ABSTRACT
A chia (Salvia hispanica L.) trial in the Santa Elena Peninsula of Ecuador consisted of 4 genotypes (Tzotzol, Iztac 1, Iztac 2 and Miztic) sown on January 15, 2007 in replicated plots to assess production and composition. Seed yield was affected by genotype, with Miztic and Tzotzol producing significantly (p<0.05) greater yields than the Iztac II genotype, but not more than Iztac I which was not significantly (p<0.05) different from Iztac II. Iztac II had the highest protein content (24.43%), however the difference was significantly (p<0.05) different only from Iztac I. Neither Iztac II nor Iztac I were significantly (p<0.05) different from either the Tzotzol or Miztic genotypes. No significant difference (p<0.05) in lipid content was found among genotypes. Miztic and Iztac II, with 20.23% and 20.03%, respectively had significantly (p<0.05) higher linoleic fatty acid percentages than the 19.23% of the Iztac I genotype. Iztac I had the highest α-linolenic fatty acid percentage (61.73) and this was significantly (p<0.05) different than the 58.37% found for the Iztac II genotype. All of the genotypes showed a similar relationship among compounds, that being caffeic acid>chlorogenic acid> quercetin>kaempherol. In summary, the effect of genotype was more evident on seed yield than protein content, oil content, fatty acid composition and phenolic compounds, hence yield needs to be the main factor when considering establishment of chia as a crop in the area.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2009.301.307
URL: https://scialert.net/abstract/?doi=ajps.2009.301.307
INTRODUCTION
Coronary Heart Disease (CHD) causes 1 of every 5 deaths in the United States and now is the greatest killer of American males and females and this year it is estimated that 700,000 Americans will suffer from CHD and about 500,000 will have a recurrence of the disease (American Heart Association, 2007). Coronary Heart Disease also is the leading cause of disability in the US labor force, accounting for 19% of all disability payments made by the Social Security Administration (American Heart Association, 2004). The estimated direct and indirect costs of CHD for 2007 are $ 151.6 billion (American Heart Association, 2007). Coronary Heart Disease is caused by arteriosclerosis and can be prevented if people make healthier dietary choices. Increased intake of saturated and polyunsaturated ω 6 fatty acids have been shown to exacerbate the risk of CHD. Conversely, increased intake of ω 3 fatty acids, including α-linolenic fatty acid and its metabolites EPA and DHA, reduce the risk of suffering CHD (Baylin et al., 2003; Lorgeril et al., 1996). Unfortunately Western populations are consuming foods low in ω 3 fatty acids, hence consumption needs to increase to decrease the risk of CHD (American Heart Association, 2004). The problem is, however, there are very few readily available ω 3 sources for human or animal consumption and most of these should only be consumed in moderation, if at all. The reason for this is contaminants such as dioxin and mercury are present in fish and anti-nutritional compounds are found in flaxseed (Hamilton et al., 2005; Bhatty, 1993). Clearly any reliable source of ω 3 fatty acids that can be found which is safe for consumption would be attractive. Chia (Salvia hispanica L.) contains ω 3 fatty acids and its oil provides the richest plant source of α-linolenic fatty acid known (Ayerza and Coates, 2005a). Studies in which chia was used as a dietary source of α-linolenic fatty acid for rats have shown improved LDL, HDL and TG levels along with a better blood fatty acid profile (Ayerza and Coates, 2007, 2005b) which should consequently reduce the risk of suffering CHD. Additionally chia seeds have not shown any of the problems associated with other ω 3 sources such as flaxseed or marine products which have introduced a fishy flavor into foods, exhibited immune reactions, animal weight loss, digestive problems, etc. (Azcona et al., 2008; Ayerza and Coates, 1999, 2002). Although, chia was one of the four main crops of the Aztecs along with corn, beans and amaranth, it was for all practical purposes eliminated as a crop. This was most likely due to the Aztec’s use of it in their religious ceremonies (Ayerza and Coates, 2005b). Efforts to incorporate chia into modern agriculture only began in 1991 through the Northwestern Argentina Regional Project (Ayerza and Coates, 2005b). As a consequence little information about chia seed production and the compounds within the seed and how they relate to the environment and genetics is available. Santa Elena Peninsula is a warm, arid area located along the south-central coast of Ecuador. Precipitation varies within the region from 112 mm year-1 in Salinas, up to 550 mm year-1 in Chongon. In this area, as is true all along the Ecuadorian coast, the El Niňo phenomenon affects rainfall patterns dramatically. At irregular periods, in general every seven years, El Niňo is much stronger and brings heavy rains which causes flooding that destroys the basic infrastructure of the area including roads, bridges, houses and crops (Ministerio del Ambiente, 2004; Cornejo, 2003). This periodic destruction of crops is one of the major obstacles to cultivation of perennial species which need 3-4 years to fruit. In the 1990s the Government of Ecuador began work on an irrigation system designed to provide water for up to 23,000 has of the approximately 540,000 has which comprise the Santa Elena Peninsula (Ministerio del Ambiente, 2004; Cornejo, 2003). This study has not met expectations and only 6,512 has are now cultivated (Cornejo, 2003). One of the problems has been an absence of data which can be used to identify crops that should do well in the area (C. Noel, 2006, Salinas, Guayas, Ecuador, personal communication). Obviously any crop for which there is a market and which appears adapted to the region, would be attractive. The objective of this study was to plant 4 genotypes of chia in the region to determine which is the best in terms of seed yield (kg ha-1), amount of protein (%), oil content (%) and phenolic compounds (quercetin, kaempherol, chlorogenic acid, caffeic acid as molecular weight in g kg-1 of seed produced), as well as fatty acid composition (%).
MATERIALS AND METHODS
The research plot was established on the Santa Marta farm, located at 020 18’ 00" South, 800 37’ 00" West, 48 m above sea level. The soil type is Entic Haplustoll according to the USDA Soil Classification System (Cornejo, 2003). Meteorological data showed that an annual mean precipitation of 220 mm year-1 evaporation of 1,627 mm year-1 and an average temperature of 25.7°C with remarkable uniformity occurring from year to year (Cornejo, 2003). For the test an average temperature of 26.5°C was recorded over the last 30 days of the crop cycle, with data being recorded on site using a portable weather station. Experimental design was a randomized complete block with 3 replications. Plots consisted of 3 rows, 10 m in length, spaced 1.6 m apart. Seeding rate was 5 kg ha-1. The plots were maintained using conventional cultural and drip-irrigation practices. No insect problems or plant diseases were encountered. Sowing was done on January 15/ 2007 and harvested on April 30/2007. Four genotypes were planted: Tzotzol, Iztac 1, Iztac 2 and Miztic. A 1 m long section from the center of the middle row of each plot was hand-harvested, cleaned and weighed for yield determination following procedures used by the researchers in similar chia studies. A sub-sample from each genotype and each replication was obtained to determine 1000-seed weight and to conduct the laboratory analysis. Crude nitrogen was determined by a standard micro-Kjeldahl method (Guebel et al., 1991) and converted into protein content using a 5.71 conversion factor.
Lipids were extracted and converted into fatty acid methyl esters using the IRAM 5-560II method (Instituto Argentino de Racionalización de Materiales (IRAM), 1982). Fatty acid methyl esters were separated and quantified by an automated gas chromatograph (Model 6890, GC; Hewlett Packard Co., Wilmington, DE, 20006) equipped with flame ionization detectors and a 30 mx530 μm i.d. capillary column (Model HP-FFAP; Hewlett Packard Co., Wilmington, DE, USA). Phenol compounds were extracted using an Ultraturrax homogenizer (IKA Works Inc., Wilmington, NC, USA). A methanol:water solution of 90:10 was utilized, with this being filtered through a 0.45 μm membrane prior to analysis. Quantification and determination of the various phenolics in the crude extract were carried out using a TSP (Thermo Separation Products Inc., Piscataway, NJ, USA) chromatograph equipped with a quaternary pump, a degasses and diode array detector (L 3000). Phenolic compounds were separated using a ODS-Hypersil C18 column, 250x4.6 mm i.d. and particle of 5 μm (Knauer, Berlin, Germany) and a Lichrospher RP-18 pre-column (Merck KgaA, Darmstadt, Germany). The column temperature was 25°C, with injection volume for the chia seed extracts and standards set at 20 μL following procedures used by the researchers in earlier chia trials. The system was run with a linear gradient of a two solvent mix according to Vitract et al. (2002). The phenolic concentrations were determinated by comparison with standard calibration curves developed using the same chromatographic system and seven concentration levels of each compound. As insufficient seed for each replication was available for phenol analysis only one sample, made by combining the three replications, was analyzed for the Iztac I, Tzotzol and Miztic genotypes. Phenol analysis was not performed on the Iztac II genotype, since insufficient seed was available. Each variable was compared by analysis of variance. When the F-value was significant (p<0.05), means were separated using Duncan’s new multiple range test (Cohort Stat, 2006).
RESULTS AND DISCUSSION
Seed yield was affected by genotype. The Miztic and Tzotzol genotypes produced significantly (p <0.05) higher seed yields than the Iztac II genotype, but not more than Iztac I which was not significantly (p<0.05) different from Iztac II. Tzotzol and Miztic genotypes, with 1.30 and 1.22 g/1000 seeds, respectively, had higher seed weights, followed by Iztac I and Iztac II genotypes with 1.20 g/1000 seeds each (Table 1). As seed weights were not replicated, no statistical significance for the differences could be established. Iztac II had the highest protein content (24.43%), however, the difference was significantly (p<0.05) different only from Iztac I. Neither Iztac II nor Iztac I were significantly (p<0.05) different from either the Tzotzol and Miztic genotypes. No significant difference (p<0.05) in lipid content was found among genotypes, however palmitic and stearic fatty acid percentages were significantly (p<0.05) different among genotypes.
| Table 1: | Crop cycle, seed yield and seed weights of four chia genotypes |
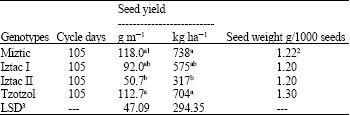 | |
| 1Means within each column did not differ significantly (p<0.05) according to Duncan´s multiple range test; 2Not replicated and 3Least significant difference for p<0.05 | |
Total saturated fatty acid percentage, was significantly (p<0.05) lower for Iztac I compared to the Miztic and Iztac II genotypes. Miztic and Iztac II, with 20.23 and 20.03%, respectively, had significantly (p<0.05) higher linoleic fatty acid percentages than the 19.23% of the Iztac I genotype. Iztac I had the highest α-linolenic fatty acid percentage (61.73) and this was significantly (p<0.05) different than the 58.37% found for the Iztac II genotype (Table 2). Total polyunsaturated fatty acid (PUFA), calculated as the sum of linoleic and α-linolenic fatty acids, was significantly (p<0.05) higher for Iztac I, compared to Iztac II. The ω 6:ω 3 and SAT:PUFA fatty acid ratios were significantly (p<0.05) lower for Iztac I compared to Iztac II, with the ω 6:ω 3 ratio for Iztac I also being significantly lower than the Miztic as well. Four phenolic compounds were detected in all genotypes namely, quercetin, kaempherol, chlorogenic acid and caffeic acid. Individual and total phenolic contents varied among genotypes, however as the analysis were not replicated, statistical significance of the differences could not be established (Table 3). However, all of the genotypes showed a similar relationship among compounds, that being caffeic acid>chlorogenic acid>quercetin>kaempherol. The sum of the chlorogenic and caffeic acids comprised 98.4, 98.6 and 97.7% of the total phenolic compounds found in the Miztic, Iztac I and Tzotzol, genotypes, respectively.
Significant (p<0.05) differences between the Miztic and Tzotzol yields and that of Iztac II show a strong genotype effect on seed yield under the environmental conditions in which the experiment was conducted. However, the seed yields measured herein were lower than those reported in other experiments. Maximum yields of 1,355, 938 and 862 kg ha-1 with growth cycles of 148, 173 and 174 days, respectively, were reported for three other experimental fields. The first 2 were in the Sub Humid Chaco ecosystem and the other, which was irrigated, was in the Semiarid Chaco ecosystem.
| Table 2: | Means of the protein and oil contents and fatty acid composition in four chia genotypes |
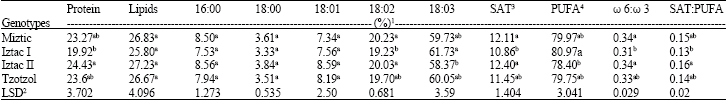 | |
| 1Means in a column within a group with the same letter(s) are not statistically different (p<0.05) according to Duncan´s multiple range test; 2Least significant difference for p<0.05; 3SAT: Saturated fatty acids; 4PUFA: Polyunsaturated fatty acids | |
| Table 3: | Phenolic content and composition in the chia genotypes |
 | |
| 1Standard deviation | |
Additionally yields were less than the 1,602 and 1,188 kg ha-1, with crop cycles of 166 and 151 days, respectively, for commercially irrigated fields established in 2 locations in the Semiarid Chaco ecosystem (Coates and Ayerza, 1996). Yields of 1,171 and 1,047 kg ha-1, with cycle durations of 124 and 140 days, respectively, were reported for two other irrigated commercial fields located in the Semiarid Chaco ecosystem (Coates and Ayerza, 1998).
Differences in seed yields between these plots and earlier trials could be a result of a combination of factors including genetics, environmental conditions, agronomic practices, seeding dates and their interactions. A decrease in biomass and seed yield was reported when chia seeding dates were compared. Coates and Ayerza (1996) determined that both biomass and total seed yields were significantly (p<0.05) higher for earlier plantings compared to later plantings. The lower seed yields reported herein could be related to the shorter crop cycle for the current study, since crop cycle has been reported as an important factor affecting crop yield (Ellis et al., 1990). In general high temperatures shorten crop cycles, resulting in a reduction in yield, decreased fruit set and rate of photosynthesis (Uzun, 2007; Ellis et al., 1990). Since, chia is a fall-winter crop in the subtropical Chaco ecosystem, the lower yields found with all four genotypes could be related to the shorter crop cycle caused by the high summer temperatures. Another factor contributing to yield differences could be row spacing. Since, the other studies were sown with 0.70-0.80 m between rows (Coates and Ayerza, 1996, 1998). This means an approximate 2.3 fold increase in planted area. Another experiment specifically set up to examine the effect of plant density under irrigated conditions in the Arid Chaco Ecosystem, found the highest seed yield with a 0.70 m spacing, as compared to greater separation between rows (Gonzalez Vera et al., 1996). The significant (p<0.05) differences between yields of the Miztic and Tzotzol genotypes and Iztac II that were found would tend to indicate that a strong genotype effect on seed yield exists, at least under the environmental conditions in which the experiment was conducted. Seed weights found herein are in agreement with that of the 1.24 g/1,000 seeds reported by Guyot and Rueda (1996) for chia produced under irrigated conditions in the Arid Chaco ecosystem. However, the seeds were much heavier than the 0.32-0.53 g/1,000 seeds reported by Coates and Ayerza (1998), for six samples of a common genotype collected from commercial farms in the Semiarid Chaco ecosystem and the average of 0.89 g/1,000 seeds (with extremes of 0.79-0.95 g/1,000 seeds) reported by Gonzalez Vera et al. (1996) for a trial in the Arid Chaco ecosystem. The differences could be a result of genetics alone, or to a genetic x environment interaction. All but the Iztac II genotype showed a higher protein content than the maximum value of 23.1% reported for seed produced in 9 sites located within 6 different tropical and subtropical ecosystems of South America (Ayerza and Coates, 2004). The higher protein content found herein could be related to the temperature difference between trials. The average temperature measured over the last 30 days of the crop cycle was 25.7°C, which is 1.5°C higher than that recorded over the last 30 days of the Ayerza and Coates (2004) trial. Another trial demonstrated that as altitude decreased, with a subsequent temperature increase, protein content of chia seed tended to increase (Ayerza and Coates, 2004). Other crops such as sorghum and soybeans have shown similar changes in protein content induced by environment (Vollmann et al., 2000; Mohammed et al., 1987). The oil contents measured herein were lower than those of chia seeds grown in other ecosystems. As demonstrated for chia and other oilseed crops, temperatures affect oil content: high temperatures decrease oil content, while low temperatures increase oil content (Ayerza, 2001; Yaniv et al., 1995; Cherry et al., 1985). The mean oil content and extreme values found for chia produced at nine sites (Ayerza and Coates, 2004) were 30.7 and 28.5-32.7%, respectively; while for five other sites in Northwestern Argentina they were 35.9 and 35.6-38.6%, respectively (Ayerza, 1995). Since, the mean temperature for the current trial was 1.7 and 6.4°C higher than those measured in the 2 trials noted above, this most likely was the reason for the lower oil content. In general, SAT and PUFA contents, as well each of their components, were slightly higher and lower, respectively, compared to oil from seeds grown under the lower temperatures reported by Ayerza and Coates (2004) and by Ayerza (1995). A SAT palmitic fatty acid increase and a PUFA α-linolenic decrease [both significant (p<0.05) were found to be positively correlated with increased temperatures during the last 30 days of the crop cycle (Ayerza, 1995). Increased oil saturation through increased palmitic and stearic acid contents, with concomitant decreases in linoleic and linolenic acids brought about by increased temperatures has been reported in soybeans (Thomas et al., 2003). Lack of a significant (p<0.05) difference in palmitic, stearic and oleic fatty acid percentages between genotypes and the small, but significant (p<0.05) difference in linoleic content found between Iztac I and Miztic and Iztac II and the α-linolenic fatty acid content between Iztac I and Iztac II genotypes could indicate a relatively close genetic relationship among genotypes. Data from a trial conducted at 3 sites located between 1,600 and 2,200 m of elevation showed the Tzotzol and Iztac I genotypes to have similar oil content and fatty acid compositions, even though significant (p<0.05) differences in seed yield were recorded (Ayerza and Coates, 2004). Hence, these data would tend to support the theory of small genetic diversity reported by Cahill (2004) using RAPD (Random Amplified Polymorphic DNA) markers for domestic accessions of chia. The current trial, however, did indicate that some fatty acid differences do exist among genotypes. As a general recommendation, one way of reducing the risk of CHD is to lower the ω 6:ω 3 ratio in the diet from typical values of 15:1 to no more than 5:1, with an ideal ratio being 1:1 (British Nutrition Foundation, 1992; Canada Health and Welfare, 1990). Given this, the significantly (p<0.05) lower ω 6:ω 3 ratio shown by the Iztac I genotype, compared to that of the Miztic and Iztac II genotypes, might be considered a nutritional advantage. Several nutritional studies support a relationship between SAT consumption and the risk of CHD exists. Hence, there is a need to reduce these fatty acids in the diet and increase the consumption of PUFAs (Canada Health and Welfare, 1990). The significantly (p<0.05) lower total SAT content (up to 12%) along with the lower SAT:PUFA ratio (up to 19%) exhibited by the Iztac I genotype also makes this genotype appear to be nutritionally superior. The phenolic compounds found in chia have been shown to possess consistently strong antioxidant properties (Reyes-Caudillo et al., 2007; Taga et al., 1984). A number of studies have shown good oxidative stability of chia seed when used as animal feed or as a food ingredient, with this being attributed to the high antioxidant activity of the phenolic compounds it contains (Bautista Justo et al., 2007; Ayerza et al., 2002; Ayerza and Coates, 2002, 1999). Interestingly the phenolic compound analysis did not determine any measurable amount of myricetin, which was reported earlier by Taga et al. (1984). Whether this is just an anomaly, or a result of environment is not known. The total phenolic amounts found herein are lower than the 0.757-0.881 mg g-1 found for 2 chia seed sources as reported by Reyes-Caudillo et al. (2007). This could be related to environment, since antioxidant content has been found to be affected by growing season for Artemisia princeps var. orientalis (Yun et al., 2008).
CONCLUSIONS
The effect of genotype is more evident on seed yield than protein content, oil content, fatty acid composition and phenolic compounds. The data herein is consistent with earlier research which has reported that high temperatures reduce oil content and degree of unsaturation, while increasing protein content. The seed yields, which were obtained in only 105 days, indicate that chia seed can be produced in this region with a comparatively, short crop cycle. Additional trials are required to fully assess the potential of chia cultivation in this region, but in general, the data indicate that chia could be an alternative to the traditional crops grown here.
ACKNOWLEDGMENTS
The authors acknowledge support for this study from the Corporation International de Comercio y Servicios S.A, Buenos Aires, Argentina and Hacienda Santa Marta, El Azucar, Ecuador. The authors are also grateful to Ing. Carlos Noel and Agr. Ricardo Weisson and Agr. Fernando Castilla for caring for the experimental plots and assistance throughout the trial and to Dr. Marina Insani for her support with HPLC tests.
REFERENCES
- Ayerza, R., 1995. Oil content and fatty acid composition of Chia (Salvia hispanica L.) from five Northwestern locations in Argentina. J. Am. Oil Chem. Soc., 72: 1079-1081.
CrossRefDirect Link - Ayerza, R. and W. Coates, 2005. Ground chia seed and chia oil effects on plasma lipids and fatty acids in the rat. Nutr. Res., 11: 995-1003.
CrossRef - Ayerza, R. and W. Coates, 2004. Composition of chia (Salvia hispanica L.) grown in six tropical and subtropical ecosystems of South America. Trop. Sci., 44: 131-135.
CrossRef - Ayerza, R. and W. Coates, 2002. Dietary levels of chia: Influence on hen weight, egg production and sensory quality, for two strains of hens. Br. Poult. Sci., 43: 283-290.
CrossRef - Bautista Justo, M., A.D. Castro Alfaro, E. Camarena Aguilar, K. Wrobel and K.E. Wrobel et al., 2007. Desarrollo de pan integral con soya, chia, linaza, y acido folico como alimento funcional para la mujer. Arch. Latinoam. Nutr., 1: 78-84.
Direct Link - Baylin, A., E.K. Kabagambe, A. Ascherio, D. Spiegelman and H. Campos, 2003. Adipose tissue 18:3n-3 acid and nonfatal acute myocardial infarction in Costa Rica. Circulation, 107: 1586-1591.
Direct Link - Bhatty, R.S., 1993. Further compositional analyses of flax: Mucilage, trypsin inhibitors and hydrocyanic acid. J. Am. Oil Chem. Soc., 9: 899-904.
CrossRefDirect Link - Cahill, J.P., 2004. Genetic diversity among varieties of chia (Salvia hispanica L.). Genet. Resour. Crop Evol., 51: 773-781.
CrossRef - Cherry, J.H., L. Bishop, P.M. Hasegawa and H.R. Leffler, 1985. Differences in the fatty acids composition of soybean seed produced in northern and southern areas of the USA. Phytochemistry, 24: 237-241.
CrossRef - Coates, W. and R. Ayerza, 1998. Commercial production of chia in Northwestern Argentina. J. Am. Oil Chem. Soc., 75: 1417-1420.
CrossRef - Coates, W. and R. Ayerza, 1996. Production potential of chia in Northwestern Argentina. Ind. Crops Prod., 5: 229-233.
CrossRefDirect Link - Gonzalez Vera, C.A., M.I. Fernandez, O.E. Roldan, L.D. Montalvan, V.L. Luque, G.E. Contreras and S.M. Gorosito, 1996. Incidencia de la epoca, distanciamiento y densidad de siembra en la produccion de semilla de Salvia hispanca L. en Catamarca. Proceedings of the 9th International Conference on Jojoba and Its Uses and of the 3rd International Conference on New Industrial Crops and Products, (JICP'96), The Association for the Advancement of Industrial Crops, pp: 358-362.
- Guyot, E.C. and M.E. Rueda, 1996. Chia: Capacidad productiva en el Valle Central de Catamarca. Proceedings os the 9th International Conference on Jojoba and Its Uses and of the 3rd International Conference on New Industrial Crops and Products, (JICP'96), The Association for the Advancement of Industrial Crops, pp: 351-354.
- Guebel, D.V., B.C. Nudel and A.M. Giulietti, 1991. A simple and rapid micro-Kjeldahl method for total nitrogen analysis. Biotechnol. Tech., 5: 427-430.
Direct Link - Hamilton, M.C., R.A. Hites, S.J. Schwager, J.A. Foran, B.A Knuth and D.O. Carpenter, 2005. Lipid composition and contaminants in farmed and wild salmon. Environ. Sci. Technol., 39: 8622-8629.
CrossRef - IRAM-Instituto Argentino de Racionalizacion de Materiales, 1982. Aceites y Grasas Vegetales y Animales: Metodo Rapido de Preparacion de Esteres Metilicos de Acidos Grasos Para Su Analisis Por Cromatografia en Fase Gaseosa. Instituto Argentino de Racionalizacion de Materiales, Buenos Aires, Argentina.
- Saeed, M., C.A. Francis, J.F. Rajewski and J.W. Maranville, 1987. Genotype × environment interaction and stability analysis of protein and oil in grain sorghum. Crop Sci., 27: 169-171.
CrossRefDirect Link - Reyes-Caudillo, E., A. Tecante and M.A. Valdivia-Lopez, 2008. Dietary fibre content and antioxidant activity of phenolic compounds present in Mexican chia (Salvia hispanica L.) seeds. Food Chem., 107: 656-663.
CrossRefDirect Link - Taga, M.S., E.E. Miller and D.E. Pratt, 1984. Chia seeds as a source of natural lipid antioxidants. J. Am. Oil Chem. Soc., 61: 928-931.
CrossRefDirect Link - Thomas, J.M.G., K.J. Boote, L.H. Allen, Jr. Gallo-Meagher and J.M. Davis, 2003. Elevated temperature and carbon dioxide effects on soybean seed composition and transcript abundance. Crop Sci., 43: 1548-1557.
Direct Link - Vollmann, J., J. Winklerb, C.N. Ritza, H. Grausgrubera and C. Ruckenbauera, 2000. Spatial field variations in soybean (Glycine max. [L.] Merr.) Performance trials affect agronomic characters and seed composition. Eur. J. Agron., 12: 13-22.
Direct Link - Yun, K.W., H.J. Jeong and J.H. Kim, 2008. The influence of the growth season on the antimicrobial and antioxidative activity in Artemisia princeps var. orientalis. Ind. Crops Prod., 27: 69-74.
CrossRef - Ayerza, R., W. Coates and M. Lauria, 2002. Chia seed (Salvia hispanica L.) as an omega-3 fatty acid source for broilers: Influence on fatty acid composition, cholesterol and fat content of white and dark meats, growth performance, and sensory characteristics. Poult. Sci., 81: 826-837.
CrossRefDirect Link








