ABSTRACT
Two strains of rhizobia, Bradyrhizobia sp. Aust 13c and Tel 8, which have dissimilar characteristics (antibiotic sensitivity and PCR-RFLP profiles) were tested for nodulation of Acacia mangium. Nodulation was carried out on in vitro seedlings growing in Broughton-Dilworth medium supplemented with different levels of nitrogen at 7, 18 and 180 ppm. Control seedlings were grown in nitrogen free nutrient solution. A total of 70 nodules were harvested, with DNA extraction from bacteria in nodules carried out and PCR-RFLP generated. A. mangium seedlings inoculated with a mixture of equal volumes of the two nitrogen fixing bacteria strains, Aust 13c and Tel 8 had 35.7% of the nodules infected only with Aust 13c, 14.3% infected singly with Tel 8 and none doubly infected with both Aust 13c and Tel 8 in medium without nitrogen. Higher levels of nitrogen were found to decrease the percentage of single and increase the percentage of double infection. Our results suggest that increasing nitrogen levels altered the promiscuity of the legume-rhizobia relationship.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.941.948
URL: https://scialert.net/abstract/?doi=ajps.2007.941.948
INTRODUCTION
Nitrogen is found in abundance in the atmosphere. However, plants cannot convert atmospheric nitrogen into useful organic forms for plant growth and often have to engage the help of rhizobial bacteria to reduce the dinitrogen into usable forms. A substantial part of the world’s supply of organic nitrogen is fixed via the symbiosis between rhizobial bacteria and leguminous host plants. The Fabaceae, or legumes are both important ecologically and agriculturally as they are responsible for a large part of the global flux of nitrogen from atmospheric nitrogen to fixed forms like ammonia, nitrate and organic compounds (Young and Haukka, 1996). Such efforts are often accomplished with the ability of bacteria to fix nitrogen, nodulate the legume host and compete with other indigeneous bacteria present in the soil for infection of the roots. To encourage symbiosis, rhizobia are frequently used to inoculate soil or seed. However the need for inoculation and the magnitude of response to the applied rhizobia may be determined by bacterial type as well as many factors which include salinity, drought or excessive use of pesticides (Saleena et al., 2001), as well as the levels of nitrogen in the soil.
In agricultural systems, fields are often treated with nitrogen fertilizers to improve crop yield. Legume plants will grow well with exogenous nitrogen applications, especially in nitrogen deficient soils. However, legumes supplied with high nitrogen levels also show a reduction in the number of nodules formed (West et al., 2002; Simms and Taylor, 2002). The effects on the types of partnerships and the choice of rhizobial partners to associate with given a change in the host’s external environment have not been studied extensively. Simms and Taylor (2002) have suggested that legumes may be able to restrict associations to particular groups of bacteria and adjust the levels of nodulation in response to the availability of nitrogen from external sources. Thus, this project seeks to compare the growth relationships in the legume host Acacia mangium inoculated with Aust 13c and Tel 8 singly or doubly in four different levels of nitrogen. The objectives are to observe: 1) the effects of nitrogen and inocula on the growth (the height and number of leaves) of Acacia mangium seedlings, 2) the effects of nitrogen and inocula on nodulation (the number of nodules formed and 3) the effect of nitrogen on diversity of bacterial partners in seedlings inoculated with mixed cultures.
MATERIALS AND METHODS
Inoculum: Bacterial strains, Bradyrhizobium sp. Aust 13c and Tel 8 were recovered from root nodules of Acacia mangium. Isolate Aust 13c was isolated from a host plant growing on clay-sand soil in Daintree, Australia and selected previously for its high nodulation efficiency (Galiana et al., 1990; Lesueur et al., 1993; Galiana et al., 1994). Tel 8 was isolated from nodules of the host growing in clay soil in Sabah Malaysia (Martin-Laurent et al., 1999). Bacterial strains were grown for one week in liquid mannitol yeast extract (YM) medium (mannitol 1%, K2HPO4 0.05%, MgSO4.7H2O 0.2%, NaCl 0.01%, yeast extract 0.05%; pH 6.8) (Vincent, 1970) at 30°C with agitation. The concentration of the bacterial inoculant was 109 cells ml-1 as determined with a counting chamber using a phase contrast microscope.
Growth of acacia seedlings: Experiments were carried out in the laboratories and plant growth rooms at the National Institute of Education, Nanyang Technological University, in Singapore. Acacia mangium seeds were bought from a commercial seed company (Seedworld, Australia). The seeds were scarified by immersion for two minutes in boiling water at 100°C. They were then surface sterilized using 30% hydrogen peroxide for 10 min. The seeds were rinsed five times with sterile deionised water and germinated aseptically on 1% tap water agar. Two week old seedlings were individually transferred into sterile assemblies set-up as follows: The roots of the seedling were inserted through a slit in a filter paper support and the seedling on the support was placed into a boiling test tube containing 5 mL of a modified Broughton-Dilworth (BD) nutrient solution (CaCl2 147 ppm, K2PO4 68 ppm, Fe(III) EDTA 3.35 ppm, MgSO4.7H2O 61.6 ppm, K2SO4 43.5 ppm, MnSO4 0.17 ppm, H3BO3 0.123 ppm, ZnSO4 0.144 ppm, CuSO4 0.05 ppm, CoSO4 0.028 ppm, Na2MoO2 0.024 ppm; pH 6.7) (Broughton and Dilworth, 1971) with no nitrogen (N), 7, 18 and 180 ppm N. Nitrogen was supplied in the form of KNO3. The seedlings were inoculated after transfer with either 2 mL of sterile deionised water for control or 2 mL of one-week old bacterial cultures. The seedlings were transferred to fresh nutrient solutions of the corresponding nitrogen concentration after 5 weeks. The in vitro grown seedlings were maintained in a growth room at 28±2°C under 12 h day-1 photoperiod at 4.18x10±2 and 3.87x10±2 μmol m-2 sec-1 produced from cool white fluorescent lamps.
Design of experiment: The seedlings were grown under four different levels of N treatment: (1) N-free nutrient solution, (2) with 7 ppm N, (3) with 18 ppm N and (4) with 180 ppm N. Within each N treatment, the seedlings were either inoculated with 1) deionised water (control), (2) only Aust 13c, (3) only Tel 8 and (4) with a 1:1 mixture of Aust 13c and Tel 8. There were ten replicates designated per treatment organized in a completely randomized design, except for the control treatments which had only three seedlings due to insufficient number of seeds successfully germinated. Seedlings were harvested 2 months after inoculation to determine the number of nodules per seedling, the number of leaves per seedling and the increase in the height of seedling. The heights of the plants were measured without taking them out of their tubes to avoid contamination. Data was statistically analysed by the ANOVA procedure (General Linear Model (GLM) and one-way ANOVA) using MINITAB programme (Ryan et al., 1985). The results are analysed such that if p-value in GLM is significant (p<0.05), an interaction plot is used to display interactions between factors; if the p-value is not significant (p>0.05), the analysis will proceed with one way ANOVA.
Isolation of rhizobia from nodules of plants inoculated with mixed bacterial inoculant: Two months after inoculation, all nodules from the replicates for each of the four N treatments were harvested and pooled. A total of 20 nodules were randomly collected from each treatment: 0 ppm N, 7 and 18 ppm N; with the exception of 10 nodules being collected from the 180 ppm N treatment due to the limited number of nodules formed. The 70 nodules were surfaced sterilized in 70% ethanol, followed by 20% household bleach (1% sodium hypochlorite) and rinsed three times with sterile deionised water. For DNA extraction, each nodule was crushed aseptically in a microcentrifuge tube with 20 μL of sterile deionised water. To this suspension, 300 μL of nodule extraction buffer (100 mM Tris; 1.4M NaCl; 20 mM EDTA; 2% CTAB) was added. The solution was vortexed, incubated at 65°C for 1 h and centrifuged at 13,000 rpm for 5 min. 250 μL of the supernatant was transferred to a fresh tube, avoiding the pellet. An equal volume of phenol:chloroform (1:1) was added and the contents were mixed vigorously and centrifuged at 13,000 rpm for 10 min. The top aqueous layer of 230 to 250 μL was transferred to a fresh tube and 1/10 volume of precipitating CTAB (CTAB 10%, NaCl 0.7M) was added. The contents were mixed and incubated for 10 min at 65°C. An equal volume of chloroform:isoamyl alcohol (24:1) was added, the contents mixed and the tube was centrifuged at 13,000 rpm for 10 min. Again, the aqueous layer was transferred to another fresh tube and 1/10 volume of 3M NaOAc and 2 volumes of cold absolute alcohol was added. The tube was incubated at 20°C for 20 min, followed by centrifuging at 13,000 rpm for 10 min. The supernatant was poured away, leaving only the DNA pellet. The DNA pellet was washed in 50 μL of 70% ethanol and centrifuged at 13,000 rpm for 3 min. The ethanol was removed and the pellet was left to air-dry. The DNA pellet was re-suspended in 30 μL TE (10 mM Tris; 0.1 mM EDTA; pH 8) and centrifuged at 13,000 rpm for 1 min. Extracted DNA was stored at -20°C for subsequent use in the Polymerase Chain Reaction (PCR). Determination of bacterial strains was carried out by PCR-restriction fragment length polymorphism (PCR-RFLP) using a modified protocol from Martin-Laurent et al. (2000).
Analysis of bacterial isolates by PCR-RFLP: The intergenic spacer (IGS) between the 16S and 23S rRNA genes of the ribosomal operon was amplified. The sequences of the primers for the amplification were derived from the 16S and 23S rRNA genes flanking the targeted IGS: 5’CCGGGTTTCCCCATTCGG3’ (Primer 1) and 5’GGAGAGTTAGATCTTGGCTCAG3’ (Primer 2). Amplifications were performed using a PTC-100TM programmable thermal cycler with a heated lid (MJ Research Inc., USA) in 50 μL reaction volumes, each containing 2 μL of DNA extract, 1xMgCl2-free reaction buffer (Invitrogen, Singapore), 3 mM MgCl2, 200 μM dNTPs, 0.2 μM Primer 1, 0.2 μM Primer 2 and 2 units of DNA polymerase (Invitrogen, Singapore). DNA amplification was carried out at 95°C for 3 min for one cycle, followed by 35 cycles of amplification at 95°C for 1 min, 55°C for 2 min and 72°C for 3 min. A final extension time at 72°C for 10 min was included. Amplification products were cut with the restriction enzyme CfoI as recommended by the manufacturer (Vivantis, Singapore). Restriction fragments were separated by electrophoresis on a 2.5% agarose gel and stained with ethidium bromide (1 μg L-1) for 30 min and destained for 10 min. Gels were photographed using an EagleEye Image Analyser (Stratagene, USA).
RESULTS
Effect of nitrogen and inocula on nodulation: The number of nodules differed significantly (one way ANOVA, p = 0.001) under the four different nitrogen treatments and also under the four inoculation treatments (one way ANOVA, p = 0.005). For different nitrogen levels, the mean numbers of nodules±standard error (SE) were: 2.97±0.67, 4.92±0.82 and 4.07±0.93, 0.77±0.25 in 0, 7, 18 and 180 ppm N, respectively (Fig. 1A). The mean number of nodules formed under mixed inoculum was 4.62±0.82 nodules which was higher than those formed under single inoculation of Aust 13c (3.52±0.80 nodules) and Tel 8 (2.45±0.43 nodules) respectively (Fig. 1B). Inoculation with Aust 13c resulted in more nodules than with Tel 8 although numbers were not significantly different. Similarly, mixed inocula appeared to enhance nodulation more than inoculation with a single isolate (the difference between Tel 8 and mixed inoculum was statistically significant).
Effect of nitrogen and inocula on growth of A. mangium: The results showed that the height of plants differed significantly among the four nitrogen and four inoculation treatments (one way ANOVA, p = 0.028). As the heights showed significant differences between the various nitrogen and inocula treatments under GLM analysis, an interaction plot was used to display the mean heights of plants (Fig. 2). A comparison across the mean heights showed that plants under 7 ppm N, 18 ppm N and 180 ppm N treatments all displayed the same mean height when inoculated singly with Aust 13c. Plants under the 180 ppm N treatment yielded the highest mean when inoculated singly with Tel 8. Plants under the 7 ppm N treatment were found with the highest mean height when inoculated with both Aust 13c and Tel 8. And plants under the 18 ppm N treatment were found with the highest mean height when no nitrogen was added.
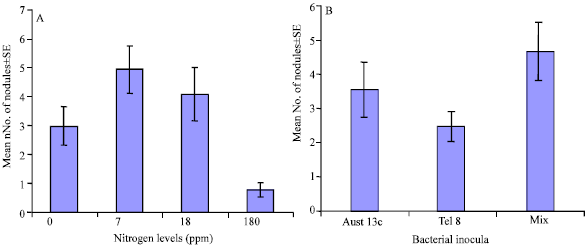 | |
| Fig. 1: | (A). Effect of nitrogen levels on overall nodulation in plants after 8 weeks. (B) Effect of different bacterial treatments on overall nodulation in plants after 8 weeks. Values are means±SE of the number of plants in each treatment. Vertical bars represent standard error |
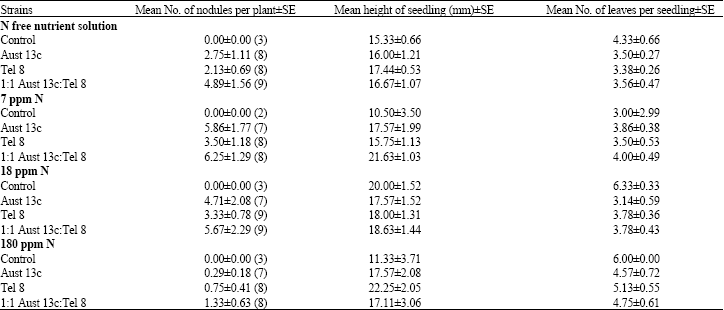
No significant difference was found for the number of leaves developed between the seedlings under various inoculation treatments (one way ANOVA, p = 0.569) but there is a significant difference (ANOVA, p = 0.031) under the different nitrogen treatments (Table 1).
Effect of nitrogen on diversity of bacterial partners in seedlings inoculated with mixed cultures: Distinct PCR-RFLP profiles were obtained from Aust 13c, Tel 8 and mixed inoculum DNA (Fig. 3). The proportion of nodules found to be infected by Aust 13c decreased from 71.4 to 33.3% as the amount of nitrogen increased in the nutrient solution (Table 2). In contrast, the proportion of nodules infected by Tel 8 increased from 28.5 to 41.6% as nitrogen levels increased. Similarly, the percentage of nodules showing double infection by both Aust 13c and Tel 8 also increased with increasing levels of nitrogen treatment from 0 to 25%. Only a total of 10 nodules were formed in treatment with 180 ppm N. Although DNA was extracted from these nodules, extremely faint or no PCR-RFLP profiles could be obtained and results could not be scored. Hence, no data is presented for this treatment.
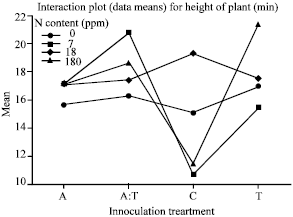 | |
| Fig. 2: | Interaction plot for mean height of plants against various nitrogen and inoculation treatments. Inoculation treatment is as follows: 1) A: Aust 13c 2) A:T: Aust 13c:Tel 8 3) C: Control 4) T: Tel 8 |
| Table 1: | Effects of nitrogen on nodulation and growth of Acacia mangium seedlings 8 weeks after inoculation. Values are means±SE of the number of replicate plants in each treatment (number of replicates in parentheses) |
 | |
| Table 2: | Effects of nitrogen on the diversity of bacterial partners within nodules of Acacia mangium |
 | |
| a: Proportion of nodules infected in parenthesis | |
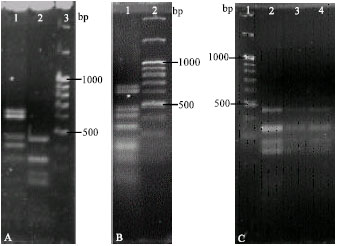 | |
| Fig. 3: | PCR-RFLP profiles obtained from (A) Aust 13c, Tel 8 (lanes 1 and 2, respectively; lane 3: 100 bp molecular weight marker), (B) mixed inocula (lane 1; lane 2: 100 bp molecular weight marker) and (C) DNA from nodules treated with mixed inocula at 18 ppm N showing single infection with Tel 8 (lane 1: 100 bp molecular weight marker; lanes 2-4: nodule 1, nodule 2, nodule 3, respectively) |
DISCUSSION
The results indicate that the level of nitrogen has a significant effect on plant growth in terms of height of plant and the number of leaves and nodules developed, with higher nitrogen concentrations yielding taller plants with more leaves. This is not unexpected as nitrogen is an important element for plant growth and development and indeed Acacia mangium saplings that were not supplied with nitrogen were shown to have very poor development even when nodulated with effective nitrogen-fixing Bradyrhizobium species (Weber et al., 2007). Improved nitrogen nutrition has also been shown to stimulate increased root and plant growth (Gentili and Huss-Danell, 2001; Indieka and Odee, 2005) as well as contribute to tolerance to stresses, for example, drought and salinity (Saneoka et al., 2004; Tabatabaei, 2006). Present results also show that all inoculated Acacia mangium plants in general (except for control plants at 18 ppm N) had higher mean heights than non-treated plants, whether inoculated with a single strain or doubly inoculated with two strains of Bradyrhizobium. This suggests general increase in nitrogen fixation caused by nodule formations on plant roots which promote plant growth. Similarly, the Bradyrhizobium strains Aust 13c and Tel 8 used in the present experiments have been previously demonstrated to be very effective in nodulating and fixing nitrogen for A. mangium and improving its growth both in field, hydroponics as well as aeroponics trials (Martin-Laurent et al., 1997, 1999, 2000).
In this study, the mean number of nodules decreased as nitrogen levels increased. Other studies have likewise recorded decreasing nodulation rates with increasing soil nitrogen availability (Caetano-Anolles and Gresshoff, 1991; Lang et al., 1993; Rubio et al., 1999; Singleton and van Kessel, 1987; Simms and Taylor, 2002; Thomas et al., 2000). Although Indieka and Odee (2005) showed that nodulation in Sesbania sesban was relatively tolerant to high nitrogen treatments and concentrations of up to 100 mg L-1 was essential in stimulating early and effective nitrogen-fixing symbiosis, concentrations of nitrogen at 200 mg L-1 and above depressed or totally inhibited nodule formation. Similarly, although plants responded to increased nitrogen levels by improved growth of root and shoot systems, it was demonstrated that inhibition of nodulation by nitrogen was systemic for both nodule number and nodule biomass in the root system of Hippophae rhamnoides (Gentili and Huss-Danell, 2001). High nitrogen levels were found to completely inhibit nodulation in some plants of this species. The result we have in the present investigation is congruent with the partner-choice theory on nitrogen fixation proposed by Simms and Taylor (2002), where the benefits of nodulation to the plant decline with increasing abundance of reduced nitrogen that the plant can obtain directly from the soil. Hence, these authors suggested that certain legumes could essentially control resource allocation to nitrogen fixation by adjusting levels of nodulation in response to the availability of nitrogen. It may also imply that nitrogen fixation by rhizobia is generally less important to the plant as nitrogen becomes less limiting to plants (West et al., 2002). The detailed mechanisms for nitrogen inhibition however are poorly understood and Parsons et al. (1993) and Baker et al. (1997) have proposed that the concentration of reduced nitrogen compounds (such as amino acids) may be involved in an internal feedback regulatory process from phloem to nodule.
Plants have a way of benefiting from obtaining as much nitrogen as possible at the lowest plausible cost in terms of inducing nodule formation or sanctioning resources for instance, photosynthates to nodules according to Simms and Taylor (2002). In other words, as long as the nitrogen level in the medium is sufficient for the plant, it will not pay the extra energetic price of conducting signal exchange with rhizobia, producing leghaemoglobin and forming nodules. Similarly in the present investigation, it also appears that under high nitrogen availability, plants do not practice unlimited infection by even compatible rhizobia such as Aust 13c or Tel 8. Aust 13c has been demonstrated to be a highly infective, competitive and effective strain of Bradyrhizobium (Martin-Laurent et al., 1999 and 2000) and the results here confirm it induces greater nodulation in A. mangium than the strain Tel 8. Interestingly, the number of nodules formed when a mixture of two strains was used for inoculation was higher than when either Aust 13c or Tel 8 was used singly. These preliminary results could suggest that Aust 13c and Tel 8 could work synergistically in enhancing nodulation in A. mangium and dual inoculations with high nodulating Bradyrhizobium strains such as Aust 13c may benefit from being “coupled” with lower nodulating strains, for example Tel 8. Having said this, it will only hold true if nitrogen fixation can be shown to be effective in these nodules, as nodule numbers are not always synonymous with higher nitrogen fixing capacity. In Parasponia andersonii, strains of Bradyrhizobium in paired or mixed inoculations also appeared to increase nodulation and frequency of dual occupancy in nodules compared to inoculations with single strains (Trinick and Hadobas, 1989). The less competitive strains were dual occupiers with the more competitive strains.
Whereas there have been reports on the competitiveness of rhizobia strains and dual infections of nodules by multiple strains of rhizobia (Lindermann et al., 1974; Moawad and Schmidt, 1987; Sato et al., 1988; Asad et al., 1991; Trinick and Hadobas, 1989; Streit et al., 1995), as far as we know, there are none that report on the effect of nitrogen on the number of bacteria partners in the legume-rhizobia partnership. The results of our study show that the proportion of nodules infected solely with Aust 13c decreased whereas the number of nodules infected solely with Tel 8 increased with increasing nitrogen content. In addition, the percentage of nodules exhibiting double infection with both Aust 13c and Tel 8 of the nodules similarly increased with increasing nitrogen levels as well. This is interesting as the plants appeared to be more discriminate with the partnerships they form, with Aust 13c being the partner of choice instead of Tel 8 at lower nitrogen levels. The host plants were more promiscuous with the diversity of partnerships forged as nitrogen levels increased. This was similarly suggested by Simms and Taylor (2002) that nod factor discrimination may be subject to changes in soil nitrogen levels. Nod factors are released by rhizobia bacteria and play a role in host-bacteria recognition as these molecules induce root hair curling and nodule formation. In low levels of nitrogen, the plants appeared to have selected Aust 13c possibly because of its stronger ability to fix nitrogen than Tel 8. Elevating the nitrogen levels surprisingly resulted in increasing nodules with Tel 8 bacteria as well as mixed dual inocula. The preliminary data suggest that perhaps the plant host may possess mechanisms that would allow it to evaluate and choose partners and this operates stringently at limiting nitrogen levels. However, at enhanced nitrogen levels, this mechanism seems to be less accurate and less effective bacteria could benefit by occurring with beneficial bacteria.
The proportion of nodules hosting double strains of Bradyrhizobium also showed an upward trend as nitrogen levels increased which may be indicative that higher nitrogen levels favour dual or mixed infections. Nevertheless, the number of nodules examined in the current study is limited and more data needs to be obtained to confirm this trend. Furthermore, as this experiment was done under artificial plant growth conditions in the laboratory, the level of incidences of double infections may not be reflective of what occurs in the field in soil with the presence of more rhizobia (Lindemann et al., 1974). In the present study, 14 to 25% double infections of nodules were observed. This is quite similar to the frequencies reported by Sato et al. (1988) on experiments with in vitro seedlings of soybean, where 13% of dual infected nodules were obtained when plants were challenged with two strains of Bradyrhizobium japonicum, using Norris and Date medium which is N-free. Incidence of double- and indeed triple-strain occupancy of nodules has also been reported to range from 12 to 32% in controlled glasshouse experiments (Moawad and Schmidt, 1987; Asad et al., 1991) showing again a range of frequencies corresponding closely to our findings reported here. In contrast, an extremely high frequency of mixed-occupancy nodules up to 70% were revealed in some laboratory studies of crop legumes (Denison, 2000).
In conclusion, plants become less dependent on association with the appropriate bacteria at high levels of nitrogen because nitrogen is in abundance and not a limiting resource. Not only do nodule numbers decline but the nature of the partnerships the plants form also changes. Plants display promiscuity in partner choice and appear to be less discriminating in the symbiotic associations they form.
The implications of the study confirm that increasing nitrogen levels through excessive use of nitrogen fertilizers is likely to be detrimental to nodulation in legumes. In addition, the results also indicate that the effects of applications of elite strains of rhizobia in agriculture and forestry (which involve additional costs) may be reduced with increasing nitrogen levels as specificity with host decreases. Hence, the preliminary evidence here suggests that application of nitrogenous fertilizers to soils in agricultural and forest systems together with use of elite strains of rhizobia need to be more carefully managed to ensure maximum benefit from the interactions of the symbionts.
ACKNOWLEDGMENTS
The authors thank the National Institute of Education, Nanyang Technological University, Singapore for financial support for this research. The authors also gratefully acknowledge the use of the bradyrhizobium cultures, Aust13c and Tel8, which were originally supplied by CIRAD-FORET, France.
REFERENCES
- Asad, S., K.A. Malik and F.Y. Hafeez, 1991. Competition between inoculated and indigenous Rhizobium/Bradyrhizobium sp. strains for nodulation of grain and fodder legumes in Pakistan. Biol. Fertility Soils, 12: 107-111.
Direct Link - Baker, A., G.F. Hill and R. Parsons, 1997. Evidence for N feedback regulation of N2 fixation in Alnus glutinosa L. J. Exp. Bot., 48: 67-74.
Direct Link - Denison, R.F., 2000. Legume sanctions and the evolution of symbiotic cooperation by rhizobia. Am. Nat., 156: 567-576.
Direct Link - Galiana, A., Y. Prin, B. Mallet, G.M. Ghahona, M. Poitel and H.G. Diem, 1994. Inoculation of Acacia mangium with alginate beads of containing Bradyrhizobium strains under field conditions: Long term effect on plant growth and persistence of the introduced strain in soil. Applied Environ. Microbiol., 60: 3974-3980.
- Gentili, F. and K. Huss-Danell, 2002. Phosphorus modifies the effects of nitrogen on nodulation in split-root systems of Hippophae rhamnoides. New Phytologist, 153: 53-61.
Direct Link - Indieka, S.A. and D.W. Odee, 2005. Nodulation and growth response of Sesbania sesban (L.) Merr. To increasing nitrogen (ammonium) supply under greenhouse conditions. Afr. J. Biotechnol., 4: 57-60.
Direct Link - Lang, P., R. Martin and M.P. Golvano, 1993. Effect of nitrate on carbon metabolism and nitrogen fixation in root nodules of Lupinus albus. Plant Physiol. Biochem., 31: 639-648.
Direct Link - Lesueur, D., H.G. Diem, M. Dianda and C. Le-Roux, 1993. Selection of Bradyrhizobium strains and provenance of Acacia mangium and Faidherbia albida: Relationship with their tolerance to acidity and aluminium. Plant Soil, 149: 159-166.
Direct Link - Martin-Laurent, F., S.K. Lee, F.Y. Tham, J. He, H.G. Diem and P. Durand, 1997. A new approach to enhance growth and nodulation of Acacia mangium through aeroponic culture. Biol. Fertility Soils, 25: 7-12.
Direct Link - Martin-Laurent, F., M. Fremont, S.K. Lee, F.Y. Tham, Y. Prin, T.K. Tan and H.G. Diem, 1999. Effect of inoculation with selected Bradyrhizobium sp. on the survival and growth of Acacia mangium saplings after 20 months in the field. J. Trop. Forest Sci., 11: 470-483.
Direct Link - Parsons, R., A. Stanforth, J.A. Raven and J.I. Sprent, 1993. Nodule growth and activity may be regulated by a feedback mechanism involving phloem nitrogen. Plant Cell Environ., 16: 125-136.
Direct Link - Rubio Arias, H.O., L. De-la-Vega, O. Ruiz and K. Wood, 1999. Differential nodule response and biomass yield of Alexandria clover as affected by levels of inorganic nitrogen fertilizer. J. Plant Nutr., 22: 1233-1239.
Direct Link - Saleena, L.M., P. Loganathan, S. Rangarajan and S. Nair, 2001. Genetic diversity of Bradyrhizobium strains isolated from Arachis hypogaea. Can. J. Microbiol., 47: 118-122.
Direct Link - Saneoka, H., R.E.A. Moghaeib, G.S. Premachandra and K. Fujita, 2004. Nitrogen nutrition and water stress effects on cell membrane stability and leaf water relations in Agrostis palustris Huds. Environ. Exp. Bot., 52: 131-138.
Direct Link - Simms, E.L. and D.L. Taylor, 2002. Partner choice in nitrogen-fixation mutualisms of legumes and rhizobia. Integr. Comp. Biol., 42: 369-380.
CrossRefDirect Link - Singleton, P.W. and C. Van-Kessel, 1987. Effect of localized nitrogen availability to soybean half-root systems on photosynthate partitioning to roots and nodules. Plant Physiol., 83: 552-556.
Direct Link - Streit, W., L. Botero, D. Werner and D. Beck, 1995. Competition for nodule occupancy on Phaseolus vulgaris by Rhizobium etli and Rhizobium tropici strains can be efficiently monitored in ultisol during the early stages of growth using a constitutive GUS gene fusion. Soil Biol. Biochem., 27: 1075-1081.
Direct Link - Tabatabaei, S.J., 2006. Effects of salinity and N on the growth, photosynthesis and N status of olive (Olea europaea L.) trees. Sci. Hortic., 108: 432-438.
Direct Link - Thomas, R.B., M.A. Bashkin and D.D. Richter, 2000. Nitrogen inhibition of nodulation and N2 fixation of a tropical N2-fixing tree (Gliricidia sepium) grown in elevated atmospheric CO2. New Phytol., 145: 233-243.
Direct Link - Weber, J., F.Y. Tham, A. Galiana, Y. Prin, M. Ducousso and S.K. Lee, 2007. Effects of nitrogen source on the growth and nodulation of Acacia mangium in aeroponic culture. J. Trop. Forest Sci., 19: 103-112.
Direct Link - West, S.A., E.T. Kiers, E.L. Simms and R.F. Denison, 2002. Sanctions and mutualism stability: Why do rhizobia fix nitrogen? Proc. Royal Soc. Lond. Bull., 269: 685-694.
Direct Link - Peter, J., W. Young and K.E. Haukka, 1996. Diversity and phylogeny of rhizobia. New Phytol., 133: 87-94.
CrossRefDirect Link








