ABSTRACT
Survey on insecticide resistance monitoring for a period of one year indicated that the Helicoverpa armigera population of Tamil Nadu developed very high level of resistance to synthetic pyrethroids, medium level of resistance to chlorpyrifos and quinalphos, low level of resistance to endosulfan, thiodicarb and profenofos and cent per cent susceptibility to the new chemistry spinosad. Synergistic studies with piperonyl butoxide, pungam oil and profenofos clearly indicated that the common MFO mechanism was responsible for the high level resistance observed to synthetic pyrethroids.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.94.100
URL: https://scialert.net/abstract/?doi=ajps.2004.94.100
INTRODUCTION
The Gram caterpillar, Helicoverpa armigera Hubner (Noctuidae: Lepidoptera) has become number one agricultural pest in India, Pakistan and China[1]. The damage by H.armigera was estimated at more than 2000 crores ($ 450m) nationally with 15 per cent decline in the cotton yield[2]. More than 75 per cent of the insecticides used in cotton are being targeted towards H.armigera[3]. This high selection pressure might induce the development of resistance in H.armigera which is considered as one of the major constraints to cotton production in Tamil Nadu[2]. Hence, a survey was carried out for one year to monitor the level of resistance and mechanism involved to commonly used insecticides including new chemistries such as spinosad and thiodicarb in Tamil Nadu.
MATERIALS AND METHODS
H.armigera population collected at weekly intervals from three different locations (Coimbatore, Poluvampatty and Madukarai) during June 2002 (23rd standard week) to April 2003 (18th standard week) was assayed for the level of resistance. Same set of population was exposed simultaneously to the discriminating doses of all the eleven chemistries (Synthetic pyrethroids-5; Organophosphates-3; Organo Chlorines-1; Carbamate-1; Spinosad) (Table 1) and synergists.
Bioassay methods
Topical bioassay: Technical grade insecticides were dissolved in analytical grade acetone and used for bioassays. Third instar larvae were assigned for topical application on the dorsal thorax with 1.0 μl of discriminating doses using Hamilton repeating dispenser. A minimum of 48 larvae were used per insecticide. After treatment, larvae were held individually in 12 well tissue culture plates containing semi-synthetic diet, at 25 (±2)°C. Control larvae were treated with acetone alone.
Ingestion method/diet contamination method: As thiodicarb is a stomach poison without any contact toxicity to H.armigera and little solubility in solvents, this insecticide was incorporated into the larval diet via surface disturbance[4]. Insecticide dilution (1.5% w/v) was put on the surface of diet @ 1 μl 2.5 mm-3 of artificial diet. Third instar larvae were allowed to feed on the artificial diet. Not less than 48 larvae were used. After treatment, the 12 well tissue culture plates were maintained at 25 (±2)°C. The mortality of treated larvae was observed at every 24 upto 144 h. Larvae were considered dead if they were unable to move in a coordinated manner when prodded.
The mechanism of resistance in field population was identified by employing the synergists. The synergists were applied along with discriminating dose of insecticide. The per cent suppression of resistance was computed as follows:
 |
RESULTS AND DISCUSSION
The level of resistance was at peak to synthetic pyrethroids irrespective of the locations surveyed (Table 2, 3, 4 and 8).
| Table 1: | Discriminating doses for insecticide resistance monitoring |
 | |
| Table 2: | Resistance frequency to synthetic pyrethroids in Coimbatore population of H.armigera |
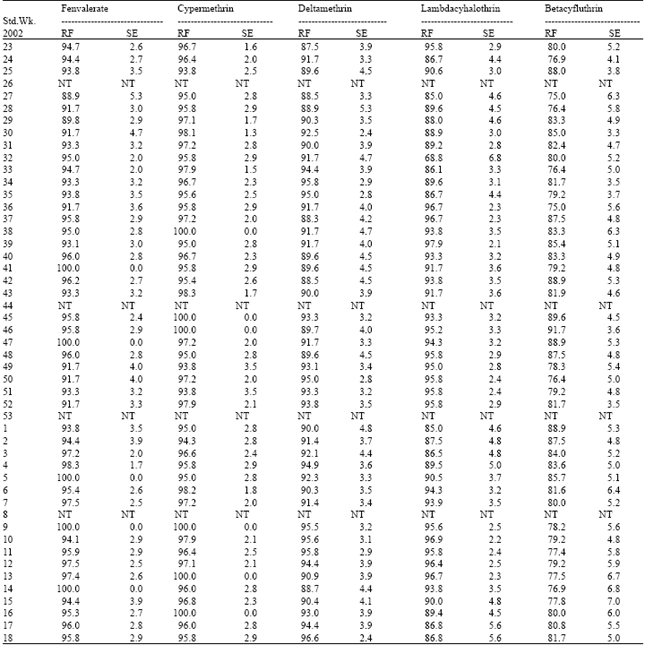 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
| Table 3: | Resistance frequency to synthetic pyrethroids in Poluvampatty population of H.armigera |
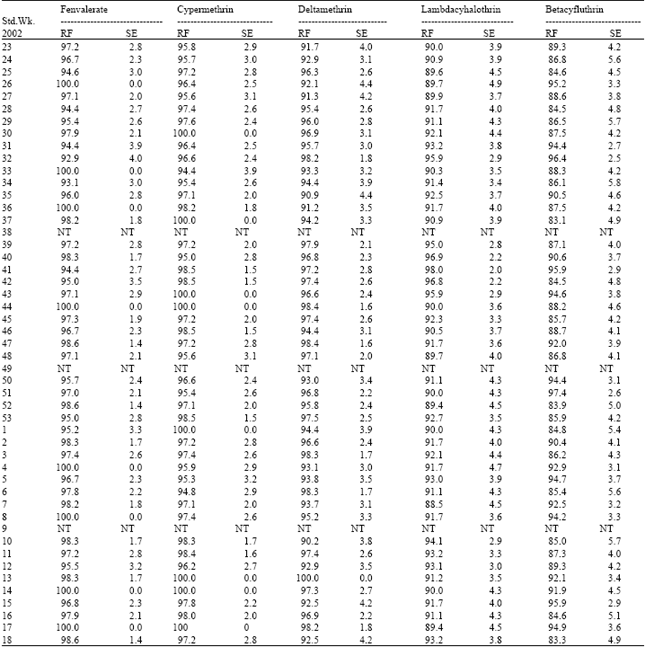 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
Cent per cent resistance was detected to fenvalerate, cypermethrin and deltamethrin during certain periods of survey. This was due to indiscriminate usage of pyrethroids over the past one decade. Pyrethroids alone constituted 50-70% of the insecticides targeted against H.armigera in cotton ecosystem[5]. This high selection pressure induced the activities of mixed function oxidases (MFO) and carboxyl esterases (CE). The synergistic suppression of resistance by piperonyl butoxide, pungam oil and profenofos clearly demonstrated that the MFO enzymes played the major role in the detoxification of pyrethroids in field population (Table 9). Scott and Georghiou[6] had shown that MFO- mediated resistance is specific to pyrethroids having phenoxy-benzyl group. Since all the five synthetic pyrethroids detected for field level resistance in the current investigation are ester bonded phenoxy-benzyl alcohols, the common MFO-mediated mechanism could be the reason for very high level of resistance observed among the pyrethroids.
The level of resistance to organophosphates was low to medium (Table 5, 6 and 7).
| Table 4: | Resistance frequency to synthetic pyrethroids in Madukarai Population of H.armigera |
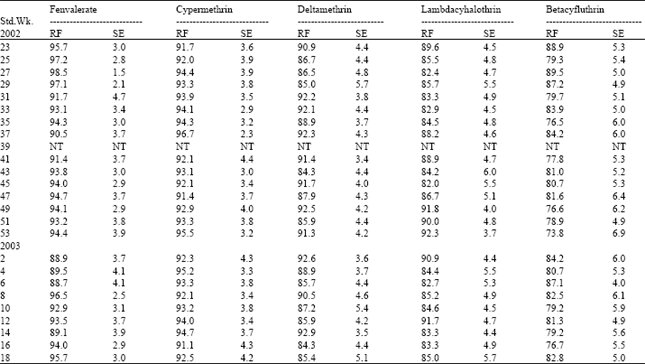 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
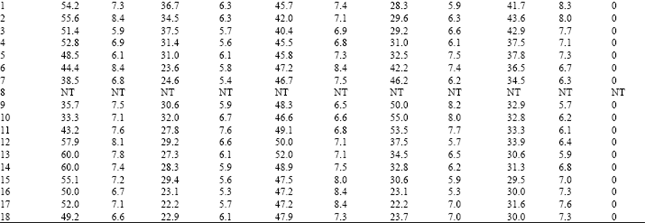
| Table 5: | Resistance frequency to OP compounds, endosulfan, thiodicarb and spinosad in Coimbatore Population of H.armigera |
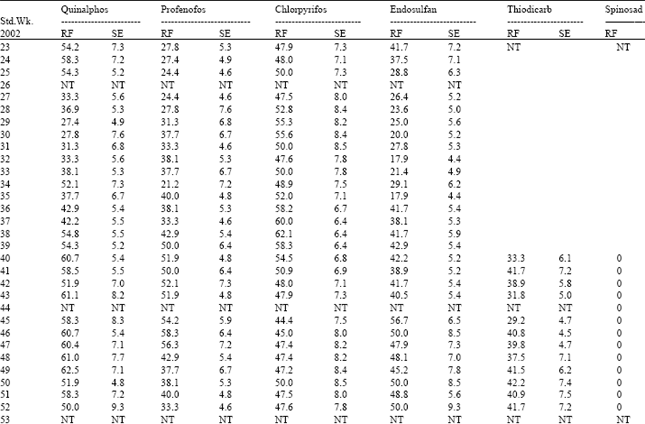 | |
 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
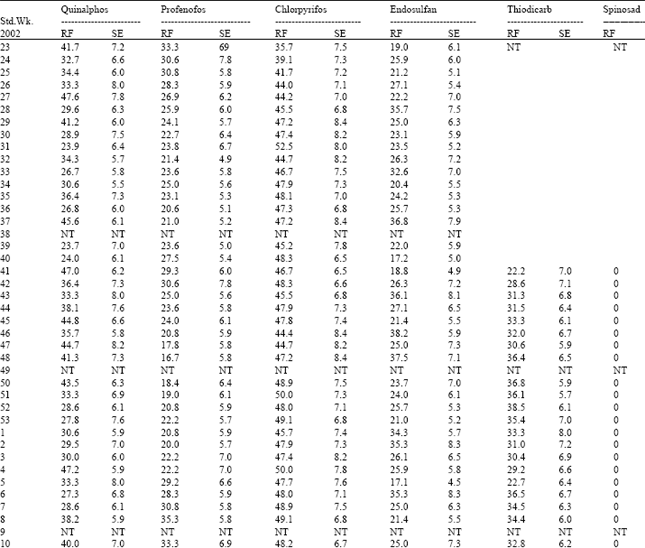
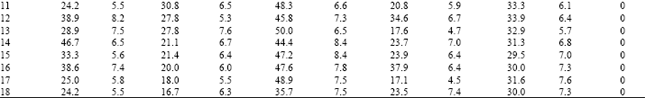
| Table 6: | Resistance frequency to OP compounds, endosulfan, thiodicarb and spinosad in Poluvampatty population of H.armigera |
 | |
 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
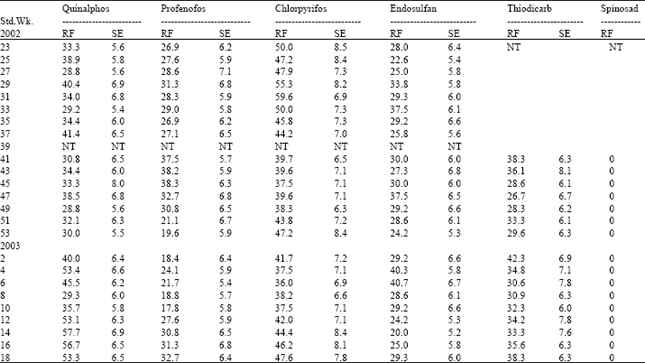
| Table 7: | Resistance frequency to OP compounds, endosulfan, thiodicarb and spinosad in Madukarai Population of H.armigera |
 | |
| RF: Resistance frequency (%); SE: Standard error; NT: Not tested | |
| Table 8: | Mean Resistance frequency*(%) to different chemistries in H.armigera population of Tamil Nadu (a comparison among the three locations) |
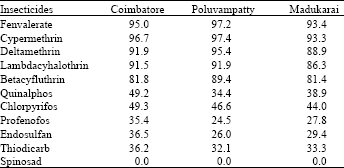 | |
| *Mean of resistance frequency data obtained for 49 weeks (June 2002-April 2003) | |
Among the three OP compounds tested, the resistance frequency was comparatively higher to chlorpyrifos followed by quinalphos. However, H.armigera population from three different locations showed low level of resistance to profenofos (24.5-35.4%) (Table 8).
| Table 9: | Mean synergistic suppression (%) of insecticide resistance by MFO and CE inhibitors |
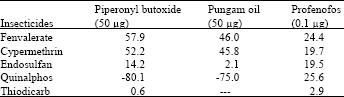 | |
Increased activity of MFO detected in these locations might activate the phosphorothionates into more toxic phosphates. The inhibition of MFO by piperonyl butoxide or pungam oil greatly reduced the toxicity of quinalphos (Table 9). This also confirmed the importance of MFO for activation of phosphorothionates. Among the three phosphorothionates, profenofos is a specific synergist recommended to inhibit the activity of CE; therefore the resistance was lower to profenofos. These are the reasons for low to medium level of resistance detected to these chemicals even though the OPs are being used from their introduction to till date against H. armigera in cotton ecosystem. The population also showed low level of resistance to endosulfan (26.0-36.5%) and the carbamate thiodicarb (32.1-36.2%) (Table 8) because of less frequent application.
The population was cent per cent susceptible to the new chemistry spinosad (Table 5, 6 and 7). The mode of action of spinosad is entirely different from the mode of action of already existing chemistries. Spinosyn-A, the primary constituent of the novel insecticide spinosad was found to elicit inhibitory action at GABA (gamma amino butyric acid) receptor[7]. It had also been established that spinosyn A had little ability to interact with other known GABAergic radio ligands such as organochlorines including endosulfan. This novel mode of action might be the reason for the lack of resistance to spinosad in H. armigera population of Tamil Nadu. Based on the results obtained in the present investigation, it may be advisable to go for rotation of insecticides incorporating new chemistries such as spinosad to manage the pyrethroid resistant population of H. armigera.
ACKNOWLEDGMENTS
The financial support from the Common Fund for Commodities (CFC) Europe, International Cotton Advisory Committee (ICAC) U.S.A. and Natural Resources Institute (NRI), U.K. is acknowledged.
REFERENCES
- Scott, J.G. and G.P. Georghiou, 1986. Mechanisms responsible for high levels of permethrin resistance in the housefly. Pestic. Sci., 17: 195-206.
Direct Link - Forrester, N.W., M. Cahill, L.J. Bird and J.K. Layland, 1993. Management of pyrethroid and endosulfan resistance in Helicoverpa armigera (Lepidoptera: Noctuidae) in Australia. Bull. Entomol. Res., 1: 1-132.
Direct Link - Gunning, R.V., C.S. Easton, L.R. Greenup and V.E. Edge, 1984. Pyrethroid resistance in Heliothis armigera (Hubner) (Lepidoptera: Noctuidae) in Australia. J. Econ. Entomol., 77: 1283-1287.
Direct Link - Regupathy, A., D.S. Rajavel, S. Rajkumar and D. Russell, 1999. Present status of insecticide resistance in Helicoverpa armigera and its management in Tamil Nadu, India. Proceedings of the ICAC-CCRI Regional Consultation Insecticide Resistance Management in Cotton, Jun. 28-Jul. 1, CCRI, Multan, Pakistan, pp: 48-55.









etienne tendeng Reply
I want a to obtain a method to study insect resitance