Research Article
Studies of Some Molecular Properties of the Vacuolar H+-ATPase in Rainbow Trout (Oncorhynchus mykiss)
Department of Biological Sciences, Faculty of Science-King Abdulaziz University, P.O. Box 80094-Jeddah 21589 Saudi Arabia
The vacuolar H+-ATPase (H+ V-ATPase) or proton pump is probably expressed in all eukaryote cells where it plays a housekeeping role in the acidification of intracellular organelles (Nelson, 1992). However, in addition to its housekeeping role, the H+ V-ATPase is thought to be specifically involved in acid-base balance and ionic regulation in a variety of secretory epithelia (Stevens and Forgac, 1997; Forgac, 1998; Nelson and Harvey 1999; Wieczorek et al., 1999; AL-Fifi et al., 1998; Choe et al., 2002, 2004) including the rainbow trout (Oncorhynchus mykiss) gill (Lin and Randall, 1995). Avella and Bornancin (1989) first suggested a physiological role for the H+ V-ATPase in the fish gill. Specifically, it was reasoned using thermodynamic arguments that the traditional model for Na+ uptake across the freshwater fish gill, involving electroneutral Na+/H+ exchange (Krogh, 1938; Choe et al., 2004), was not tenable. Instead, it was postulated (Avella and Bornancin, 1989) that Na+ uptake across the apical membrane of gill epithelial cells was linked energetically to active H+ extrusion via the H+ V-ATPase. According to their model, H+ secretion across the apical membrane establishes a favourable electrochemical gradient that permits the inward entry of Na+ through epithelial Na+ channels (AL-Fifi et al., 1998, 2002).
Despite scarce empirical evidence for apical membrane Na+ channels, this newer model for Na+ uptake is now generally accepted (Marshall, 1995). Less certain, however, is the epithelial location of the fish gill the H+ V-ATPase. Indeed, arguments have been made for a specific localisation to the chloride cell (Lin and Randall, 1991), the pavement cell (Laurent et al., 1994; Sullivan et al., 1995; Kultz and Somero, 1995; Sullivan et al. 1996) or both cell types (Lin et al., 1994; Lin and Randall, 1995; Perry, 1997). Regardless of its location, evidence is accruing that the activity of the H+ V-ATPase in the fish gill is regulated in accordance with acid-base and ionic uptake requirements (Lin and Randall, 1995; Perry and Fryer, 1997; Choe et al., 2002, 2004). For example, H+ V-ATPase activity is increased in the trout gill during respiratory acidosis (Lin and Randall, 1993; Sullivan et al., 1995) and decreased during seawater exposure (Lin and Randall, 1993). Because cortisol (Lin and Randall, 1993) and growth hormone (Perry and Fryer, 1997) are known to influence branchial H+ V-ATPase activity, the reported changes in its activity during environmental changes may reflect hormonal regulation. Changes in H+ V-ATPase activity could reflect transcriptional or post-transcriptional regulation of mRNA levels and/or translational or post- translational regulation of H+ V-ATPase protein activity/localisation. Sullivan et al. (1996) provided evidence in support of transcriptional/post-transcriptional regulation by demonstrating, using in situ hybridisation, that the levels of H+ V- ATPase mRNA were increased during exposure of rainbow trout to external hypercapnia. That study, however, was limited by the use of a heterologous oligonucleotide probe that was derived from the sequence of bovine renal H+ V- ATPase E subunit. Recently, the B sub-unit of the H+ V-ATPase was cloned from trout gill (Perry et al., 2000) and thus an homologous cDNA probe is now available to quantitatively assess the levels of H+ V-ATPase mRNA.
In this study, we have used the previously cloned H+ V-ATPase cDNA to: i) establish its inter-specific distribution among marine and freshwater species using northern analysis and ii) assess its expression in rainbow trout gill during a variety of acid-base, ionic and hormonal disturbances using slot blot analysis.
Experimental animals: Rainbow trout (Oncorhynchus mykiss Walbaum; 200-300 g, <2 years old) of both sexes were obtained from Linwood Acres Trout Farm. American eels, Anguilla rostrata, of either sex (~ 500 g) and brown bullhead catfish, Ictalurus nebulosus, (~ 300 g) were obtained from a commercial supplier (Lancaster Ontario) and were transported on ice (eels) or in oxygenated water (bullhead) to University of Ottawa. All species were held indoors (using a 12:12 h light-dark photoperiod cycle) in large 1300 l fiberglass tanks supplied with flowing, aerated and dechlorinated tap water. Trout and eel were maintained at a temperature of 14°C, bullhead were kept at 20°C. While trout and bullhead were fed commercial diets ad libitum on alternate days, eels were not fed.
In addition, total RNA was obtained (courtesy of Dr. Pat Walsh, University of Miami) from the following species; winter flounder (Pseudopleuronectes americanus), Pacific hagfish (Eptatretus stoutii), lingcod (Ophiodon elongatus), long-horned sculpin (Myoxocephalus octodecimspinosus), Lake Magadi tilapia (Oroechromis alcalicus Grahami), plainfin midshipman (Porichthys notatus), bigfin eelpout (L. cortezianus), Dover sole (Microstomus pacificus), shiner perch (Cymatogaster aggregata), green cod (T. macellus), gulf toadfish (Opsanus beta), brown Irish lord (H. spinosus), black finned poacher (Bathyagonus nigripinnus) and lumpfish (Cyclopterus lumpus).
Experimental protocol: In addition to a broad species survey and an assessment of H+ V-ATPase tissue distribution in rainbow trout, three separate series of experiments were performed.
Exposure to external hypercapnia: To induce rapid respiratory acidosis, separate groups of fish (N = 6 in each group) were exposed to external hypercapnia for 1, 2 or 3 h. Hypercapnia was achieved by gassing a water equilibration column with 1.3% CO2 in air (Cameron flowmeter) to reach a final water PCO2 (PwCO2) of ~7.5 mm Hg. The PwCO2 was monitored continuously using a CO2 electrode and associated meter (Cameron Instruments Inc.). Deviations in PwCO2 from the target of 7.5 mm Hg were corrected by adjustments of gas and/or water flows through the equilibration column. Control fish were subjected to continuing normocapnia for 1- 3 h. At 0, 1, 2 and 3 h, fish were euthanised and perfused with saline to remove blood from the tissues (Perry et al., 2000). Tissues were removed, frozen in liquid N2 and stored at 80°C until total RNA was prepared.
Exposure to ion-poor water: Ion-poor water was prepared by mixing tap water with deionised water derived from a reverse osmosis unit. Regular tap water and the ion-poor water were analysed daily for Na+, Ca2+ and K+ levels using flame emission spectroscopy (Varian model 250 atomic absorption spectrophotometer). For regular tap water, the ion concentrations (mmol L-1) were Na+ = 0.135, Ca2+ = 0.391 and K+ = 0.025. For ion-poor water, the ion concentrations (mmol L-1) were Na+ = 0.03-0.06, Ca2+ = 0.09-0.13 and K+ = 0.006-0.009. Fish were not fed during the duration (6-72 h) of the ion-poor water treatment. Fish were sacrificed (N = 6 in each group) after 6, 12, 24, 48 and 72 h of exposure to ionpoor water.
Cortisol treatment: Fish were anaesthetised in a 1:12,000 (weight/volume) solution of benzocaine (ethyl-p-aminobenzoate) cooled to 10°C. After cessation of breathing movements, the fish was transferred to an operating table and the gills were irrigated with the same anaesthetic solution throughout the brief period (<1 min) required to inject cortisol implants. To permit chronic elevation of circulating cortisol levels to a target level of 200-300 ng mL-1, fish were injected intraperitoneally with 150 mg kg-1 body weight (injection volume = 2 ml kg-1) of cortisol (hemisuccinate salt) dissolved in coconut oil (Perry and Reid, 1994). Control fish were injected with equivalent volumes of coconut oil. Control and cortisol-treated fish were kept in separate tanks (100 L) for 4 days prior to blood sampling and tissue removal.
Fish were killed by a blow to the head and a blood sample (~1 mL) was rapidly withdrawn from the caudal vessels by percutaneous puncture. After centrifugation (30 sec at 12,000 G), plasma was removed and frozen (20°C) for subsequent determination of cortisol levels. Tissues were harvested and immediately frozen in liquid nitrogen prior to storage at 80°C.
RNA isolation, gel electrophoresis and Northern analysis: Total RNA was isolated by homogenisation in guanidinium thiocyanate (Chomcyzynski and Sacchi, 1987) as modified by Chomcyzynski and Mackey (1995).
Northern analysis was performed using an 810 bp homologous H+ VATPase cDNA plasmid insert as the probe (Perry et al., 2000). RNA samples (20±g) were incubated in loading buffer at 65°C and electrophoresed through 1.5% (w/v) agarose gels in MOPS (morpholinopropanesulfonic acid) buffer containing 0.6 mol L-1 formaldehyde as described in Sambrook et al. (1989) and then transferred to GeneScreen+ membranes (NEN Life Sciences) by capillary action (Sambrook et al., 1989). Membranes were pre-hybridised at 65°C for 2-4 h in a buffer containing 6 X SSC (0.9 mol L-1 NaCl, 0.09 mol L-1 sodium citrate, pH 7.0), 5 X Denhardt’s (1X Denhardt’s is 0.1% Ficoll 400,000, 0.1% polyvinylpyrrolidone, 0.1% bovine serum albumin), 100 μg mL-1 single-stranded herring sperm DNA, 1% Sodium Dodecyl Sulfate (SDS) and 10% dextran sulfate. After the addition of probe to a concentration of 1-2x106 cpm mL-1, hybridisation proceeded for 16 h at 65°C in the same solution. Following hybridisation, the membranes were washed several times at 65°C with 0.1 X SSC, 0.1% SDS and exposed to BioMax film plus intensifying screen (Kodak) at 80°C for up to 4 days.
In order to confirm equal loading between samples, membranes were re-probed with a homologous β-actin probe (a 514 bp PCR fragment corresponding to 63- 576 of the trout β-actin sequence; GenBank accession #AF254414) under similar conditions but with an exposure time of hours.
DNA for probes was labelled with [-32P] dCTP (Amersham, 370 MBq mL-1) by the random-primer method (Feinberg and Vogelstein, 1983). Unincorporated nucleotides were removed by spin columns (Sambrook et al., 1989). Prior to use, the labelled DNA was denatured by boiling for 5 min and then quick chilled in ice.
Slot blot analysis: A slot blot apparatus (Schleicher and Schuell, Minifold II) was used for semiquantitative analysis of H+ V-ATPase mRNA levels in fish exposed to (treated with) hypercapnia, ion-poor water or cortisol. A preliminary series of experiments revealed that the ratio of H+ V-ATPase/β-actin phosphorescence was constant only over a relatively narrow range of total RNA loaded (0.5-2.0 μg). Consequently, all subsequent slot blot experiments used loading quantities of 0.5 and 1.0±g total RNA; samples were loaded in duplicate. Thus, for any given sample, an average H+ V-ATPase/β-actin phosphorescence ratio was obtained using the mean data from both loadings.
After hybridisation and washing, membranes were exposed to a phosphor screen for >20 h. The screens were subsequently scanned (BioRad Molecular Imager FX) and the signals were quantified using associated BioRad software. Replicate membranes were probed in a similar manner with radiolabelled β-actin but were exposed to the phosphor screens for 30-45 min.
Cortisol analysis: Plasma cortisol concentrations were measured on duplicate samples using a commercial radioimmunoassay kit (ICN).
Statistical analysis: All data are represented as means±1 SEM unless otherwise stated. Data were analysed using one-way ANOVA followed by Bonferroni’s multiple comparison. P-values<0.05 were considered to be statistically significant. Calculations were performed using the SigmaStat (SPSS; version 3.0) software package.
Northern blots of gill total RNA, performed under conditions of high stringency, revealed cross hybridisation between the trout probe and 11 of 16 species that were examined. Cross hybridisation was not observed in the pacific hagfish (Eptatretus stoutii), Lake Magadi tilapia (Oroechromis alcalicus Grahami), bigfin eelpout (Lycodes cortezianus), blackfin poacher (Bathyagonus nigripinnus) or freshwater American eel (Anguilla rostrata). A representative Northern blot showing distribution of the H+ V-ATPase amongst selected fish species is depicted in Fig. 1. The lack of detectable cross hybridisation in several of the species cannot be explained by inadequate RNA loading (based on the more-or-less equivalent intensities of the 18 and 28 S ribosomal RNA bands;data not shown) but may reflect insufficient sequence homology given the stringent hybridisation conditions that were used. For example, the trout gill cDNA probe that was used in this study shares 83-85% nucleotide identity with similar regions of the eel swim bladder gas gland tissue H+ V-ATPase B2 and B1 isoforms, respectively (Niederstaetter and Pelster, 2000).
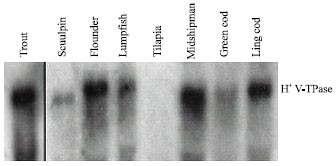 | |
| Fig. 1: | Representative Northern blot of gill tissue from selected fish species showing the degree of cross hybridisation of vacuolar H+ V-ATPase B subunit mRNA. Transfers were hybridized with a trout H+ V-ATPase B subunit cDNA probe (810 bp). Gels were loaded with 20 μg per lane of total RNA obtained from individual fish |
It is unclear as to whether this dissimilarity in nucleotide sequence would prevent hybridization under stringent conditions.
A representative Northern blot depicting tissue distribution of H+ V-ATPase B subunit mRNA is illustrated in Fig. 2. Detectable levels of H+-ATPase B subunit mRNA were observed in all tissues that were examined. H+ V-ATPase mRNA expression was high in the gill, kidney (anterior or posterior), intestine, heart, spleen and blood but lower in liver and white muscle.
Exposure of rainbow trout to external hypercapnia caused a transient elevation of gill H+ V-ATPase mRNA levels at 1 h (Fig. 3). These results are consistent with data from a preliminary study (Perry et al., 2000) that demonstrated a trend toward elevated gill H+ V-ATPase mRNA levels after 2 h of hypercapnia. It is likely, therefore, that the increased activity of branchial H+ V-ATPase observed in trout during hypercapnia (Lin and Randall, 1993; Sullivan et al., 1995) is caused, at least in part, by an increase in the steady-state levels of mRNA. The most probable cause of the elevated mRNA levels is an increase in gene transcription. The physiological benefit of increased branchial H+ V-ATPase activity during hypercapnia is an associated increase in acid excretion by the gill (Sullivan et al., 1995) that serves to raise internal pH and thus correct the hypercapnicPlasma cortisol levels were elevated in the hypercapnic fish (from 45±15 to 83±4 ng mL-1) and because cortisol was previously implicated as a regulator of H+-ATPase activity (Lin and Randall, 1993), mRNA levels were quantified in fish subjected to chronic cortisol acidosis.
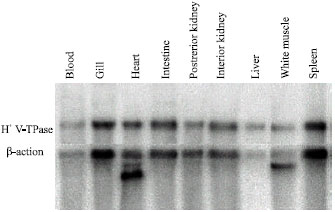 | |
| Fig. 2: | Northern blot of RNA isolated from selected rainbow trout (Oncorhynchus mykiss) tissues hybridized with a homologous H+ VATPase B subunit cDNA probe (810 bp) and/or a trout β-actin clone. Gels were loaded with 20 μg per lane of total RNA obtained from perfused fish (i.e., blood was removed from tissue via saline perfusion |
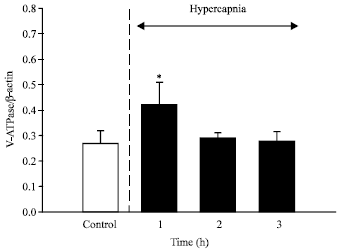 | |
| Fig. 3: | The temporal effects of external hypercapnia (N = 6 at each sampling time) on the levels of vacuolar H+ V-ATPase B subunit mRNA in trout (Oncorhynchus mykiss) gill tissue. Transfers were hybridized with a homologous H+ V-ATPase B subunit cDNA probe (810 bp) or with a homologous ß-actin clone. Slot blots were loaded with 0.5 or 1.0 μg of total RNA obtained from perfused fish. * Denotes a significant difference from the control value |
Although debated, the cell type that is responsible for increased H+ excretion during respiratory acidosis excretion is probably the pavement cell rather than the chloride cell (Goss et al., 1992; 1995; 1998; Laurent et al., 1994; Choe et al., 2004). elevation.
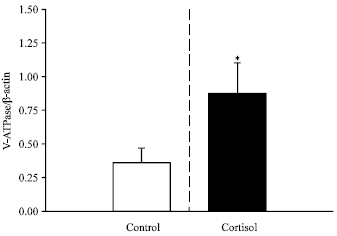 | |
| Fig. 4: | The effects of chronic cortisol elevation (N = 6) on the levels of vacuolar H+ V-ATPase B subunit mRNA in trout (Oncorhynchus mykiss) gill tissue. Transfers were hybridized with a homologous H+ V-ATPase B subunit cDNA probe (810 bp) or with a homologous ß-actin clone. Slot blots were loaded with 0.5 or 1.0 μg of total RNA obtained from perfused fish. * Denotes a significant difference from the control value |
An increase in plasma cortisol concentration from 90±10 (sham implants) to 300±60 ng mL-1 (cortisolimplants) for 4 days was associated with an approximate doubling of gill H+- ATPase mRNA levels (Fig. 4). Thus, it is conceivable that the rise in cortisol levels during hypercapnia may act as a trigger for increased H+ V-ATPase gene expression.
Although a previous study has examined the impact of elevating the ionic composition of freshwater on branchial H+ V-ATPase activity (Lin and Randall, 1993), this is the first study to address the effects of reducing the ionic strength of water. Figure 5 shows that exposure of trout to ion-poor water was associated with a pronounced and sustained decrease in the levels of H+ V-ATPase mRNA in gill tissue. Under normal conditions, there is an obligate linkage between H+ excretion and Na+ uptake. In the present study, the level of Na+ in the ambient water was lowered to values well below the Km of the Na+ uptake mechanism and consequently Na+ uptake is depressed under such conditions (Perry and Laurent, 1989). Thus, the decrease in gill H+ V-ATPase mRNA levels during exposure to ion-poor water may simply reflect a decreased need to excrete H+ in the face of lowered Na+ uptake rates. Interestingly, exposure of trout to ion-poor water is accompanied by proliferation of chloride cells on gill lamellae (Perry and Laurent, 1989). Thus, the decrease in H+ VATPase mRNA levels at such times is further evidence that the pavement cell may be a more important site of H+ V-ATPase expression.
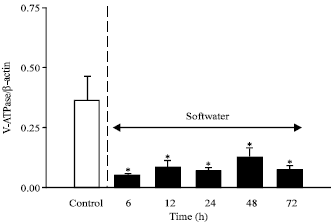 | |
| Fig. 5: | The temporal effects of exposure to ion poor water (N = 6 at each sampling time) on the levels of vacuolar H+ V-ATPase B subunit mRNA in trout (Oncorhynchus mykiss) gill tissue. Transfers were hybridized with a homologous H+ V-ATPase B subunit cDNA probe (810 bp) or with a homologous β-actin clone. Slot blots were loaded with 0.5 or 1.0 μg of total RNA obtained from perfused fish. * Denotes a significant difference from the control value |