ABSTRACT
Summer weeds, Abutilon theophrasti Medik., Amaranthus retroflexus L., Echinochloa colonum (L.) Link., Hibiscus trionum L., Setaria verticillata (L.) P. Beauv., Solanum nigrum L. and Sorghum halepense (L.) Pers. and winter weeds Avena sterilis L. and Sinapis arvensis L., are important weed species in the Cukurova Region of Turkey. In order to detect seed longevity of these weeds, a seed burial experiment was initiated in 1994, except for A. sterilis and S. arvensis, which were buried in 1995. Seeds were recovered 7 years after burial and seed longevity was determined using by germination tests and TTC test. Burial depth, 15 and 30 cm had no significant effect on seed viability. Seed longevity varied among weed species: .A. theophrasti 6.71%; A. retroflexus 0.26%; A. sterilis 0.26%; E. colonum 1.42%; H. trionum 22.74%; S. verticillata 1.66%; S. arvensis 38.18%; S. nigrum 5.38% and S. halepense 8.83% after 7 years.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.1.5
URL: https://scialert.net/abstract/?doi=ajps.2005.1.5
INTRODUCTION
Weeds are always present in agronomic systems and most owe their beginning to weed seed in the soil[1]. Weed seeds are distributed in different depths of soil layers by tillage implements and they germinate when the conditions are favorable and resulting plants compete with crops for sources such as water and nutrients[2].
Information on weed seed longevity in the soil is one of the essential parts of understanding weed/seed biology. It helps to understand weed population dynamics, to establish economic thresholds, to design management strategies, to prepare models, to plan effective weed control programs and to make management decisions[2-8]. Even it is expected that information on seed longevity in the soil will be more needed in the future for decision making in weed management programs[8].
Studies on seed longevity lie back to the 1800s. The best known and the world’s oldest study on seed longevity in the soil was Beal’s trial which was initiated in 1879 in Michigan, the United States of America. Beal buried seeds of 23 plant species in sand in bottles mouth downward at depth of 46 cm. His study is still in progress and will continue until 2100[9].
It has been shown that seeds of some weed species buried in the soil can remain viable for 20 years or more[10,11]. Buried seeds of 36 species out of 107 were still viable after 39 years[12]. Furthermore, Livingston and Allesio[13] concluded that seeds of some weed species buried in the soil can remain viable for 50-100 years or more. Seeds of 3 of 23 species buried were viable after 100 years[14].
The longevity of seeds varies considerably depending upon the species, depth of seed burial, soil type and tillage[1]. Ozer[15] reported that seeds of P. oleracea L., Sinapis arvensis L., Solanum nigrum L. and Setaria verticillata (L.) P. Beauv. buried in the soil can remain viable for 30 years or more, but Avena spp. only 3-8 years. Egley and Chandler[3] found that seeds of 48% of Sorghum halepense (L.) Pers., 36% of Abutilon theophrasti Medik., buried in soil were still viable after 5.5 years but, the viable seed ratio was less than 1% for P. oleracea L. and A. retroflexus.
There are different reports about the longevity of the same species. Results of buried seed studies may not be transferable from one region to another[3,16], because differences in soil conditions may have considerable influence on seed survival although weeds have the same features[17]. Seeds of A. retroflexus remained viable after 10 years in an experiment[12] while it was viable after 40 years in another study[18].
The Cukurova Region with a wide range of crop pattern is one of the foremost agricultural areas of Turkey. Weeds are among major problems in summer and winter crops[19-26]. There are several studies related to soil seedbank in the region[27-29]. However, seed longevity has not been studied. This study was started to investigate the seed longevity of nine important weed species in the region in order to create data for future modeling and strategy developing works in 1994. Earlier findings of the study have been published elsewhere[30,31]. This paper discusses the findings on seed longevity at the 7th year of burial trial.
MATERIALS AND METHODS
Seed preparation and burial: Seeds of Abutilon theophrasti Medik., Amaranthus retroflexus L., Echinochloa colonum (L.) Link., Hibiscus trionum L., Setaria verticillata (L.) P. Beauv., Solanum nigrum L. and Sorghum halepense (L.) Pers., which are summer weeds, were collected in 1994 and of Avena sterilis L. and Sinapis arvensis L., which are winter weeds, collected in 1995 in Adana province, which is the main part of the Cukurova Region. The seeds were air dried, hand separated, cleaned and stored in the dark room at 15 or 25°C for winter weeds or summer weeds, respectively, until burial time.
Soil at the burial site in Adana was sandy loam with pH 7.2, 0.95% salt, 8.32% lime and 3.02% organic matter content. Seeds of each weed species were mixed with 0.3 kg of sterilized soil and put into a polypropylene mesh bags. Each bag contained 400 seeds, except for A. sterilis, which were only 100 seeds were put because of seed size. Bags were located 15 and 30 cm depths of soil in November 1, 1994 and May 1, 1995, for summer and winter weed species, respectively. Bags were replicated four times for each species in each burial depth.
Exhuming bags and seed viability tests: After 7 years of burial, seed bags from depths of 15 and 30 cm were carefully exhumed without disturbing neighbouring burials in 2001. The bags were transferred to the laboratory, opened and washed over a nematode sieve (0.20 mm).
Seed viability was determined at the burial time and after exhuming using three tests: Germination test with intact seeds, germination test with damaged seeds and TTC (3,5-triphenyltetrazolium chloride) test.
The seeds recovered from each bags were placed onto two layers of filter paper in 9 cm petri dishes and added 5 mL of distilled water. Petri dishes were placed in a growth chamber Incubation conditions were 30 °C for A. theophrasti, A. retroflexus, E. colonum, H. trionum, S. nigrum and S. halepense; 35°C for S. verticillata and 10 °C for A. sterilis and S. arvensis, which temperatures were determined by Uremis and Uygur[32]. Germination tests were lasted for 14 days in the dark. Seed was considered germinated when radicle was over 0.5 mm. Seeds that germinated were recorded and removed daily.
Remaining seeds (i.e. non germinated) after 14 days were subjected to further examination. Seed coats were pierced at the radicle end of the seed for A. sterilis, E. colonum, S. verticillata, S. nigrum and S. halepense with a dissecting needle and scarified for A. theophrasti, A. retroflexus, H. trionum and S. arvensis with emery paper. Seeds in petri dishes were placed into incubators which, were adjusted the same temperatures as previous test. Again, germinated seeds were recorded and removed daily. Tests were lasted seven days.
After these two procedures, the remaining non-germinated seeds underwent viability test with 0.3% TTC. Seeds were dissected and those with pink to red embryos were assumed as viable[16].
Data analysis: The number of viable seeds was calculated using the following formula for each species:
Where, RVS is the number of recovered viable seeds; GI the number of germinated seeds, which were intact; GD stands for the number of seeds germinated after damaging; TT is the number of seeds, which accepted viable in TTC test.
The rate of viability of exhumed seeds was calculated for each species using the equation below:
Where, PV is viability percentage for a given species; RVS, the number of recovered viable seeds and BVS, the number of buried viable seeds.
Initial Viability (IV) rate was calculated:
Where, TS is the number of seeds which were subjected to viability test just before burying.
Data were subjected to analysis of variance.
RESULTS AND DISCUSSION
Viability of seeds before burial was over 90%, except for E. colonum of which viability rate was 88% (Table 1).
After 7 years of burial, all species kept viability in both depth (data not presented). There was no significant relationship between species and burial depth. Burial depth itself did not affect viability either.
| Table 1: | Viability of seeds before burying and after 7 years burial duration |
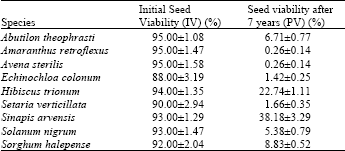 | |
Soil depth findings of current experiment including earlier findings of it Uremis and Uygur[30], Uremis and Uygur[31] were similar to that of earlier researchers. Egley and Chandler[3] found that there were no significant differences between viabilities of seeds buried at 23 and 38 cm after 5.5 year of burial but the viability was differed between 8 and 23 cm for some species. Although, there have been reports that showed the effect of burial depth on viability[2,5,33], those experiments compared several layers within top 10 cm of soil which might be affected by any agricultural practices and environmental changes and seeds kept in soil for shorter durations[2] or many layers from 0 to 34 cm, which can cause significant difference at environment in each depth[33]. Taylorson[34], Horng and Leu[35] reported that seeds in top levels of soils lost their viability fast whereas seeds below 15 cm could live longer. In another burial experiment comparing 2 and 15 cm burial depth, burial depth significantly affected seed viability in the first two years[5] but after 6.7 and 9.7 years[7]. Conn and Deck[7] attributed those findings to low number of viable seeds and variability among replicates. However, in our experiment, comparisons for depth effect on each species did not give any significant difference for the species which still have higher viability rate and less variability among replicates. Conn[6] found varying results related to soil depth for different species and/or different burial duration such as 2.7, 3.7 and 4.7 years in above mentioned long term burial trial. We speculate that the depth effect of 15 and 30 cm was similar because environmental effects in both depth might be similar.
Seed viability significantly varied among weed species. Species variation for viability is similar to the earlier studies[3,5,7,8].
Amaranthus retroflexus, A. sterilis, E. colonum and S. verticillata showed very low viability, under 5% (Table 1). Actually viability of A. retroflexus seeds drastically dropped after the first year of burial[31]. Egley and Chandler[3], Burnside et al.[8] found similar results. But for S. verticillata, Burnside et al.[8] found 18 and 26% viability in two locations after 7 years of burial. Such variabilities for seed longevity can be seen. For instance, at the same study they found different results between two locations for A. retroflexus and A. theophrasti in some years. Egley and Chandler[16] concluded that A. retroflexus, Sida spinosa and E. cruss-galli are problem in the fields because they produce enormous amount of seeds in spite of their shorter seed longevity. But, this explanation does not match A. sterilis which is a common and dense weed. It has short seed longevity and it does not produce seeds as much as A. retroflexus does[20,36,37].
On the other hand Darlington and Steinbauer[18] reported 40 years of longevity for A. retroflexus, Toole and Brown[12] 10 years and Burnside et al.[8] 17 years. It shows that seeds will live longer even very small rate of viability.
Abutilon theophrasti, S. nigrum and S. halepense showed 6.71, 5.38 and 8.83% of viability (Table 1). Burnside et al.[8] reported two different rate for two locations 5 and 40% for A. theophrasti after 7 year. Toole and Brown[12], Egley and Chandler[3] found that seed viability was 36% in 20 cm depth after 6 and 5.5 years, respectively. Lueschen and Andersen[38] found that seed viability was 37% after 4 years of burial; but, under tillage conditions longevity was less. Viability of S. halepense seeds reported 48% after 5.5 years of burial[3]. This is also three times higher than it was found in this trial at the 5th year[31].
Sinapis arvensis, one of the most common and dense weed in wheat and other winter crops in the region[22,24,25], had the highest viability, 38.18%, among 9 species (Table 1). The viability of S. arvensis seeds did not change compared with the 5th year of exhuming[31]. Brenchley[39] cited that the viability ratio was 87% in 30 cm after 10 years. One of the causes of seed longevity might be hard seed coats such as A. theophrasti, H. trionum and S. arvensis. Egley and Chandler[3], also stressed a relation between seed coats and longevity. However, a short-seed life species, A. retroflexus also have a hard seed coat.
The other species which maintained high viability is H. trionum. Unlike S. arvensis, viability of H. trionum decreased almost half from 5th to 7th years after burial[31].
Present findings for 9 species sometimes are in agreement with the earlier literatures; but most of the time, different results were obtained. It is a nature of longevity studies. Westra et al.[40] found that not only burial location but also seed sources’ location affected burial seed persistence.
It has been speculated that soil temperature and humidity can effect the longevity[16,38,41]. However, any of the studies has not established a world wide accurate explanation including this study. Dawson and Bruns[17] did this experiment yielded no explanation for these surprising differences. However, the results show clearly that the environmental factors that control the life and death of seeds in soil below the level favorable for germination are very subtle.
The trial will continue some more years. The data obtained so far and will be obtained will help to improve weed control in a sustainable way.
In the future, to produce high yielding crops with modern techniques might require more biological and ecological data. So, seedbank data could be used even more.
REFERENCES
- Uludag, A., I. Uremis, E. Aksoy, O. Gonen and I. Kadioglu, 2004. Relations between seedbank and weed flora in cotton areas. Zeitschrift Pflanzenkrankheiten Pflanzenschutz, 19: 57-64.
Direct Link








