Research Article
Phytohaemagglutinins in Membrane Signalling, Biomedical and Genetic Engineering Research
University of Reading, WhiteKnight, Reading, UK
R.A. Rastall
School of Food Science, University of Reading, WhiteKnight, Reading, UK
The onset of the scientific discipline called ‘lectinology’ dates back to the observation by Stillmark in 1888 that preparation from castor bean extracts agglutinated red blood cells. As more and more agglutinating factors were discovered in other plants, the term ‘haemagglutinin’ was introduced as a common name for all substances that exhibited this particular biological activity (Van Damme et al., 1998). However, when the important discovery was made that some haemagglutinins selectively agglutinated blood cells of a particular human blood group within the ABO blood group system, the novel term ‘lectin’ (derived from the Latin verb ‘legere’, which means ‘to select’) was introduced to denote this aspect of selectivity. (Boyd and Reguera, 1949).
Plant lectins have long been used as powerful and specific tools in carbohydrate chemistry and histology. However, in addition to these traditional uses, lectins are now also recognised in their own right as a class of communication proteins which are involved in interactions between plants and their environment and which may also have physiological functions within the plant (Goldstein et al., 1980). Fundamental biological studies in the last decades revealed that plants can have many and varied interactions with a range of other life forms from the simplest micro-organisms to insect and mammalian predators, including humans and that in these, lectins may be one of the principal communication molecules which developed during evolution for precisely this reason. The fulfilment of this basic physiological role is the inextricably linked with their ability to specifically recognise and bind to carbohydrate structures (Putsztai, 1991). When these are present on cellular membranes of other organisms, lectins can act as biological signals in cell- to-cell interactions and send messages to these exogenous organisms by mimicking the actions of endogenous hormones and growth factors (Peumans and Van Damme, 1995). Using molecular modelling techniques, new opportunities have arisen to explore similarities, make predictions and establish correlations in sugar-binding activity and/ or specificity between well-studied and newly discovered lectins (Etzler, 1986).
Subdivision of lectins
With the progress in the purification and characterisation of lectins, evidence accumulated that lectins are a very heterogenous group of proteins, artificially classified together solely on the basis of their capability to agglutinate cells (Van Damme et al., 1998). However, as soon as it was demonstrated that the agglutination properties of lectins were based on their specific recognition of and binding to carbohydrates (Watkins and Morgan, 1952), the question arose as to what criteria had to be met by a protein in order for it to be regarded as a lectin. The first definition of lectins was based primarily on the sugar specificity and inhibition of the agglutination reaction. According to this definition, lectins are carbohydrate-binding proteins of non-immune origin which agglutinate cells or precipitate glycoconjugates (Golstein et al., 1980). This definition was too restrictive as it excluded some poorly agglutinating toxins (such as ricin, abrin, modecin, etc) which were known to contain lectin subunits and so the definition was extended to include these toxins (Kocourex and Horejsi, 1983).
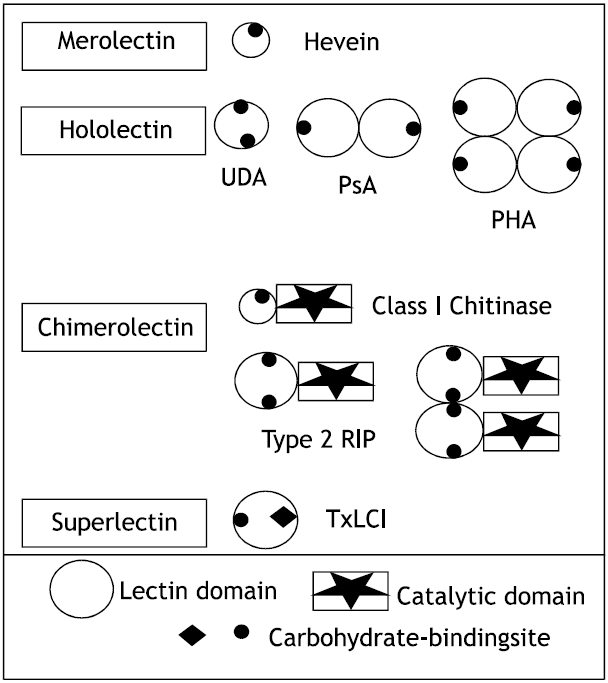
On the basis of the overall structure of lectin subunits (or more precisely the primary translation products of the lectin genes) four major types of lectins are distinguished, namely merolectins, hololectins, chimerolectins and superlectins (Fig. 1).
Merolectins are small proteins consisting exclusively of a single carbohydrates binding domain. Due to their monovalent nature, merolectins cannot precipitate agglutinate cells. At present, only a few merolectins have been described. Well known examples are the chitin- binding hevein from latex of the rubber tree (Hevea brasiliensis) (Van Parijs et al., 1991) and the monomeric mannose-binding proteins from orchids (Van Damme et al., 1994).
Hololectins are also exclusively composed of carbohydrate-binding domains. However in contrast to merolectins, they contain two more carbohydrate-binding domains which are identical or very homologous and bind either the same or structurally similar sugar. Since hololectins have multiple binding sites they are fully capable of agglutinating cells and precipitate glycoconjugates. Most plan lectins belong to the subgroup of hololectins (Goldstein et al., 1908).
 | |
| Fig. 1: | SchematicrepresentationofPlantLectinFamily(Merolectins,Hololectins, Chimerolectins and superlectins (Van Damme et al., 1998) |
Chimerolectins are basically fusion proteins composed of a carbohydrate-binding domain tandemly arrayed with an unrelated domain. The latter domain may have a well-defined catalytic activity but acts independently of the carbohydrate-binding domain (Kocourek and Horejsi, 1983). Chimerolectins with multiple carbohydrate-binding sities bahave as hololectins, e.g. Type 2 RIP (Ribosome activating protein), which possess two carbohydrate-binding sites on each B chain and agglutinate cells.
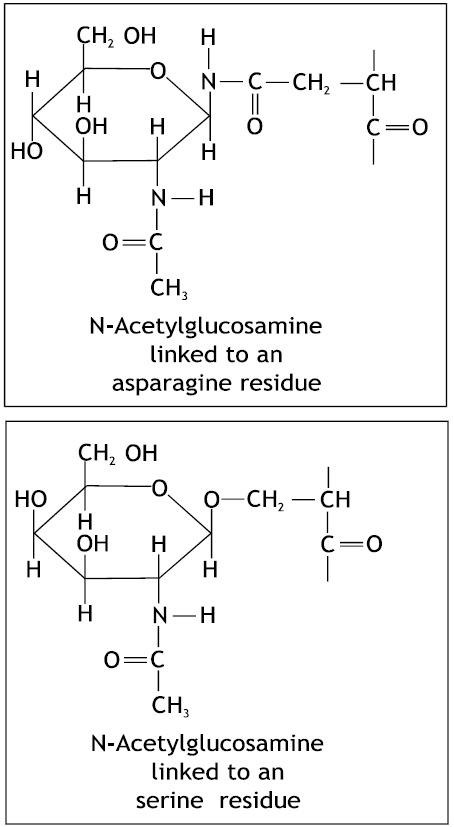 | |
| Fig. 2: | Sugar binding residue linkage to lectin structures (Stryer, 1981) |
 | |
| Fig. 3: | Electron micrograph of lectin Concanavalin A conjugate with ferritin (protein with a very dense core of iron hydroxide) (Adopted from Stryer, 1981) |
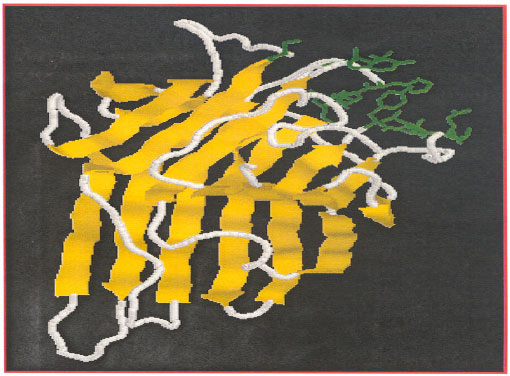 | |
| Fig. 4: | Surface loop structure of concanavalin A-lectin (highlighted in green) involved in carbohydrate-binding site. (http://.food.reading.ac.uk/online/fs916/lect11/lect11.htm) |
 | |
| Fig. 5: | Interaction between cell-adhesion molecules during the binding process of T-cells Leucocyte (Hydes and Lander, 1992) |
 | |
| Fig. 6: | Major families of Cell membrane adhesion-communication molecules (CAMs) (Lodish et al., 2000) |
On the contrary, chimerolectins with a single carbohydrate-binding site bahave as merolectins. Class 1 plant chitinase, for instance, which have only one chitin-binding domain per molecule, cannot precipitate glycoconjugates or agglutinates cells (Van Damme et al., 1994).
 | |
| Fig. 7: | Mitotic division of isolated leucocyte stimulated by plant lectins glycoprotein complex, from seed of Phaseolus vulgaris (Bowman and Rand, 1990) |
Superlectins are special type of chimerolectins. They are fusion proteins built up of two tandemly arrayed carbohydrate-binding domains which are structurally different and recognize structurally unrelated sugars. At present, only one superlectin has been described, namely a lectin from tulip bulbs (Cammue et al., 1986). The N-terminal mannose-binding domain tandemly arrayed with an unrelated GalNAc-binding domain (Van Damme et al., 1996).
Occurrence and distribution of plant lectins
Lectins are usually considered as a very large and heterogenous group of proteins (Goldstein and Poretz (1986). Numerous plant species of different taxonomic groupings contain lectins, but the total number of documented cases is less than 500. Agglutinating lectins are relatively scarce, while the chimero lectins belonging to the Class 1 chitinase seem to be present in almost all plant species (Collinge et al., 1993).
Plant lectins for a long time has been regarded as typical seed proteins. However, the occurrence of lectins in vegetative tissues is well documented. A short survey of the occurrence and concentration of lectins in seeds as well as in different types of vegetative tissues reveals striking differences in the location and relative abundance of the individual lectins (Etzler, 1986). Seed lectins, for instance, are located in the cotyledons (e.g. in legumes) or in the endosperm (e.g. castor bean), or are confined to the primary axis (e.g. in wheat). Typically, lectins usually account for 0.1-5% of the total seed protein. However, some seed lectins are predominant proteins representing up to 50% of the total protein (e.g. in some Phaseolus species), whereas others are only minor, quantitatively unimportant proteins, this also applies to non-seed lectins, which have been found in virtually all kinds of vegetative tissues such as leaves, stem, bark, bulbs, tubers, corms, rhizomes, roots, fruits, flowers, ovaries, phloem sap and even nectar (Peumans and Van Damme, 1995). Non-seed lectins may occur in different tissues of the same plant. For instance, the snowdrop and daffodil lectins have been found in virtually all vegetative tissues, although the lectin is most abundant in the bulbs (Van Damme and Peumans, 1990).
Carbohydrate-binding specificity of plant lectins
The specificity of plant lectins is usually expressed in terms of their ability to bind one or more specific monosaccharides. On this basis, lectins have been subdivided into glucose/mannose-specific, Gal/GalNAc-specific, GlcNAc-specific, fucose-specific-specific and sialic acid specific lectins (Goldstein and Poretz, 1986). Classification of lectins in specificity groups on the basis of their binding to monosaccharides suffers another drawback, due to the fact that some lectins bind simple sugars does not imply that these monosaccharides are their true receptor molecules, since even these lectins usually have a much higher affinity for oligosaccharides containing two or more monosaccharides units. Plant lectins have prefererence for carbohydrates from animal or microbial origin. This preference has two important consequences. First, it indicates that most lectins are destined to bind foreign glycoconjugates rather than endogenous receptors. Second, it makes plant lectins important and useful tools for the isolation and analysis of human and animal glycoconjugates (Peumans and Van Damme, 1995).
In membrane glycoproteins, one or more chains of sugar residues are attached to serine, threonine, or asparagine sode chains of the protein, usually through N-acetylglucosamine or N- acetylgalactosamine. The location of these carbohydrate groups in membranes can be determine by specific labelling techniques. The lectins, which are plant proteins with high affinity for specific sugar residues, are valuable probes in this regard. For example concanavalin A binds to internal and nonreducing terminal;-mannosyl residues, whereas wheat-germ agglutinin binds to terminal N-acetylglucosamine residues. These lectins can be readily seen in electron micrographs if they are conjugated to ferritin, a protein with a very electron dense core of iron hydroxide (Stryer, 1981).
The ferritin conjugate of concanavalin A binds specifically to outer surface of the erythrocyte membrane and not to the inner cytoplasmic surface (Fig. 3). Sugar residues are always located on the external surface of plasma membranes. Plant lectins involved in carbohydrate binding sites is formed from a surface loop structure suitable for the binding stability and catalytic activity. An example of lectin with an surface loop structure is Concanavalin A (Fig. 4).
The physiological role of plant lectins
The breakthrough in the search for the physiological role of plant lectins was achieved when it was realized that most plant lectins not only play a role in the plant itself (e.g. as a store of nitrogen or as a specific recognition factor) but are also capable of interfering with the functioning of foreign organism through an interaction with glycoconjugates on the surface or in the digestive tract of these organisms (Taylor and Kennewell, 1993). Most plant lectins are probably involved in the plant defence. The direct interference with viruses and micro-organisms is rather exceptional and the deleterious effects of plant lectins on both predatory invertebrates and higher animals are well documented. Not all lectins are involved in defensive role, for instance lectins occurring at low concentrations might well be involved in specific recognition processes either within or outside the plant (Fischer et al., 1984). Legume root lectins, for instance, could be involved in the recognition and /or binding of Rhizobium and Bradyrhizobium species and hence be a determining factor in establishing symbiosis (Diaz et al., 1989).
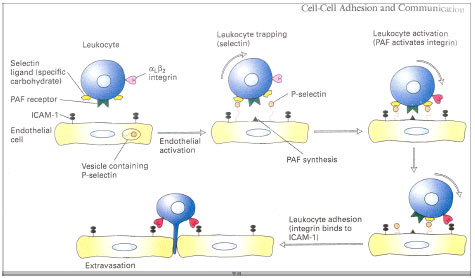
Lectins and other cell adhesion molecules (CAMs) participation in leucocyte extravasation
There are five principal classes of CAMs (Fig. 5). Cadherins, the immunoglobulin (Ig), superfamily, selectins, mucins and integrin (Lodish et al., 2000). Cell to cell adhesion involving cadherins and selectins depends on Ca2+ ions whereas interactions involving integrin and Ig- superfamily CAMs do not. P-selectin is a lectin, a protein that binds to carbohydrates. Each type of selectin binds to specific oligosaccharide sequences in glycoproteins or glycolipids. As with cadherins, binding of selectins to their ligands is Ca2+-dependent. The sugar binding lectin domain in selectin generally is at the end of the extracellular region of the molecule (Fig. 5). The ligand for P-selectin is a specific oligosaccharide sequence, called the sialyl Lewis-x antigen, that is part of longer oligosaccharides present in abundance on leucocytes glycoprotiens and glycolipids.
In normal endothelial cells P-selectin is localised in intracellular vesicles and are not present in plasma membrane. These cells are activated by various inflammatory signals released by surrounding cells in areas of infection or inflammation (Fig. 6). Once the endothelial cells are activated, the vesicles containing P-selectin undergo exocytosis within seconds and P-selectin appears on the plasma membrane. As a consequence, passing leucocytes adhere weakly to the endothelium; because of the force of the blood flow, these ”trapped” leucocytes are slowed but not stopped and seem to roll along the surface of the endothelium.
In order for tight adhesion to occur between activated endothelial cells and leucocytes, β2-containing intergrins on the surface of leucocytes also must be activated. For example, activation of the αLβ2 integrin, which is expressed by T lymphocytes, is induced by platelet- activiaitng factor (PAF), a phospholipid released by activiated endothelial cells at the same time that P-selectin is exocytosed. Binding of PAF to its receptor on T lymphocytes activates integrin αLβ2 through the Rho signalling pathway. The activiated integrin then binds to ICAM-1 and ICAM-2, which are Ig-superfamily CAM expressed contitutively on the surface of the endothelial cells (Lodish et al., 2000)
Plant lectins in medical research
Role of plant lectins in karyotype (chromosome pattern)
The mitotic division of isolated lymphocytes is stimulated by specific antigens to which they are sensitive and plant lectins which are glycoprotien complexes extracted from seeds of Phaseolus vulgaris (Kidney beans and from the bacterium Proteus vulgaris (Bowman and Rand, 1990). Plant lectin is used to test the capacity of lymphocytes to divide as a diagnostic aid in certain diseases of lymphoid tissue and in preparing chromosome spreads for examination of karyotype (chromosome pattern). Plant lectins also contain factors causing agglutination of red cells and precipitation of serum proteins. It has been tried in the treatment of aplastic anaemia (failure of bone marrow cells to proliferate), but it causes many adverse reactions and little positive response.
The ability of lectins to agglutinate red blood cells is used as a basis for assays of blood types. One of the lectins, convanavalin A, is a tetrameric glycoprotien which binds to glucose, mannoside or fructoside groupings in glycoproteins and glycolipids associated with the cell surface. This binding initiates cloning in T-cell lymphocytes (Bowman and Rand, 1990).
Phytohaemagglutinins extracted from seeds of Phaseolus vulgaris (kidney beans) has been shown to stimulate mitotic division of isolated lymphocytes. Plant lectins are is therefore used to test the capacity of lymphocytes to divide as a diagnostic aid in certain diseases of lymphoid tissue and in preparing chromosome spreads for examination of the karyotype. In addition to the factor causing stimulation of lymphocytes mitosis, plant lectins also contains factors causing agglutination of red cells and precipitation of serum protein. It has been tried in the treatment of aplastic anaemia (failure of bone marrow cells to proliferate), but it causes many adverse reactions and little positive response. (Bowman and Rand, 1990).
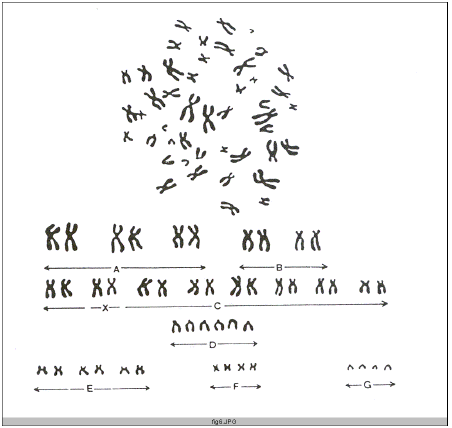
The karyotype, or chromosome pattern, can be examined during metaphase when the chromosomes have double but are still held together by their centromerse. Samples may be taken from tissues in which cells are normally actively dividing (e.g. bone marrow, the testes or neoplasm) or cell division can be produced in vitro in lymphocytes from a drop of blood (using phytohaemagglutinin to stimulate division), connective tissue fibroblasts from a small piece of skin (which may be stimulated by an embryo extract) or amniotic fluid cells. The cultured cells are treated with colchicines or one of its derivatives to cause arrest in metaphase (Fig. 7).
The chromosomes may be arranged, by convention, into seven groups (A to G) distinguished based on size and shape of chromosomes. Matched pairs are usually identifiable in groups A and E and are placed together where possible; otherwise, the chromosome of male karyotype is in the group G and can often be identified. The X chromosomes (two in females and one in males) are in group C and can not usually be identified except with special methods. Chromosomes other than sex chromosomes are termed autosomes (Bowman and Rand, 1990).
Possible use of plant lectins in biotechnology and transgenic plants
Plant lectins are used in two different areas of biotechnology. First, lectins can be used for the isolation of recombinant glycoproteins and second lectin genes can be expressed in transgenic plants either for the production of large quantities of lectins or to confer resistance against insect, nematodes or mammalian pests. (Gatehouse et al., 1995).
Other applications of plant lectins in biotechnology are based on the expression of lectin genes in transgenic (crop) plants. Some lectins may have beneficial effects on humans or animals when present in the diet, e.g. the snowdrop lectin strongly reduces the bacterial overgrowth in the rat gut by mannose-sensitive fimbriated E coli (Pusztai et al., 1993). The incorporation of sufficient snowdrop lectin in the diet of farm animals can only be achieved through the use of transgenic feed plants (e.g. soybean, cereals or oil seed rape), which over express the snowdrop lectin gene.
Most lectins with promising potential in crop protection are only found in wild species which cannot be crossed with our common crop plants. Therefore. lectin genes can be used as resistance factors only when transferred to and expressed in desired crop plants. For instance, genetically modified potato plants which express the snowdrop lectin genes at sufficiently high level are protected against sucking insects and nematodes (Hilder et al., 1995).
The use of a plant lectin, prohevin, the principal allergen of latex rubber, being coded into tomato plants for its fungicidal properties (Lee and Raikhel, 1995). The sudden onset of intolerance to tomatoes in previously stable individuals who are latex sensitive may provide substantial challenges to occupational physicians in the future.
Heterologous expression of genes for membrane proteins can provide useful approach to analyse ligand binding and other cell surface characterisitics. An analysis of the expression and processing of a barley lectin gene in mammalian cells showed that this cytotoxic plant lectin could be expressed in a functional state using a transient expression system (Audrey et al., 2000). The mammalian cells did not recognise all the processing signals on the lectin and as a result, the protein was secreted into the medium. The lectin expression studies suggest that it would be feasible to use the mammalian system for the expression and identification of plant genes encoding proteins that are able to bind the Nod factor, a bacterially-produced signal molecule required for the establishment of the legume-Rhizobium symbiosis (Audrey et al., 2000). A Nod factor-binding assay was developed and specific sets of transfected mammalian cells were shown to exhibit Nod factor- binding activity.
Plant lectin has been shown to influence pathogenicity of fungal species. The significance of mannose-substituted glycoprotiens is emphasized by the fact that the mannose-binding rice lectin can interfere with the development of the appressorium in Magnaporthe grisea which causes rice blast disease, one of the major diseases of cultivated rice (Teroaka et al., 2000). This appressorium is important in fungal pathogenicity and field establishment of infection (Ou, 1985).
Lectin toxicity
Although lectins are a group of substances, which have been recognized because of their ability to agglutinate red blood cells, some of these substances are also highly toxic to animals. For example, lectins isolated from black beans produce growth retardation when fed to rats at 0.5% of the diet and lectins from kidney beans produce death in rats fed on lectins at 0.5% of the diet for 2 weeks (Goldstein et al., 1986). Ricin, a lectin from the castor oil bean, is one of the most toxic natural substances with an LD 50 by injection of 0.05 mg/kg. Because of their high toxicity, castor beans (not a legume) must be thoroughly heated to deactivate their ricin before they can be used as animal feed (Takayuki and Bjeldanes, 1993).
Peanut (Arachis hypogeae), agglutinin (PNA), stimulates proliferation of rectal mucosa (Ryder et al., 1998). Intact PNA can be detected at up to 5 ug/mL in the systemic circulation after eating peanuts. Similar concentration of PNA stimulates proliferation of epithelial cells and detection of lectin with a haemagglutination assay implies that it has retained biological activity. The rapid appearance of the lectin in the blood after ingestion suggests that uptake occurs high in the gastrointestinal tract, probably in the stomach, in which there are any PNA receptors. Previous studies have shown phytohaemagglutinin, a toxic dietary lectin from red kidney beans and tomato lectin, in the systemic circulation after ingestion (Kilpatrick et al., 1985). Although concentrations in blood of individual lectins are likely to be low, the overall effect of the uptake of dietary lectins into the circulating might lead to important interactions with circulation and cellular glycoproteins and therefore, affect cell-to-cell adhesion and proliferation (Quiang et al., 1998).
Lectins are toxic, inflammatory and resistant to cooking and digestive enzymes present in much of our food (Takayuki and Bjeldanes, 1993). The mechanism by which lectins produce an ultimate toxic effect is also open to controversy. It is well established that lectins from various sources present on the intestinal epithelium adsorb nutrients and thus reduce the absorption of those nutrients by the intestine. The resulting inefficient use of nutrients may in itself account for the poor growth promoted by diets rich in uncooked legumes. This effect may also magnify the protein losses induced by pancreatic hypersecretion caused by trypsin inhibitors also present in the legumes.
The microflora of the gut also play a role in legume-and lectin-induced toxicity. Germ-free birds (birds free of intestinal bacteria) used as test species show less growth depression when fed raw legumes or isolated lectins than do conventional birds Taylor and Kennewell, 1993). For example, diets containing raw jack beans meal produce high mortality in Japanese quail. However, germ-free birds exhibit no toxic effects under exactly the same experimental conditions; these observations have led some investigators to suggest that the lectins may impair the body’s defence system against bacterial infection, resulting in an increased tendency for an invasion by gut and other bacterial flora (Watson, 1987).
A number of significant clinical conditions are caused by recognized food components containing lectins eg glutens contains gliaden which is recognized to be one of the main components of coeliac disease (Pustzai et al., 1989). Wheat gliaden causes coelic disease, contains a lectin like substance that binds to intestinal mucosa (Kolberg and Sollid 1985).
Food avoidance treats celiac disease, but wheat lectins are also found bound to capillary walls in the kidney glomerulus and tubule and has been shown in rodents to bind immunoglubulin A (IgA) and can induce IgA mesangial deposits. IgA nephropathy can be managed partially by the use of a gluten free diet. Austistic children are IgA deficient (Etzler, 1986).
Detoxcification of the toxicity of lectins in animal feed has been done directly by thermal deactivation or by intra-venous administration of antitoxicants in infected rats (Takayuki and Bjeldanes, 1993). Indirect detoxification by the introduction of low toxic cultivar for animal feed through selective breeding methods and biotechnology via transgenesis is also possible (Gatehouse et al., 1995).
Plant lectins plays an important role in the understanding of carbohydrate chemistry and histology. Its significance as a communication protein and in biological signalling is well established.
The ability for lectins to agglutinate red blood cells has made a significant contribution for assays of blood types, study of chromosome patterns which is vital in the treatment of certain human diseases such as aplastic anaemia (failure of bone marrow cells to proliferate).
Plant lectins have contributed in biotechnology and transgenic plants through isolation of recombinant glycoproteins and the expression of lectin genes in transgenic plants for the production of either large quantities of lectins or to confer resistance against insects, nematodes or mammalian pests. This biotechnological input has contributed in the improvement of animal feed production and increased yield of food crop, which is an attribute for sustainable food crop production.