[17] reported a reduction in the stomatal conductance and transpiration rate of Atriplex prostrata as result of salinity stress. Likewise, the rate of stomatal conductance was directly correlated to the rate of photosynthesis. In addition to affecting stomatal conductance, salinity has been shown to inhibit nonstomatal factors, which influence photosynthesis. A reduction in photosynthesis was observed in Prunus salicina due to increasing salinity levels and was attributed to an increase in leaf chloride levels[20]. Furthermore, a decline in the activity of ribulose 1,5-bisphosphate carboxylase (Rubpcase) and pool size of triosephosphate, ribulose 1,5-bisphosphate (Rubp) and phosphoglycerate was observed in response to increasing salinity levels. A rise in leaf dark respiration was also detected in association with elevated leaf chloride concentration that might further contribute to a decline in net CO2 assimilation and plant growth. The light reaction mechanism of photosynthesis also found to be hindered by increasing salinity stress[21]. Specifically, irreversible inactivation of PSI and PSII resulted of osmotic and ionic effects of NaCl[22]. Chlorophyll a and b were each sustained similar concentrations with the increase in NaCl up to 50 mM, which was followed by a significant decline in the concentration of both chlorophylls (Table 3). The chlorophylls were impacted similarly by the various salt concentrations as indicated by the insignificant difference in chlorophyll a/b ratio. Chlorophyll b is found exclusively in association with the light-harvesting complex of photosystem II[23]. The increase in the pigment concentration of photosystem II was considered to be as an energy balance between the two photosystems. Krishnaraj et al.[3] reported that increased salt concentration resulted in an increase in chlorophyll a and b in wheat (Triticum aestivum) salt resistant variety, whereas no notable variations were shown in the susceptible variety. Increased chlorophyll concentration in stressed plants can be interpreted as an index of tolerance to NaCl; however, it should not be the only consideration. In this, the increase in chlorophylls in association with salt increase up to 50 mM was not statistically significant.
Solute accumulation in the leaf was directly proportional to the increase in the salt concentration in the growth medium (Table 4). The significant increase in molality, number of moles of solute/ weight of solvent in kg, was shown to coincide with a decrease in water potential values. The capacity to alter tissue water potentials to a lower level than that of the soil is one of the common responses of plants to salinity stress to sustain the water uptake[17]. Osmoregulation involves synthesis and accumulation of various organic and inorganic solutes in order to reduce water potential and maintaining high turgor that is necessary for plant growth[24,25]. Proline, which is commonly accumulated in the cytoplasm in response to water stress, was found to contribute with other amino acids to the adaptation process to increased salinity[26]. An increase in the soluble sugars, such as glucose and mannitol in response to elevated salt content was found to play an active role in osmotic adaptation to salinity[27]. A highly negative leaf osmotic potential was shown in pigeon pea as result of increase NaCl concentration[18]. Allakhverdiev et al.[22] reported that NaCl induced osmotic effects resulting in decreasing cytosol water content and increasing the intercellular salt concentration. In addition, the increase in intercellular salt concentration was caused by an influx of Na+ ion through potassium/sodium channels.
Kudzu responds to elevated salt concentration by increasing root carbohydrate accumulation (Fig. 1). The highest soluble sugar increase (16.89%) was obtained in plants exposed to 75 mM NaCl, followed by 13.16% increase at 50 mM. However, there is no difference in soluble sugar concentration between those obtained of the control and 25 mM. Starch concentration was similar at 25 and 50 mM, which were significantly different from the control and 75 mM NaCl. The trend of the starch accumulation seemed to be a major influence on the pattern of total nonstructural carbohydrate accumulation as indicated by the similar outcome. Similarly, an increase in soluble and insoluble carbohydrate accumulation in salt stressed Hygophila polysperma was reported by Kovach et al.[28]. Increased carbohydrate accumulation in kudzu was also observed in response to Cr stress[29]. Azcon-Bieto[13] reported that lower rates of carbon assimilation and a decrease in yield were associated with carbohydrate accumulation in many plant species. In addition, increased carbohydrate accumulation was observed in response to Cr stress in azolla (Azolla caroliniana)[15]. Carbohydrates accumulation might influence by reduction in carbohydrate utilization due to decline in growth and overall energy demanded by stressed plants.
Kudzu uptake of Na was significantly increased in both root and shoot with increased NaCl concentration (Table 5). In general, Na accumulation in the root was higher than that in the shoot. This with the exception at 75 mM NaCl, Na concentration was similar. The gradual increase in NaCl concentration in the growth media from 25, 50, to 75 resulted in a 1.4 and 1.5 fold increase in root Na uptake, respectively. In contrast, at these concentrations the increase in Na shoot uptake was 2.2 and 2.7 fold. This can be interpreted that Kudzu responds to increased NaCl concentrations by increasing Na export to the shoot. This might be an adaptation measurement to avoid the toxicity of the Na by distributing it to different regions within the plant.
In conclusion, the findings of this study showed that salt stress negatively impacted kudzu growth and photosynthesis. However, this negative influence was significant at only concentrations exceeding 25 mM NaCl. This finding coincided with the visual observations of the kudzu plants grown under the different salt concentrations, which exhibited stress symptoms such as leaf tip burning mainly at 75 mM. Carbon dioxide assimilation continued, although at a decreasing rate, even with the relatively high salt concentration. Kudzu responds to increased salt concentration by decreasing water potential, possibly to ensure water uptake. Furthermore, kudzu might be considered as a successful replacement crop in areas where NaCl concentrations are less than 50 mM. Planting kudzu in any area should consider the vigorous growth characteristic of the kudzu, especially in the absence of the natural enemy such as in the southern U.S.
REFERENCES
- Hoagland, D.R. and D.I. Arnon, 1938. The water culture method for growing plants without soil. Calif. Agric. Exp. Station, C347: 1-39.
Direct Link - Wang, L.W., A.W. Showalter and I.A. Ungar, 1997. Effect of salinity on growth, ion content and cell wall chemistry in Atriplex prostrata (Chenopodiaceae). Am. J. Bot., 84: 1247-1255.
Direct Link - Chartzoulakis, K.S., I.N. Therios, N.D. Misopolinos and B.I. Noitsakis, 1995. Growth, ion content and photosynthetic performance of salt-stressed kiwifruit plants. Irrig. Sci., 16: 23-28.
CrossRefDirect Link - Ziska, L.H., L.H. Seeman and T.M. DeJong, 1990. Salinity induced limitations on photosynthesis in Prunus salicina, a deciduous tree species. Plant Physiol., 93: 864-870.
Direct Link - Allakhverdiev, S.I., A. Sakamoto, Y. Nishiyama, M. Inaba and N. Murata, 2000. Ionic and osmotic effects of NaCl-induced inactivation of photosystems I and II in Synechococcus sp. Plant Physiol., 123: 1047-1056.
Direct Link - Tattini, M., R. Gucci, A. Romani, A. Baldi and J.D. Everard, 1996. Changes in non-structural carbohydrates in olive (Olea europaeaa) leaves during root zone salinity stress. Physiol. Plant, 98: 117-124.
CrossRefDirect Link - Kovach, C.W., J.P. Kurdziel, R. Bowman, J. Wagner and J.M. Lawrence, 1992. The effects of stress and disturbance on proximate composition, allocation of producationn photosynthesis, respiration and chlorophyll levels in Hygrophila polysperma (Roxb.) Anders. (Acanthaceae). Environ. Exp. Bot., 32: 479-486.








