ABSTRACT
The antagonistic activity against in vitro growth of Fusarium oxysporum f. sp. ciceri was determined for 90 isolates florescent Pseudomonas obtained from the rhizosphere and rhizosphere of healthy, partially wilted and completely wilted chickpea. Based on zone of inhibition, isolates were categorized into four categories, i.e. highly, moderately, least and non-antagonists. Maximum number of highly antagonistic isolates was obtained from rhizosphere and rhizoplane of healthy chickpea plants, whereas, maximum number of non-antagonistic isolates were obtained from completely wilted chickpea plants. Isolate H-Pf5 and C7R12 were selected for green house and field bioassays. Cell free culture filtrate of both the selected isolates was able to inhibit conidial germination of pathogen. Under green house and field conditions the isolates of fluorescent Pseudomonas, significantly enhanced seed germination, reduced disease incidence and promoted plant growth of chickpea as compared to control.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.446.454
URL: https://scialert.net/abstract/?doi=ajps.2007.446.454
INTRODUCTION
Fusarium wilt caused by Fusarium oxysporum Schlechtend Fr. f. sp. ciceri (Padwick) Matuo and K. Sato is a major constraint to chickpea (Cicer arietinum L.) cultivation through out the world and especially in Indian subcontinent, like in Punjab state, where chickpea is commonly grown pulse crop. Presently, the area under chickpea is 8.3 million hectare giving a total yield of 8.1 million tons but it decreased from 35 to 8.1 million tons from 1970 to 2004 due to attack of different diseases including fusarial wilts (Anonymous, 2005). Rhizosphere bacteria have been proved as effective biocontrol agents of various root diseases of agricultural important crops (Weller, 1988; Whipps, 2001). Specific strains of Pseudomonas spp. has been reported to suppress plant diseases such as F. oxysporum f. sp. raphani (de Boer et al., 1999), F. oxysporum f. sp. raphani (Duijff et al., 1998, Lemanceau and Alabouvette, 1991), F. oxysporum f. sp. lini (Duijff et al., 1999), Gaeumannomyces graminis (Hamdom et al., 1991; Raaijmakers and Weller, 1998), Pythium aphanidermatum (Moulin et al., 1996; Ongena et al., 1999) and Pythium ultimum (Bagnasco et al., 1998). Pseudomonas spp. produce cyanides/antibiotics, such as Pyrolnitrin and Pyolutiornin against plant pathogens (Schippers et al., 1985; Lemanceau et al., 1988; Lemanceau and Alabouvette, 1991; Leyns et al., 1990). The iron chelating system of fluorescent Pseudomonas deprives pathogens of iron under low availability of iron in the ecosystem (Sharma and Johri, 2003). Application of selected strains of fluorescent Pseudomonas spp. to seeds/cutting has lead to increased plant growth and increase crop yield due to increased availability of mineral nutrients (de Weger et al., 1995) and plant growth promoters and pathogens suppression as a result of antibiotic and siderophore production (Bakker et al., 1990; Moulin et al., 1994; Nowak et al., 1999) and induced plant resistance (Srivastava et al., 2001).
The objectives of the present studies were to isolate the fluorescent Pseudomonas bacteria from the chickpea rhizosphere and rhizoplane with maximum antagonistic activity against F. oxysporum f. sp. ciceri under Indian environmental conditions and determine the ability of selected bacterial isolates to suppress the Fusarium wilt of chickpea under greenhouse and field conditions and ultimately their effect on plant growth parameters of chickpea crop.
MATERIALS AND METHODS
Isolation of fluorescent Pseudomonas isolates: Isolation of fluorescent Pseudomonas was done from rhizosphere and rhizoplane soil samples collected from different chickpea fields at Plant Pathology Research Area in Punjab Agricultural University, Ludhiana, Punjab, India. Chickpea plants were categorized into three different plants types i.e., healthy, partially wilted and completely wilted plants on the basis of extent of wilting. For the rhizosphere samples, plants were uprooted and extra soil adhered to roots was removed by gentle shaking. One gram of rhizosphere soil was suspended in 10 mL of sterilized water and suspension was serially diluted to 10-5. For isolation of bacteria from these soil samples 1 mL of aliquot from each dilution was spread on King’s B medium (King et al., 1954) in 9 mm Petri plate and incubated at 28±2°C for 48 h in BOD incubator. For isolation of fluorescent Pseudomonas from rhizoplane of roots, the roots were chopped into small pieces and one gram of sample were shook in 100 mL of sterilized water for 30 min in shaker before using them for isolation. This suspension was serially diluted and spread over King’s medium in Petri plates for bacterial isolation. Plates were incubated and were screened under UV light for fluorescent colonies. Fluorescent colonies were picked up at random and transferred to King’s B medium in slants. The isolates were varying from fluorescent green, deep fluorescent green and fluorescent yellowish green. A total 90 isolates of florescent Pseudomonas were collected, 15 were from each rhizosphere and rhizoplane of healthy plants, partially wilted plants and completely wilted plants. The isolates were designated according to the type of soil samples and condition of plant from which they were isolated. Each culture was purified by streak culture method on King’ B medium. For storage of isolates of fluorescent Pseudomonas, a single colony of each isolate was transferred to 3 mL broth using a sterile needle and incubated at 28±2°C for 48 h with shaking at 120 rpm. The 700 μL of culture was added to 300 μL of sterile glycerol (30%) aseptically. The mixture was vortexed thoroughly and stored at 20°C. A loopful of culture was streaked on solid plate whenever needed.
Isolation of pathogenic isolate of Fusarium oxysporum f. sp. ciceri: The isolate designated as Foc was selected as pathogenic isolate of F. oxysporum f. sp. ciceri based on its high pathogenic ability to cause wilt in chickpea plant (Kaur, 2003).
Selection of fluorescent Pseudomonas for their ability to inhibit in vitro growth of F. oxysporum f. sp. ciceri: The isolates obtained from rhizosphere of healthy plants were designated as H-Pf1 to H-Pf15 and those from rhizoplane were named as H-Pf16 to 30. Similarly, those obtained from rhizosphere and rhizoplane of partially wilted and completely wilted plants were designated as PWPf1-15, PWPf16-30, CWPf1-15 and CWPf16-30, respectively. Discs of 5 mm diameter from actively growing culture of highly pathogenic isolate F. oxysporum f. sp. ciceri was inoculated on PDA in petri plates and after two days of its growth an isolate of fluorescent Pseudomonas was streaked to one of its sides.
| Table 1: | Categorization of isolates of fluorescent Pseudomonas obtained from rhizosphere and rhizoplane of chickpea plants on the basis of zone of inhibition against Fusarium oxysporum f. sp. ciceri |
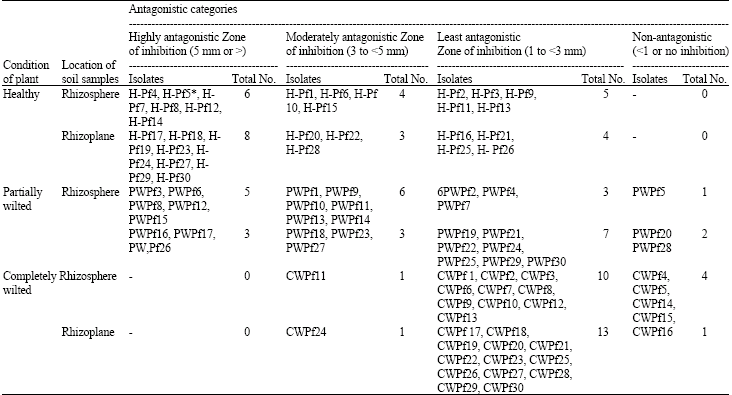 | |
| *The isolate H-Pf5 showed maximum zone of inhibition up to 7 mm between bacterial and fungal growth, thus selected for further studies | |
The observations were made on formation of zone of inhibition between fungus and the bacteria. The isolates were grouped into 4 categories i.e., highly (>5 mm) moderaterly (3 to <5 mm), least (1 to <3 mm) and non-antagonistic (<1 mm) based on zone of inhibition.
Effect of bacterial culture filtrate on conidial germination and hyphal growth of F. oxysporum f. sp. ciceri: Fluorescent Pseudomonas isolates H-Pf5, with maximum zone of inhibition and C7R12 obtained from INRA, Dijon France were selected and grown in King’B broth (KBB) at 25°C on a rotatory shaker at 120 rpm for 48 h. KBB without bacteria was used as control. Culture for each isolate was centrifuged at 10,000 rpm for 20 min and the supernatant was filtered through sterile 0.4 μm pore size millipore filter. Conidia of F. oxysporum f. sp. ciceri were taken from actively growing cultures and concentration was adjusted to 10-5 conidia per mL using haemocytometer. A 0.5 mL of aliquot of micro conidia suspension was mixed with 0.5 mL of a cell free bacterial culture filtrate. For control, 0.5 mL of KBB was added instead of cell free bacterial cultural filtrate. A 40 μL of bacterial filtrate-conidia mixture was placed on a glass slide and incubated at 25°C in moist chamber for 12 h. Later on a drop of acid fuchsine in lactophenol was added to kill and stain conidia. The mixture was examined with light microscope to determine conidial germination in five randomly chosen microscopic fields per glass slide. Four (40 μL) drops were assayed for each bacterium-fungal isolate combination. The ability of cell free cultures to inhibit hyphal growth of F. oxysporum f. sp. ciceri was tested using PDA cultures. A 5 mm plug of the medium was removed from the center of a plate and replaced with similar plug from the leading edge of a 7 day old fungal culture on PDA. Six equidistant 3 mm diameter wells were made 2.5 cm away from the centre of the plate and numbered from 1-6. A 40 μL drop of cell free culture filtrate (crude filtrate) was placed in well 1 and similar drops of 1/2, 1/4, 1/8 and 1/16 dilutions of crude filtrate and PDB were placed in well 2, 3, 4 and 5. Well 6 was filled with 40μL PDB as control. This experiment was replicated four times and plates were incubated at 25±2°C for 5 days. Hyphal growth inhibition was determined after 5 days of fungal inoculation using the formula.
Bioassay in greenhouse
Preparation of bacterial and fungal inoculum for in vivo greenhouse and field conditions: Charcoal based formulations of bacterial isolates were used to assess antagonistic activity of selected isolates under greenhouse and field conditions. Selected bacterial isolates were grown in 250 mL Erlenmeyer flasks containing 100 mL of sterilized King’B broth on a shaker at 120 rpm for 48 h. Bacterial cells were harvested by centrifugation (12 000 x g, 20°C for 10 min) and the pellet was suspended in 10 mL of sterile distilled water. The suspension was mixed with sterilized activated charcoal and CFU was adjusted by pour plate method to 1x108 CFU g-1 formulation (Kaur, 2003). For preparation of fungal inoculum, the pathogenic isolate F. oxysporum f. sp. ciceri (Foc) was grown at 25±2°C in 250 mL Erlenmeyer flasks containing sterilized maize-sand medium (20 g maize, 20 g sand and 30 mL Di H2O). Highly susceptible chickpea cultivar, JG62 was used for bioassays.
Seed treatment with antagonists: The surface sterilized JG62 seeds were treated with charcoal based formulation (Kaur, 2003) containing 1x108 CFU g-1 of fluorescent Pseudomonas isolates @ 8 g kg-1 seed. One percent Carboxymethyl Cellulose (CMC) was added as sticker. The treated seeds were dried in the shade before sowing. The loamy sand (type Utchrepts) soil with pH 8.2, electrical conductivity 0.4 sec cm-1, organic matter 0.2 % and 72 mg KMNO4 oxidizable N, 5.6 mg NaHCO3 extractable Olsen-P and 79 mg ammonium acetate extractable K per 1 kg of soil was used for the experiment. Sterilization of the soil was done at 121°C at 15 psi for 40 min 3 d prior to sowing and later on was mixed with Foc grown on maize-sand medium. The concentration of pathogen in the soil was adjusted to 1x103 CFU g-1. Seven treated seeds of chickpea were grown in pots (9 dia) filled with inoculated soil and thinned to 4 plants after germination. Seven replications for each bacterial isolate was used and untreated seeds were used as control. Plants were watered based on need. Wilt incidence, root length, shoot length and dry weight of aerial plant parts were recorded at 30 days intervals from date of sowing.
Soil treatment with antagonists: Soil was autoclaved twice in the same way as described above and pots were inoculated with the wilt pathogen Foc @ 1x103 CFU g-1 3 day prior to inoculation of antagonists. The inoculum of selected isolates of fluorescent Pseudomonas was mixed with soil to achieve the inoculum density of 1x108 CFU g-1 soil. Seven seeds of chickpea cultivar, JG62 (surface disinfected with 0.1% HgCl2 for 1 min) were planted two days after the addition of antagonists per pot and later on thinned to 4 plants after germination. Seven replications for each bacterial isolate was used and soil without bacterial treatment ware used as control. Plants were watered based on need. Wilt incidence, root length, shoot length and dry weight of aerial plant parts were recorded at 30 days intervals from date of sowing.
Bioassay under field conditions: Seed treatment of both isolates of fluorescent Pseudomonas was also under field conditions. Each plot, 2x1.5 m of size had five rows. Twenty five seeds of chickpea cultivar, JG62 (surface disinfected with 0.1% HgCl2 for 1 min) in a row in a plot (2x1.5 m) consisted of five rows, but thinned to 15 plants after germination. Foc inoculum (100 g) @ 1x103 CFU g-1 (grown on maize sand media) was mixed thoroughly in each plot up to 5 cm depth before sowing. Plots of both bacterial isolates were replicated thrice and plot with untreated seeds was kept as control. Plants were watered based on need. The wilt incidence was recorded 4 times during crop at 30 days intervals.
Statistical analysis: The data for microconidia germination, relative inhibition of mycelial growth, seed germination and disease incidence were square root transformed before analysis. Transformed data were analyzed by ANOVA using PROC GLM (SAS Institute 1999). Fisher’s least significant difference test with a significance level of α = 0.05 (SAS Institute, 1999) was used for means separation. All experiments were duplicated and the results shown here represent one representative experiment.
RESULTS
Selection of fluorescent Pseudomonas for their ability to inhibit in vitro growth of F. oxysporum f. sp. ciceri: Antagonistic potential of 90 isolates collected from rhizosphere and rhizoplane soil of chickpea plants was tested against F. oxysporum f. sp. ciceri in dual culture under in vitro conditions (Table 1). The isolates formed zone of inhibition ranging from 0-7 mm. On the basis of zone of inhibition, the isolates were divided further into four groups i.e., highly, moderately, least and non-antagonistic isolates. The maximum number of highly antagonistic isolates (14 isolates) were obtained from healthy chickpea plants followed by partially wilted plants (8 isolates). However, no potential antagonistic isolate as obtained from completely wilted plants. In contrast, the non-antagonistic isolates were obtained from completely wilted plants (5 isolates) and partial wilted plants (3 isolates) while none of isolate was non-antagonistic from soil samples obtained from healthy plants (Table 1). Isolate H-Pf5 gave maximum zone of inhibition upto 7 mm in dual culture, thus selected for further studies.
Effect of bacterial culture filtrate on conidial germination and hyphal growth of F. oxysporum f. sp. ciceri: Two isolates of fluorescent Pseudomonas H-Pf5 and C7R12 were employed for studying their effect on micro conidial germination and hyphal growth inhibition of wilt causing pathogen (F. oxysporum f. sp. ciceri) in chickpea. Cell free culture filtrate from both the selected isolate of fluorescent Pseudomonas significantly inhibited the conidial germination and hyphal growth of F. oxysporum. f. sp. ciceri (Table 2). There was 89.5% (10.5% germination) and 88% (12% germination) inhibition of conidial germination in glass slides having fungal culture amended with cell free culture of C7R12 and H-Pf5, respectively as compared to 100% conidial germination in control. However, the C7R12 isolate gave higher conidial germination but it did not differ significantly from isolate H-Pf5 (p>0.05). In case of mycelial growth inhibition the crude cell free filtrate from both the isolates of bacteria provided maximum growth inhibition. However, there was no significant difference within the isolates but observations were significantly different (p<0.05) from control (Table 2). Both the isolates proved highly antagonistic to F. oxysporum f.sp. ciceri. With the increase in the dilution of cell free culture filtrate of bacteria the antagonistic activity was also reduced. However, it differed significantly from control.
| Table 2: | Effect of cell free culture filtrates of selected isolates of fluorescent Pseudomonas on micro conidia germination and hyphal growth of Fusarium oxysporum f. sp. ciceri |
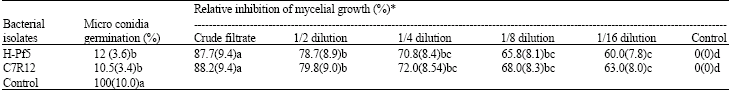 | |
With in the columns, means with a common lower case letter do not different significantly (p = 0.05) according to Fisher’s least significant different test. *Data on microconida germination are means of four replication. Figures in the parenthesis are inhibition of conidial germination yGrowth in inhibition expressed as the ratio of radius of hyphal growth in the direction of the well with crude filtrate relative to the radius of hyphal growth in the direction of control well. Each value is the mean of four replication bacterial isolate. Figures in the parenthesis are square root transformed value
Bioassay in the green house
Efficacy of selected Pseudomonas isolates as seed treatment under green house conditions against F. oxysporum f.sp. ciceri: Effect of seed treatment with selected isolates of fluorescent Pseudomonas i.e., H-Pf5 and C7R12 was observed on seed germination and wilt incidence in chickpea under green house conditions (Table 3).
The seeds treated with of H-Pf5 and C7R12 showed 72.9 and 73.9% seed germination, respectively as compared to 48.7% in control. Rate of germination in seeds treated with both the isolates was significantly (p<0.05) different from that of control. Both the isolates were also able to reduce the wilt incidence (approx 50%) in comparison to control. The maximum of 47.9 and 52.7% disease inhibition was observed with application of H-Pf5 and C7R12, respectively after 120 days of sowing whereas, in control 100% of plants showed wilting (Table 3) after 120 days of sowing.
Effect of seed treatment on plant growth parameters: The seed treatment with both isolates of fluorescent Pseudomonas H-Pf5 and C7R12 greatly enhanced the plant growth in chickpea plants (p<0.05) (Fig. 1). The root length and shoot length of chickpea plants treated with isolates of H-Pf5 and C7R12 was significantly higher than the control treatment. Similarly, the dry weight in treated plants was also significantly (p<0.05) higher than the untreated plants (Fig. 1A-C).
Efficacy of selected Pseudomonas isolates as soil treatment under green house conditions against F. oxysporum f. sp. ciceri: Soil treatment with antagonists, H-Pf5 and C7R12 showed 78.6 and 78.5% seed germination as compared to control where only 45.8% seeds were germinated. Rate of germination in seeds treated with both the isolate was significantly (p≥0.05) different from that of control. There was no significant difference within the isolates (Table 4). Both the isolates were also able to reduce the wilt incidence in chickpea plants in comparison to control (p<0.05). The maximum wilt incidence 47.6% was observed after 120 days of plants in comparison to control (p<0.05).
| Table 3: | Effect of seed treatment with fluorescent Pseudomonas on the incidence of chickpea wilt in green house |
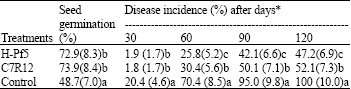 | |
| *With in the columns, means with a common lower case letter do not differ significantly (p = 0.05) according to fisher’s least significant difference test. Figures in the parenthesis are square root transformed value | |
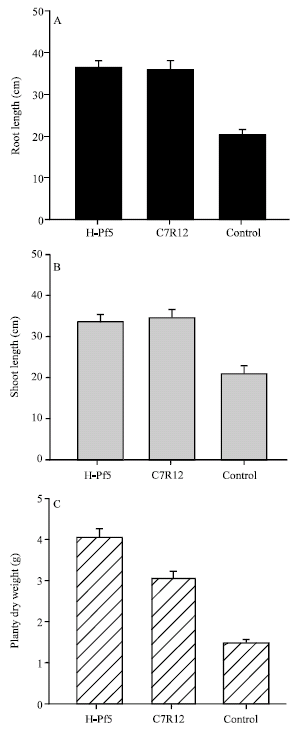 | |
| Fig. 1: | Effect of seed treatment with flourescent Pseudomonas isolates on the root length, shoot length and dry weight of chickpea plants in green house after 90 days of sowing |
The maximum wilt incidence 47.6% was observed after 120 days of sowing in plants treated with H-Pf5 and c7R12 in comparison to 96.7% in control plants (Table 4).
Effect of soil inoculation on plant growth parameters: The soil treatment with isolates of fluorescent Pseudomonas also greatly enhanced the plant growth in chickpea (Fig. 2).
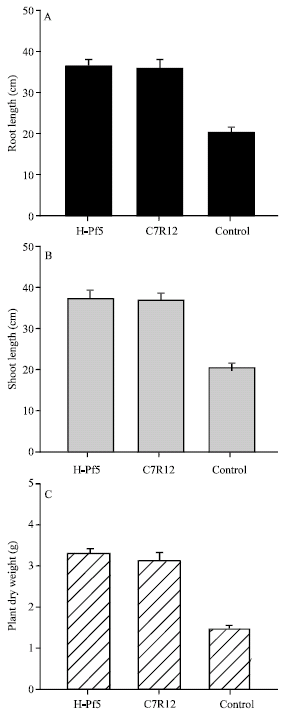 | |
| Fig. 2: | Effect of soil treatment with flourescent Pseudomonas isolates on the root length, shoot length and dry weight of chickpea plants in green house after 90 days of sowing |
The root length and shoot length in chickpea plants treated with isolates of H-Pf5 and C7R12 was significantly higher than the control treatment. Similarly the dry weight in treated plants was also much higher to that of untreated plants.
Bioassay under field conditions: Effect of fluorescent Pseudomonas isolates on wilt incidence was also recorded in field conditions.
| Table 4: | Effect of soil inoculation with selected isolates fluorescent Pseudomonas on incidence of chickpea wilt in green house conditions |
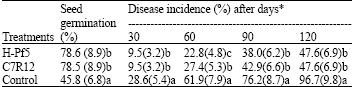 | |
| *With in the columns, means with a common lower case letter do not differ significantly (p = 0.05) according to Fisher’s least significant difference test. Figures in the parenthesis are square root transformed value | |
| Table 5: | Effect of seed inoculation with selected isolates of fluorescent Pseudomonas on incidence of chickpea wilt in field conditions |
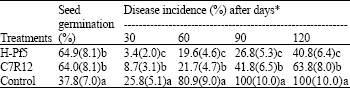 | |
| *With in the columns, means with a common lower case letter do not differ significantly (p = 0.05) according to Fisher’s least significant difference test. Figures in the parenthesis are square root transformed value | |
There was no significant difference seed germination percentage in seeds treated with both the antagonists. A maximum of 64.9 and 64.0% seeds germinated when treated with H-Pf5 and C7R12, respectively, where as only 37.8 % in case of control (Table 5). Both the isolates were highly effective in checking the wilting in comparison to control (p<0.05). However, the Indian isolate H-Pf5 was more effective than the French isolate C7R12. The difference in their biocontrol efficacy in field conditions was significant even after 30 days of sowing. The treatment with H-Pf5 showed 3.4% wilt incidence as compared to 8.7% with C7R12. However, in both the treatments, the disease incidence was significantly lower than that of the control. Even after 120 days of sowing, 40.8 and 63.8% wilting observed with the seed treatment with fluorescent Pseudomonas H-Pf5 and C7R12, respectively in comparison to 100 % wilting in control plots.
DISCUSSION
As it has been observed earlier that the rhizosphere environment may be helpful in selecting the effective antagonistic strain of bacteria in the same environment as where they will be used eventually (Weller, 1988; Edward et al., 1994; Marilley and Aragno, 1999), Thus, during the present studies, the major objective was to select an antagonistic bacterial isolate from rhizosphere and rhizoplane of chickpea and which can be effective against Fusarium wilt of chickpea under Indian conditions. Approximately 91% of 90 bacterial isolates from the chickpea rhizosphere inhibited in vitro growth of F. oxysporum ciceri in dual cultures. This appears to be a higher proportion than that inhibitory of P. megasperma Dreschsler f.sp. medicaginis obtained by Myatt et al. (1992) and F. oxysporum f. sp. ciceris by Landa et al. (1997) from a similar environment. Isolates were categorized into four categories based on their antagonistic activity, i.e., highly, moderately, least and non-antagonists. Maximum number of highly antagonistic isolates was obtained from rhizosphere and rhizoplane of healthy chickpea plants. Whereas, maximum number of non-antagonistic isolates were obtained from completely wilted chickpea plants. The isolate H-Pf5 selected in this study was isolated from rhizosphere soil of healthy chickpea plant and this supports the fact that the rhizosphere zone helps in selecting the antagonistic strains of bacteria from the natural environment as where they are being used eventually. The inhibition of hyphal growth of fungal isolate in dual cultures suggests the involvement of antibiotics and/or other antifungal substances by these bacteria. Adhikari et al. (2001) isolated three different Pseudomonas species from rhizosphere of rice and these significantly reduced the in vitro growth of two rice pathogens (Achyla klebsiana and Pythium spinosum) and proved to be effective biological control agents. Similarly Weller et al. (1985 and 2002) demonstrated that wheat roots grown in a take all suppressive soil yielded higher numbers of take all suppressive bacterial strains than those grown in take-all conducive soil. This is supported by fact that all bacterial isolates selected initially, there was no direct contact between fungal mycelium and bacterial colonies, so that the inhibition of fungal growth was due to substances that diffused into the agar medium. Reading the current studies the involvement of antifungal compounds produced by bacteria in the inhibition of fungal growth was confirmed by the ability of cell-free culture filtrates of bacteria to inhibit in vitro conidia germination and hyphal growth of F. oxysporum f. sp. ciceri. The concentration of culture filtrate of both the bacterial isolates influenced the mycelial growth inhibition of pathogen. The crude filtrate showed utmost antagonistic activity and this reduced with increase in the dilution of culture filtrate.
Results from seed and soil treatments are corroborated with the earlier studies that had also suggested that fluorescent Pseudomonas inhibited Fusarium wilt of flax caused by F. oxysporum f. sp. lini in vitro and in vivo and also increased emergence of radish and cucumber (Scher and Baker, 1980). Similarly during the current study seed and soil treatment under green house conditions significantly enhanced the growth of chickea plants. Bakker et al. (1990) established that fluorescent Pseudomonas sp. producing siderophores called pseudobactines and pyroverdines were very efficient competitors for iron and the competition for iron was one of the mechanism responsible for soil suppressiveness to Fusarium wilts (Scher and Baker, 1982; Latour et al., 2003). Fluorescent Pseudomonas during present studies isolates showed great potential in suppression of chickpea wilt and this suggests that these isolates can be used as attractive biological control agents of plant diseases caused by phytopathogenic fungi. Shekhar (2002) screened the chickpea rhizosphere competitive bacteria having biological control property and observed that P. fluorescens NBRI 1303 (ATCC 55939) was effective in suppressing plant pathogens, F. oxysporum f. sp. ciceri, Rhizoctonia bataticola and Pythium sp. in chickpeas and recommended the use of purified bacterial strain as active agent for biocontrol compositions. He also observed the enhanced plant growth and yield of chickpea and the production of antibiotics against phytopathogenic fungal diseases. Kloepper et al. (1980) reported the stimulating effect of P. putida WCS 358 on potato growth and suppressive effect on flax wilt. Similarly bacterisation of carnation rots by Pseudomonas sp. strain WCS417r had been found to reduce number of diseased plants. Scher and Baker (1982) isolated strains of fluorescent Pseudomonas spp. from mycelial mats buried in suppressive soil and demonstrated the ability of some strains to induce suppressive ness in a conducive soil. The use of fluorescent Pseudomonas isolate, C7R12 to control Fusarium wilts of different plant species had already been described by Alabouvette et al. (1996, 1998) and Siddiqui and Ehteshamul (1999). It has also been observed that seed bacterization with fluorescent Pseudomonas enhanced the synthesis of flavonoid-like compounds and played a role in induction of systemic resistance as well as improving nodulation (Nowak et al., 1999; Goel, 2001; Nguyen, 2003; Saikia et al., 2003). This study provided an initial assessment of the potential of rhizoplane and rhizosphere bacteria associated with chickpea in Punjab (India) to control wilt of chickpea and promote plant growth under controlled as well as natural conditions. Both the isolates appeared to be the most promising biocontrol agents against chickpea wilt. But further we need to investigate the improved methods of antagonists delivery and its establishment in field.
REFERENCES
- Adhikari, T.B., C.M. Joseph, G. Yang, D.A. Phillips and L.M. Nelson, 2001. Evaluation of bacteria isolated fromrice for plant growth promotion and biological control of seedling disease of rice. Can. J. Microbiol., 47: 916-924.
Direct Link - Bagnasco, P., L. de la Fuente, G. Gualtieri, F. Noya and A. Arias, 1998. Fluorescent Pseudomonas spp. as biocontrol agents against forage legume root pathogenic fungi. Soil Biol. Biochem., 30: 1317-1322.
Direct Link - De Boer, M., I. van der Sluis, L.C. van Loon and P.A.H.M. Bakker, 1999. Combining fluorescent Pseudomonas spp. strains to enhance suppression of Fusarium wilt of radish. Eur. J. Plant. Pathol., 105: 201-210.
CrossRefDirect Link - De Weger, L.A., A.J. van der Bij, L.C. Dekkers, M. Simons, C.A. Wijffelman and B.J.J. Lugtenberg, 1995. Colonization of the rhizosphere of crop plants by plant-beneficial Pseudomonads. FEMS Microbiol. Ecol., 17: 221-228.
CrossRefDirect Link - Duijff, B.J., D. Pouhair, C. Olivain, C. Alabouvette and P. Lemanceau, 1998. Implication of systemic induced resistance in the suppression of Fusarium wilt of tomato by Pseudomonas fluorescens WCS417r and by non-pathogenic Fusarium oxysporum Fo47. Eur. J. Plant. Pathol., 104: 903-910.
Direct Link - Duijff, B.J., G. Recorbet, P.A.H.M. Bakker, J.E. Loper and P. Lemanceau, 1999. Microbial antagonism at the root level is involved in the suppression of Fusarium wilt by the combination of nonpathogenic Fusarium oxysporum Fo47 and Pseudomonas putida WCS358. Phytopathology, 89: 1073-1079.
Direct Link - Hamdan, H., D.M. Weller and L.S. Thomashow, 1991. Relative importance of fluorescent siderophores and other factors in biological control of Gaeumanomyces graminis var triticii by Pseudomonas fluorescens 2-79 and M 4-80. Applied Environ. Microbiol., 57: 3270-3277.
Direct Link - King, E.O., M.K. Ward and D.E. Raney, 1954. Two simple media for the demonstration of pyocyanin and fluorescin. Transl. Res., 44: 301-307.
PubMedDirect Link - Kloepper, J.W., J. Leong, M. Teintze and M.N. Schroth, 1980. Enhanced plant growth by siderophores produced by plant growth-promoting rhizobacteria. Nature, 286: 885-886.
CrossRefDirect Link - Landa, B.B., A. Hervas, W. Bettiol and R.M. Jimenez-Diaz, 1997. Antagonistic activity of bacteria from the chickpea rhizosphere against Fusarium oxysporum f. sp. Ciceris. Phytoparasitica, 25: 305-318.
Direct Link - Latour, X., S. Delorme, P. Mirleau and P. Lemanceau, 2003. Identification of traits implicated in the rhizosphere competence of fluorescent Pseudomonads: Description of a strategy based on population and model strain studies. Agronomie, 23: 39-405.
CrossRef - Moulin, F.P., C. Lemanceau and C. Alabouvette, 1994. Control by Fluorescent Pseudomonas of Pythium aphanidermatum Root Rot, Responsible for Yield Reduction in Soil less Culture of Cucumber. In: Improving Plant Productivity with Rhizosphere Bacteria, M.H. Ryder, P.M. Stephens and G.D. Bowen (Eds.). Csrio, Adelaide, pp: 47-50.
- Nguyen, C., 2003. Rhizodeposition of organic C by plants: Mechanisms and controls. Agronomie, 23: 375-396.
CrossRef - Nowak, T.B., N. Chaney, J.S. Wing, S.J. Gould and J.E. Loper, 1999. Characterization of the pyoluteorin biosynthetic gene cluster of Pseudomonas fluorescens Pf-5. J. Bacteriol., 181: 2166-2174.
Direct Link - Ongena, M., F. Daayf, P. Jacques, P. Thonart and N. Benhamou et al., 1999. Protection of cucumber against Pythium root rot by fluorescent pseudomonads: Predominant role of induced resistance over siderophores and antibiosis. Plant Pathol., 48: 66-76.
Direct Link - Raaijmakers, J.M. and D.M. Weller, 1998. Natural plant protection by 2,4-diacetylphloroglucinol-producing Pseudomonas spp. in take-all decline soils. Mol. Plant-Microbe Interactions, 11: 144-152.
Direct Link - Saikia, R., T. Singh, R. Kumar, J. Srivastava, A.K. Srivastava, K. Singh and D.K. Arora, 2003. Role of salicylic acid in systemic resistance induced by Pseudomonas fluorescens against Fusarium oxysporum f. sp. ciceri in chickpea. Microbiol. Res., 158: 203-213.
CrossRefDirect Link - Scher, F.M. and R. Baker, 1982. Effect of Pseudomonas putida and a synthetic iron chelator on induction of soil suppressiveness to Fusarium wilt pathogens. Phytopathology, 72: 1567-1573.
CrossRefDirect Link - Sharma, A. and B.N. Johri, 2003. Combat of iron-deprivation through a plant growth promoting fluorescent Pseudomonas strain GRP3A in mung bean (Vigna radiata L. Wilzeck). Microbiol. Res., 158: 77-81.
CrossRef - Srivastava, A.K., T. Singh, T.K. Jana and D.K. Arora, 2001. Induced resistance and control of charcoal rot in Cicer arietinum (chickpea) by Pseudomonas fluorescens. Can. J. Bot., 79: 787-795.
Direct Link - Weller, D.M., J.M. Raaijmakers, B.B.M. Gardener and L.S. Thomashow, 2002. Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu. Rev. Phytopathol., 40: 309-348.
CrossRefPubMedDirect Link - Whipps, J.M., 2001. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot., 52: 487-511.
PubMedDirect Link - Weller, D.M., 1988. Biological control of soilborne plant pathogens in the rhizosphere with bacteria. Annu. Rev. Phytopathol., 26: 379-407.
CrossRefDirect Link








