Research Article
Micropropagation of Curcuma zedoaria Roscoe and Zingiber zerumbet Smith
University sains Malaysia, Malaysia
Chan Lai Keng
University sains Malaysia, Malaysia
Curcuma zedoaria Roscoe and Zingiber zerumbet Smith are two important medicinal plants of the family Zingiberaceae. Curcuma zedoaria has been used as a stimulant, carminative, expectorant, demulcent, diuretic and rubifacient (Kapoor, 1990). In addition to these, it has been reported to have analgesic (Ali et al., 2004), hepatoprotective (Kim et al., 2005), anti-inflammatory (Yoshoika et al., 1998), anti-allergic (Matsuda et al., 2004), anti-metastatic (Seo et al., 2005) and anti-oxidant (Mau et al., 2003) activities. In South East Asia, Z. zerumbet has been traditionally used for the treatment of fever, constipation and to relieve pain. It possess anti pyretic and analgesic properties (Somchit et al., 2005). It also possess anti-inflammatory (Somchit and Shukriyah, 2003) and chemo-preventive activities (Nakamura et al., 2004).
C. zedoaria and Z. zerumbet are propagated vegetatively by underground rhizomes at a slow rate. Besides, Zingiberaceae plants are frequently infected with pathogens (Chan and Thong, 2004), such as rhizome rot caused by Pythium species and the leaf spot due to Coleotrichum species. This had also posed problems for the conservation and storage of germplasm of other Zingiberaceae species such as Curcuma aromatica (Nayak, 2000). The objective of this study is to establish a micropropagation protocol that can be applied to both C. zedoaria and Z. zerumbet for the germplasm conservation of these two species.
Establishment of aseptic explants: The rhizomes of C. zedoaria and Z. zerumbet were collected during rainy season from various parts of Penang, Malaysia since 2005. Buds from the rhizomes were used as explants. The bud explants were thoroughly washed with detergent several times and rinsed under running tap water for 30 min. They were then surface sterilized with HgCl2 solution (100 mg L-1) under continuous agitation for 5 min followed by rinsing three times with sterile distilled water.
The buds were subsequently surface sterilized in 20% Clorox® with a few drops of Tween 20, a wetting agent under constant agitation for 10 min, followed by washing three times with sterile distilled water. The buds were further surface sterilized in 10% Clorox® with few drops of Tween 20 for 10 min and again rinsed three times with sterile distilled water. They were then inoculated into 350 mL glass jar containing MS (Murashige and Skoog, 1962) medium supplemented with 30 g L-1 sucrose and 7.5 g L-1 agar (Algas, Chile) for 14 days. The pH of the medium was adjusted to 5.7-5.8 prior to autoclave at 121°C and 1.06 kg cm-2 for 13 min. All the cultures were maintained at 25±2°C in a culture room with continuous lighting provided with cool white fluorescent lamps at 35 μmol m-2 s-1.
After 14 days, the aseptic buds of both species were transferred onto MS medium supplemented with 2 mg L-1 BA and 2 mg L-1 IBA, the optimum proliferation medium, formulated for Zingiber officinale and other Zingiberaceae species by Chan and Thong (2004) for shoot multiplication for 6 weeks. The multiple shoots formed were separated and transferred into MS medium devoid of plant growth regulators for another 4 weeks and were used for all subsequent experiments.
Induction of multiple shoots: The aseptic shoots of C. zedoaria and Z. zerumbet were trimmed of new leaves and roots and inoculated onto MS medium supplemented with different concentration of benzyladenine (BA) and 3-indole butyric acid (IBA) ranging from 0, 2, 4, 6, 8, 10 mg L-1 in 6x6 factorial block design. A single shoot was cultured into each 350 mL glass jar and 8 experimental units were used for each combination medium. The number of shoots formed from each shoot explant was recorded after 4 weeks of culture. Data obtained were analyzed using two-way ANOVA and the means were compared using Tukey’s HSD test at p = 0.05.
Effect of reduced concentration of BA and IBA on shoot proliferation of C. zedoaria and Z. zerumbet: The aseptic shoots of C. zedoaria were trimmed of new leaves and roots and cultured on MS medium supplemented with lower concentration of BA and IBA ranging from 0.0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0 mg L-1 in 7x7 factorial block design. A single shoot was cultured into a 350 mL glass jar and 8 experimental units were for each combination medium. The number of shoots produced was recorded after 4 weeks. Data obtained were analyzed using two-way ANOVA and the means were compared using Tukey’s HSD test at p = 0.05. The optimum medium obtained for the multiple shoot formation of C. zedoaria (MS + 0.5 mg L-1 BA and 0.5 mg L-1 IBA) was applied to Z. zerumbet by culturing the aseptic shoots of Z. zerumbet onto this medium. The aseptic shoots were also cultured on MS medium supplemented with 6 mg L-1 BA, the best shoot proliferation medium for Z. zerumbet, determined in the previous section. A single shoot was inoculated into a 350 mL glass jar and 8 experimental units were used for each medium. The number of shoots produced was recorded after 4 weeks of culture. Data obtained were analyzed using independent t-test at p = 0.05 to determined whether lower concentration of BA and IBA could still be used to induce formation of multiple shoots from Z. zerumbet shoot explants.
Effect of liquid medium on the shoot proliferation of C. zedoaria and Z. zerumbet: The aseptic shoots of C. zedoaria and Z. zerumbet were trimmed of new leaves and roots and were cultured in liquid MS medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA in 100 mL conical flasks and 8 units were used for each species and a single shoot was inoculated in each flask. The aseptic shoots of each species were also cultured on MS solid medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA. A single shoot was inoculated into a 350 mL jam jar and 8 experimental units were used for each species. The number shoots produced from each shoot from the liquid medium and the solid medium was recorded after 4 weeks of culture and the means were compared using independent t-test at p = 0.05.
Effect of divided and undivided shoots on shoot proliferation: In vitro aseptic shoots of C. zedoaria and Z. zerumbet were longitudinally divided into two and cultured into 100 mL conical flask containing MS liquid medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA. A single half shoot or divided shoot was inoculated in each flask and 8 experimental units were used for each species. Undivided aseptic shoots were also cultured into MS liquid medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA and 8 experimental units were used for each species. The number of shoots formed from each explant of each species was determined after 4 weeks of culture. The data was analyzed using independent t test at p = 0.05 to determine the effect of divided and undivided shoots on shoot proliferation for each of the species.
Acclimatization: In vitro rooted plantlets of C. zedoaria and Z. zerumbet were removed from the solid MS medium and the roots were washed under running tap water to remove any remaining agar. Each plantlet was then planted into pots containing mixture of organic soil and sand (1: 1) without direct sunlight. The percentage of surviving plantlets was recorded after 4 weeks of transferring.
The buds from the rhizomes of C. zedoaria and Z. zerumbet obtained from the field that were surface sterilized with 100 mg L-1 mercury chloride and 20% Clorox® solution for 10 min in the first stage followed by 10% Clorox® solution for 10 min in the second stage could establish more than 80% aseptic and surviving explants. Chan and Thong (2004) also reported that the use of mercury chloride with two-stage surface sterilization using Clorox® solution was very effective for establishing aseptic buds of other Zingiberaceae species. Chan et al. (2005) also used mercury chloride with two-stage surface sterilization using Clorox® solution for establishing the aseptic buds of Cymbopogon nardus.
The aseptic shoots of C. zedoaria and Z. zerumbet cultured on MS medium supplemented with different concentration of BA and IBA (2-10 mg L-1) produced roots and shoots simultaneously. The simultaneous production of roots and shoots were also reported in other Zingiberceae species (Balachandran et al., 1990; Chan and Thong, 2004; Bharalee et al., 2005). The shoots of C. zedoaria that were cultured on MS medium supplemented with 2-10 mg L-1 of BA and IBA induced the formation of 2-3 shoots per explant (Table 1) with the formation of stunted roots with callus formation at the base of the shoots. However, the buds of Z. zerumbet cultured on MS medium supplemented with higher concentration of BA and IBA (6-10 mg L-1) formed 4-5 shoots per explant and also showed abnormalities like C. zedoaria. The shoots of Z. zerumbet cultured on MS+6 mg L-1 BA induced the most multiple shoots formation (5.3 shoots per explant) (Table 2). This result indicated that high concentration of plant growth regulators was not suitable for the in vitro culture of Zingiberaceae species. Balachandran et al. (1990) also reported that higher concentration of Kinetin was not suitable for Zingiber officinale.
When the aseptic shoots of C. zedoaria were cultured on MS medium plus a lowest concentration of BA (0.5 mg L-1) and IBA (0.5 mg L-1) an average of 2.3 shoots were produced from each explant (Table 3). Since there was no increase in the number of shoots formed and also since it would be economically feasible, low concentration of BA and IBA (0.5 mg L-1) supplemented into the MS medium was chosen as the shoot multiplication medium for C. zedoaria. The aseptic shoots of Z. zerumbet cultured on MS medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA produced 3.9 shoots per explant. Since there was no significant difference in shoot number formation as compared to MS medium plus 6.0 mg L-1 BA (Table 4), hence MS plus 0.5 mg L-1 BA and 0.5 mg L-1 IBA was also chosen as a shoot proliferation medium for Z. zerumbet. Other researchers were using higher concentration of plant growth regulators for the induction of multiple shoots formation from some of the Zingiberaceae species. Nalawade et al. (2003) used higher concentration of BA (MS+4 mg L-1) to induce the same number of shoots formation as ours from the adventitious buds of Z. zerumbet within 6 weeks. Loc et al. (2005) reported that MS medium supplemented with 20% (v/v) coconut water, 3 mg L-1 BA and 0.5 mg L-1 IBA could induce the formation of 5.6 shoots per explant for C. zedoaria. Bharalee et al. (2005) found that MS medium supplemented with 4 mg L-1 BAP and 1.5 mg L-1 NAA was the best medium for shoot multiplication of C. caesia (3.5 shoots per explant) and MS plus 1 mg L-1 BAP + 0.5 mg L-1 NAA for C. zedoaria (4.5 shoots per explant). Balachandran et al. (1999) reported that C. domestica could produce 3.4 shoots per explant, C. caesia produced 2.8 shoots per explant and C. aeruginosa produced 2.7 shoots per explant using MS medium supplemented with 3 mg L-1 BAP. Nayak (2000) reported MS medium supplemented with 5 mg L-1 BAP was most effective for shoot multiplication of C. aromatica producing an average of 3.3 shoots per explant. Our results indicated that BA and IBA as low as 0.5 mg L-1 supplemented into the MS medium was sufficient for the induction of multiple shoots from the buds and shoots of C. zedoaria and Z. zerumbet.
The number of shoots produced from C. zedoaria and Z. zerumbet shoot explants cultured on the proliferation medium, MS medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA, was found to be not significantly different after 4 weeks or 8 weeks of culturing period. Thus the in vitro plantlets could be sub cultured every 8 weeks in terms of conservation purpose. We also observed that the in vitro plantlets could be maintained on solid basic MS medium for 5 months without subculturing with the plants still remained healthy and hence MS medium without the addition of plant growth regulator could be used for the in vitro germplasm conservation of C. zedoaria and Z. zerumbet.
| Table 1: | Effect of MS medium supplemented with BA and IBA (0-10 mg L-1) on multiple shoots formation (mean±SD) of Curcuma zedoaria after 4 weeks of culture |
 | |
| Mean values followed by the same alphabet are not significantly different (Tukey’s HSD test, p = 0.05) | |
| Table 2: | Effect of MS medium supplemented with BA and IBA (0-10 mg L-1) on multiple shoots formation (mean±SD) of Zingiber zerumbet over a period of 4 weeks |
 | |
| Mean values followed by the same alphabet are not significantly different (Tukey’s HSD test, p = 0.05) | |
| Table 3: | Effect of MS medium supplemented with BA (0-3 mg L-1) and IBA (0-3 mg L-1) on multiple shoots formation of Curcuma zedoaria over a period of 4 weeks of culture |
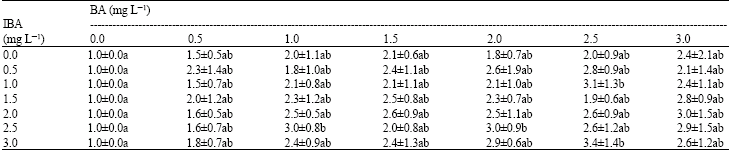 | |
| Mean values followed by the same alphabet are not significantly different (Tukey’s HSD test, p = 0.05). | |
| Table 4: | Effect of BA and IBA supplemented into MS medium on shoot multiplication of Z. zerumbet |
The plantlets of both the species that were sub cultured in proliferation medium every 4 weeks showed decrease in multiple shoot formation in each subculture cycle. Similar observation was observed by Thong (2001) for other Zingiberaceae species.
The aseptic shoot explants of C. zedoaria and Z. zerumbet cultured in liquid MS medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA produced significantly higher number of multiple shoots than those cultured on the solid medium with the same formulation. C. zedoaria formed 6.1 shoots per explant in liquid medium and 2.4 shoots per explant on solid medium. While Z. zerumbet produced 6.4 shoots per explant in liquid medium and 3.3 shoots per explant on solid medium (Fig. 1). Nalawaade et al. (2003) also reported that shoot multiplication rate of Z. zerumbet in liquid medium was higher than the semi solid medium. Improved rate of shoot multiplication in liquid medium had also been reported in other plant species such as the Curcuma species (Chan and Thong, 2004), Artemisia species (Liu et al., 2004) and Centaurium species (Piatczak et al., 2004).
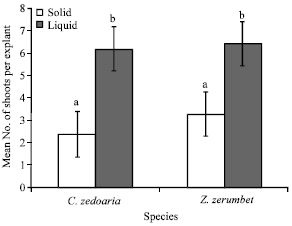 | |
| Fig. 1: | Effect of liquid and solid medium on shoot proliferation of Z. zerumbet and C. zedoaria. Mean number of shoots of the same species followed by different letter(s) are significantly different (t-test, p = 0.05) |
Piatszak et al. (2004) explained that the fast proliferation of shoots in liquid medium was due to the tissues were completely submerged in the liquid medium presenting a large surface area for the uptake of nutrients and plant growth regulators. However, we observed some of the plantlets of both C. zedoaria and Z. zerumbet became hyperhydric when cultured in the liquid proliferation medium. But the hyperhydricity of the plants disappeared after acclimatization and transferred to the soil.
By dividing the shoots of C. zedaoria into halves could induce more multiple shoots formation with an average of 5.9 shoots per half shoot after cultured for 4 weeks in the proliferation medium and therefore approximately 12 shoots could be produced from a single shoot by longitudinally dividing it.
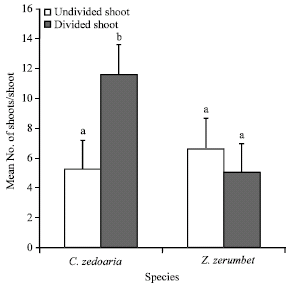 | |
| Fig. 2: | Effect of divided and undivided explant on shoot proliferation of C. zedoaria and Z. zerumbet mean No. of shoots of the same species followed by different letter(s) are significantly different (t-test, p = 0.05) |
 | |
| Fig. 3: | Half shoot of C. zedoaria forming multiple shoots in liquid MS medium supplemented with 0.5 mg L-1 BA and 0.5 mg L-1 IBA after 4 weeks of culture |
While each undivided shoot could produce only 5.3 shoots within the same duration. But for Z. zerumbet, divided shoots did not show an increase in the number of multiple shoots formed. Undivided shoots of Z. zerumbet produced 6.6 shoots per explant while the divided shoots produced only an average of 2.5 shoots per explant that was equivalent to 5 shoots which could be obtained from a single shoot by longitudinal division (Fig. 2). This result showed that plants of the same family had different in vitro growth response.
 | |
| Fig. 4: | Acclimatized plantlets (90 days old) of C. zedoaria |
The plantlets with multiple shoots derived form the half-shoot explants remained normal and healthy as shown in C. zedaoria culture (Fig. 3).
In vitro plantlets of C. zedoaria and Z. zerumbet with well developed roots were successfully acclimatized with 100% of the plantlets survived when transplanted to the outside environment. In vitro plantlets were morphologically similar to their respective mother plants. The acclimatized C. zedoaria plants developed their characteristic purple colouration in the midrib of the leaf (Fig. 4). The C. zedoaria and Z. zerumbet plants showed uniform micro rhizome formation after 90 days transplanting to the soil.
The authors would like to thank University Sains Malaysia for the research facilities.