Research Article
In vitro Direct Shoot Organogenesis and Regeneration of Plantlets from Leaf Explants of Sentang (Azadirachta excelsa)
Multimedia University (Melaka Campus)
Rofina Yasmin Othman
The University of Malaya, Malaysia
Azadirachta excelsa (A. excelsa) is an indigenous forest species found in South East Asia. It belongs to the family Meliaceae. A. excelsa is suitable for use as timber and was found to be one of the fastest growing tree species in lowland tropical forests with an average girth growth rate of 8 to 10 cm per year (Ng and Tang, 1974; Banasopit et al., (un published report) and has been designated as a priority forest genetic resource species for South East Asia (Koskela et al., 2002). The rapid expansion of forest plantation in this region has boosted demand for uniform high quality planting stocks of A. excelsa. Currently, planting stocks are supplied in the form of rooted cuttings or seeds. The method of rooting of cuttings is limited by the availability of donor plants (ortets). Another limiting factor is the rapid maturation of the ortets (Zuhaidi and Mohd Noor, 1997). In most forest tree species, the true-to-type propagation by the rooting of cuttings is possible only when the ortets are in a juvenile state. In addition, A. excelsa seeds do not store well and are recalcitrant. The seeds collected from trees need to be sown immediately to preserve its germination viability (Jøker, 2000). Therefore A. excelsa seeds are not suitable to be distributed widely as planting stock. Plantlets derived from seed also tend to exhibit a wide range of variability in phenotypic appearance. There are also no breeding trials or known provenance trials for A. excelsa. Current planting materials originate almost exclusively from unselected trees (Jøker, 2000). In vitro propagation provides a potential means of supplying a large number of high quality and uniform A. excelsa planting stock to the plantation. In this study, in vitro propagation via direct shoot regeneration from leaf cuttings to produce A. excelsa plantlets was investigated.
The study was conducted at the laboratory of University Malaya and tissue culture laboratory at TropBio Research Sdn Bhd between the year 2000 and 2002.
Shoot tips were excised from six months old A. excelsa plants grown in the nursery and used as explants. The explants were surfaced sterilized by washing with FlowLab non-toxic detergent followed by stirring in 0.125 mg L-1 Benlate for 1 h. Then, the explants were submerged in 70% ethanol for 1 min followed by 15 min agitation in 20% Chlorox with a few drops of Tween-20 added. The explants were rinsed once with sterilized distilled water. These were further submerged in 30% Chlorox plus a few drops of Tween-20 for 15 min. The explants were then rinsed three times with sterilized distilled water. The sterilized shoot tips were then cultured in Murashige and Skoog (MS) media supplemented with 1 mg L-1 benzylaminopurine (BAP).
Leaves were excised from the expanding A. excelsa shoots after 4 months in culture. These leaves were cut into 1 cm2 pieces before culturing on MS medium supplemented with a range of BAP, kinetin and adenine sulphate concentrations. Each petri dish contained 10 leaf cuttings and there were 2 petri dishes per treatment. The cultures were placed in a 30±2°C growth incubator at continuous darkness.
Subsequently, these in vitro shoots (>4 cm in height) were used for root induction experiments. These shoots were culture in MS media with varying concentrations of (1, 2, 5 and 10 mg L-1) α-naphthaleneacetic acid (NAA) or (1, 2, 5 and 10 mg L-1) indole-3-butyric acid (IBA). Rooted plantlets were removed from the culture vessels and rinsed under running water to wash off any traces of agar attached to the roots. These were then planted in plastic trays (with transparent covers) and placed in a 30±2°C growth incubator.
RESULTS
Formation of callus was observed on the cut edges of leaf pieces after 3 weeks of incubation in continuous darkness (Fig. 1a). The leaves curled but did not expand. The calluses were friable and white, however, these turned green upon exposure to light. Some of the calluses grew into globular shaped structures that eventually became shoot primordias after 5 weeks of culture (Fig. 1b). At 7 weeks the primordial shoots can be clearly seen (Fig. 1c) with formation of true leaves first observed at 11 weeks (Fig. 1d). Figure 1e shows the subsequent development of the primordias into shoots.
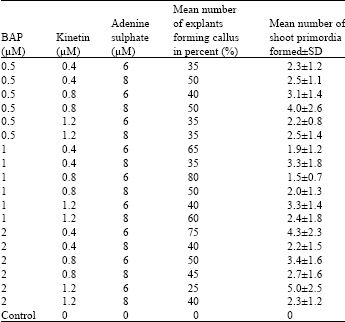
Analysis of results shows that a significantly higher mean number of shoots were formed on MS medium supplemented with 2 mg L-1 BAP, 1.2 mg L-1 kinetin and 6 mg L-1 adenine sulphate (p<0.001, n = 10). An average of up to 5 shoot primordias per explant was obtained after 12 weeks of culture (Table 1).
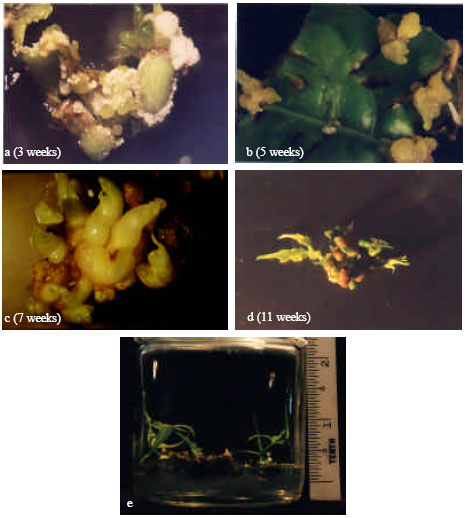 | |
| Fig. 1: | Induction of shoots from leaf cuttings-(a) Development of callus on the edges of leaf cutting. (b) Some protruding structures start to form (c) Formation of primordia shoots (d) Formation of true leaves (e) Development of primordia shoots into shoots. Time from leaf culture is shown in parenthesis |
| Table 1: | Callus and shoot formation from leaf explants cultured on various concentrations of BAP, kinetin and adenine sulphate (After 12 weeks of culture) |
 | |
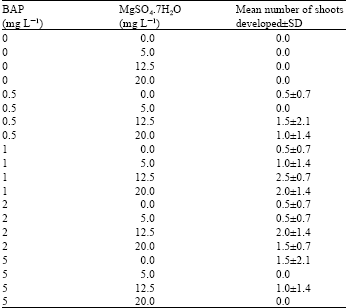
| Table 2: | Shoots developed from shoot primordial cultured on various concentrations of BAP and magnesium sulphate |
 | |
However, the highest percentage number of leaves that formed callus was recorded at 80% with the application of 1 mg L-1 BAP, 0.8 mg L-1 kinetin and 6 mg L-1 adenine sulphate.
The shoot primordias were transferred onto MS medium supplemented with various concentrations of BAP for shoot development and elongation. Various concentrations of magnesium sulphate were tested, because in preliminary tests, application of higher concentrations of this mineral has been observed to produce better shoot growth in A. excelsa (Data not shown). Overall, the mean number of shoot primordias that developed into complete shoots upon culture on all media was low.
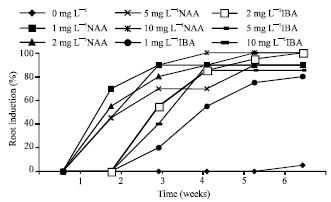 | |
| Fig. 2: | Roots formation with the application of NAA or IBA |
 | |
| Fig. 3: | Formation of in vitro plantlet ready for weaning |
The highest mean number of shoots obtained was 2.5 using medium with 1 mg L-1 BAP and 12.5 mg L-1 magnesium sulphate (Table 2).
Shoots formed were transferred to MS medium supplemented with various concentrations of NAA or IBA. The highest percent of shoots formed roots on medium supplemented with 10 mg L-1 IBA after 4 weeks (Fig. 2). A higher number of shoots were rooted earlier (approximately 1 week) on medium supplemented with NAA than with IBA.
Rooted plantlets (Fig. 3) were removed from culture vessels and roots washed to remove agar residual prior to planting in a potting tray containing most peat (crushed Jiffy material). Plants were placed in a 30°C growth chamber. All (100 %) of the rooted plantlets had survived at the end of the first month. All of them had developed new leaves and appeared healthy.
Present results show that induction of shoots from leaf cuttings by direct shoot regeneration is a viable means of producing a potentially large number of genetically identical individuals of A. excelsa. The present study found that the highest shoot number induced was five shoots per leaf explant under the optimized conditions described earlier. Forty percent of these shoots developed into plantlets. Although a higher percentage of plantlets (64.1%) has been reported in shoots induced from A. indica leaf explants (Eeswara et al., 1998) the difference may be due to the different genetic background of the trees and reaffirms the need for developing individualized protocols for different species.
It can be seen from the results obtained that only a low number of plantlets (average 2.5) are successfully formed per leaf explant. Effort therefore needed to be made to increase the number of plantlets formed. Since leaf explant can be obtained in abundant supply, one strategy that can be adopted in future is to use liquid culture to produce the in vitro plantlets. In liquid culture, a large number of leaf explants can be used as starting materials for the in vitro shoot production.
Successful application of clonal propagation depends heavily on the assumption that the genotypes propagated are identical to the selected trees with desirable characteristics. While in vitro shoot culture has been known to produce “true-to-type” planting materials (Vasil and Vasil, 1980; Ahuja and Muhs, 1985), the multiplication rate was found to be low and supply of shoot tips as starting material can be limiting. The in vitro culture by shoot organogenesis therefore will be potentially useful for providing future supply of clonal propagules of A. excelsa due to an abundance of leaves available as starting material.
This study was supported by the Malaysian Teaching Company Scheme (MTCS) grant from the Ministry of Science, Technology and Environment, Malaysia. We would like to thank Dr. Kodiswaran for reviewing the manuscript.