Research Article
Production of Carotenoids by a Newly Isolated Marine Micrococcus sp.
Department of Chemistry, University of Prince Edward Island, 550 University Avenue, C1A4P3, PEI, Canada
Carotenoids are yellow to red colored pigments which originate from the terpenoid biosynthetic pathway. They are synthesized by plants, algae and by some fungi and bacteria. They are involved in photosynthesis as accessory pigments, functioning as antioxidants, light protection pigments and membrane stabilizers. Over 600 different carotenoids are known at present (Krubasik et al., 2001). Most consist of 40 carbon atoms. The enzymatic reactions leading to their formation are well understood in plants (Sandmann, 2001) and bacteria (Armstrong, 1997). The characterization of the enzymes catalyzing their formation was facilitated after the corresponding genes had been cloned and heterologously expressed. Several groups of Gram-positive bacteria, including species from the genera Micrococcus, Corynebacterium and Flavobacterium, synthesize cyclic and acyclic C45 and C50 carotenoids (Krubasik et al., 2001). The branchpoint from the typical C40 pathway to the elongated carotenoids is lycopene, but very little is known about the details of the subsequent reactions. The C50 carotenoid decaprenoxanthin, a cyclic carotenoid with two substituted ε-rings, was first identified in Flavobacterium dehydrogenans (Miki et al., 1994). Later it was found in a few other bacteria including Corynebacterium glutamicum (Krubasik et al., 2001). The most important biological function of carotenoids is as antioxidants owing to their potential to inactivate singlet oxygen and to quench carboxy radicals (Britton, 1995; Van den Berg et al., 2000). More and more evidences are being accumulated to show that carotenoids play an important role in human health. A number of epidemiological studies have revealed that an increased consumption of a diet rich in carotenoids is correlated with a diminished risk for various types of cancer, cardiovascular or ophthalmological diseases (Mayne, 1996; Sandmann, 2001). Carotenoids with unsubstituted β-ionone end groups are precursors of vitamin A. Vitamin A may inhibit tumor promotion, carotenoids are also protective because of their antioxidant effects to prevent cells and tissues from oxidative damage (Stahl and Sies, 2003). Carotenoids also influence cellular signaling and may trigger redox-sensitive regulatory pathways (Stahl et al., 2002). Their value in medical and pharmaceutics has triggered an increased interest in the synthesis of new carotenoid structures and the economic production of compounds in engineered cells. The goals of the present research are to isolate carotenoids producing bacteria from marine source, structure elucidation and optimization of the culture conditions for carotenoids production.
Isolation and enumeration of coral-associated bacteria: Sediment samples were collected in sterile bag under water by SCUBA diving in 2006 from different sites in Bimini, Russel Cay, Eleuthera (south end), Sweetings Cay, Cross Harbour Sandi point and Peterson Cay around The Bahamas. For bacterial isolation, the different sediment samples were suspended in sterile seawater (1 g/100 mL) and stirred for one hour Ten-fold serial dilutions of the suspension were prepared to a dilution of 10-4 and 100 μL of each dilution spread-plated in triplicate on nutrient rich agar media, Marine Agar 2216 (Difco Laboratories, Detroit) and Nutrient Agar in seawater (Difco) and nutrient poor media, 1/10 strength Marine agar and 1/10 strength Nutrient Agar in seawater (Difco), Plates were incubated at 30 °C for 3-4 weeks. Representatives of each colony morphotype were serially streak-plated onto fresh marine agar media to obtain pure cultures. All pure isolates were grown in Marine broth media overnight and stored in 20% glycerol at -80 °C.
Identification of the carotenoid producing strains by 16S rDNA sequencing: The bacterial strains were identified by 16S rRNA gene sequence analysis. The bacterial isolates were grown overnight in 2mL Marine Broth. Total DNA was extracted with the UltraClean Microbial DNA Isolation Kit (Mo Bio Laboratories Inc., Carlsbad, CA) according to the manufacturer's procedure. Eubacterial-specific primers, forward primer 16F27 (5´- AGA GTT TGA TCC TGG CTC AG-3´) and reverse primer 16R1525 (5´- AAG GAG GTG ATC CAG CCG CA-3´) derived from E. coli 16S-rDNA sequence (Lane, 1991) were used to amplify 16S rDNA gene. The reaction mixture of 50 μL contained at least 100 ng of genomic DNA (in 10 mM Tris-HCl, pH 8), 0.2 μM of each primer and PCR Supermix High fidelity (Taq and Go, Promega, CA). PCR fragments were purified using a QIAquick Gel Extraction Kit (Qiagen, Valencia, CA) and sequenced on an ABI 377 automated sequencer using the PRISM Ready Reaction Kit (Applied BioSystems, Foster City, CA). Sequence data were analyzed by comparison with 16S rRNA genes in the GenBank database. The nearest relatives of each organism were obtained by BLAST searches (Altschul et al., 1997).
Fermentation conditions and optimization of carotenoid production: The microorganism was grown on sterilized marine broth medium (5 mL) and incubated overnight at 30 °C and 100 rpm. One milliliter of the inoculum (2%) was transferred to 250 Erlenmeyer flask containing 50 mL marine broth medium and incubated at 30 °C. For the optimization of the carotenoid production, several nutritional and incubation conditions were studied including medium initial pH, incubation temperature, agitation rate, carbon source, poor and rich media and effect of some additives.
Analytical methods: The carotenoid was analysed by a modification of the method reported by Aksu and Eren (2007). Ten milliliters sample was taken from each flask at definite time intervals. Great care was taken to protect the samples from light. For the measurement of bacterial growth, dry biomass concentration was determined by measuring turbidity of the diluted samples at 600 nm using a standard curve of absorbance against dry cell mass weight. Absorbance measurements were carried out by using a Nanodrop ND-1000 spectrophotometer. The medium without cell growth was used as the blank. The total carotenoids content was also determined spectrophotometrically. For this purpose the sample was centrifuged to remove supernatant and the cell pellet was washed three times with deionized water. Cells were resuspended in methanol and blended to prevent clotting. Samples were then wrapped with aluminum foil to protect them from light and samples were vortexed until the methanol layer turned yellow (within 15 min). The methanol extract was purified from cell debris by further centrifugation at 8,000 g for 10 min, extraction were done several time until no yellow colour was extracted from the cells. Methanol solution was collected and absorbance was measured at 440 nm.
HPLC analysis: Methanol extract was evaporated to dryness under vacuum at 30 °C in a Buchi rotavapor and analysis of carotenoids was carried out without saponification. Dry pigments were dissolved in 1 mL methanol and injected (50 μL) onto a μBondapak C18 column (300x4.6 mm, 5 μm particle size, RCM type; Water, USA). All other HPLC apparatus (Series 200 pump, 785A UV/Vis detector at 440 nm) was from Perkin Elmer. Separation was achieved using reverse phase HPLC at a flow rate of 1.0 mL min-1. Solvents and conditions for separation were as follows: 0 to 10 min, 90% methanol: H2O; 10-20 min, 90% methanol: H2O to 100% methanol; 20-40 min, 100% methanol (Sander et al., 1994).
LC-MS: For analysis of carotenoids by HPLC-APCI-MS, an Accela instrument (Thermo Electron Corporation) equipped with automatic sample injector, photodiode-array detector (PDA) and Finnigan LXQ series APCI mass selectivity detector was used. The mass spectra were recorded in the positive ion mode in the mass range from m/z 50 to 1500. The voltage of the corona needle was +3.9 kV, cone voltage of +17 V, probe temperature of 300 °C and source temperature of 200 °C. Separation was performed on 50x2.1 mm x1.9 um Hypersil Gold C18 column using the gradient solvent system as follows: 0-1 min, 70% methanol: H2O; 1-10 min, 100 % MeOH; 10-15 min, 100 % MeOH. The flow rate was set to 0.4 mL min-1 and injection volume was 1 μL. The PDA was operated at 200-800 nm.
RESULTS AND DISCUSSION
In the present study, a total of 75 bacterial strains were isolated from different sediment samples. Total bacterial count ranged from a highest of 3x105 to a lowest of 3x107CFU per gram of sediment. Of the 75 isolates, 4 strains were found to be carotenoid producers, termed (PAH81, PHAH82, PAH83 and PAH84). The isolates were identified by complete 16S rRNA gene sequence. Analysis of the 16S rDNA sequence of strain PAH81 showed 99% similarity with Halomonas sp., PAH82 (98%) similarity with Bacillus aquimaris. PAH83 showed highest similarity with Micrococcus sp. (98%), PAH84 showed 99% with Micrococcus luteus. Strain PAH83 was selected for further studies. The physiological and biochemical features of strain PAH83 is shown in Table 1.
Identification of the major carotenoids produced by isolate PAH83: Carotenoids of the cells of the isolated Micrococcus sp. PAH83 were extracted and analyzed by HPLC (Fig. 1A). The elution profile indicated the presence of three major carotenoid peaks, with the same absorption spectra. LC-MS analysis of the major peaks indicated their relative molecular mass (m/z) to be 1028, 866 and 706 with the absorption maxima of 418, 440 and 470 nm (Fig. 1B). This data with proton NMR (data not shown) indicted that the structure of the major carotenoids to be decaprenoxanthin diglucoside, decaproxanthin monoglucosides and decaprothannthin, respectively. The deccaprenoxanthin biosynthesis pathway of by Micrococcus luteus has been identified by Sandmann (2001).
Production and optimization of carotenoids production by isolate PAH83: An experiment on the growth pattern and the time course of carotenoid production by Micrococcus sp. using marine broth was carried out. The carotenoids production was increased during cultivation and reached maximum level at the end of the stationary phase (30 h). The effects of aeration, initial pH, temperature, initial sugar and incubation period on the growth and carotenoids biosynthesis of the marine Micrococcus sp. were investigated.
| Table 1: | Physiological and biochemical characteristics of the marine isolate PAH83 |
 | |
Effect of initial aeration on the growth and production of carotenoids: If the microorganism requires oxygen, aerating the growth medium is very important for the successful progress of the fermentation. As Micrococcus sp. is an aerobic microorganism, the effect of the aeration rate on the growth and total carotenoids formation of the bacterium was examined and the results obtained were shown in Table 2. Both the growth and total carotenoids formation changed significantly with increasing the aeration rate from 0-300 rpm. Maximum carotenoids production rate (188.31 mg g L-1) was shown
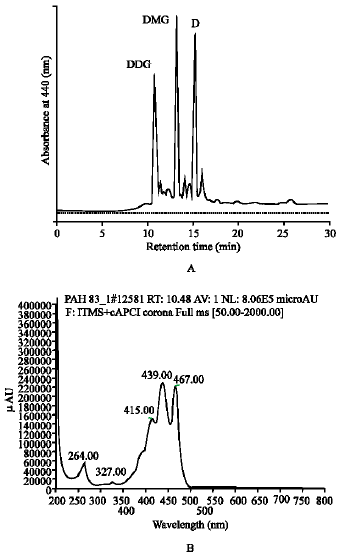 |
| Fig. 1: | A: Separation of carotenoid of Micrococcus sp. PHA83 on μBondapak C18 column. DDG: Decaprenoxanthin diglucoside, DMG: Decapromanthin monoglucosides and D: Decaprothannthin. B: Absorption spectra of DDG, DMG and D with maxima at 418, 440 and 470 nm |
| Table 2: | Effect of aeration (rpm) on the cells dry weight, total carotenoid and carotenoid production rate by the marine Micrococcus sp. |
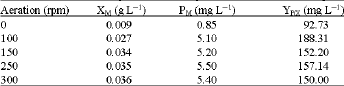 | |
| XM: cell dry weight (g L-1); PM: Total carotenoid; YP/X: Carotenoid production rate Standard deviation is 2-4% | |
to be at rpm of 100, which was two fold more that obtained in anaerated culture (90.73 mg g-1). The maximum dry cell biomass was shown at 250 rpm. These results are consistent with that reported by Aksu and Eren (2007) where the carotenoid production by Rhodotorula glutinis was increased by increasing the aeration level. Aeration
| Table 3: | Effect of initial pH on the cells dry weight, total carotenoid and carotenoid production rate by the marine Micrococcus sp. |
 | |
| XM: cell dry weight (g L-1); PM: Total carotenoid; YP/X: Carotenoid production rate. Standard deviation is 4-6% | |
| Table 4: | Effect of incubation temperature on the cells dry weight, total carotenoid and carotenoid production rate by the marine Micrococcus sp. |
 | |
| XM: cell dry weight (g L-1); PM: Total carotenoid. YP/X: Carotenoid production rate. Standard deviation is 2-6% | |
could be beneficial to the growth and performance of microbial cells by improving the mass transfer characteristics with respect to substrate, product and oxygen (Davoli et al., 2004).
Effect of initial pH on the growth and production of carotenoid: The pH value of growth medium affects not only biosynthesis activity of culture, but also culture growth rate. Table 3 shows that with ranging from 3-8. With raising the pH, carotenoid production rates and growth increased and reached to a maximum level at pH 6 and 8, respectively. On the other hand, a further increase of the pH resulted in a reduction of both rates. Although the optimal pH of 6 yielded the highest values of total carotenoids concentration and product yields, maximum biomass concentration was obtained at an initial pH value of 8. It has been reported that the optimal pH value of carotenoid production was different of that of growth in carotenoids production by Rhodotorula glutinis (Aksu and Eren, 2007).
Effect of temperature on the growth and production of carotenoid: Temperature is another important parameter affecting the performance of cells and product formation. The temperature of the growth medium had also a considerable effect on both the growth and carotenoid production of the marine Micrococcus sp. the cell biomass increased notably with raising the temperature with up to 37 °C and lessened sharply at 45 °C due to the denaturation of the enzyme system of microorganism at higher temperatures (Table 4). The total carotenoids production rate was also affected by the variation of fermentation temperature; enhanced strictly up to 30 °C (220.77 mg g L-1). Maximum cell biomass where found to be at 37 °C (0.31 mg L-1).
Effect of different media on the growth and production of carotenoids: The effect of nutrient rich (Marine broth and Nutrient broth media) and nutrient poor media (1/10 strength marine broth and 1/10 strength Nutrient broth) was investigated. The results presented in Table 5 showed that nutrient poor media showed less cell biomass but more carotenoids production rate, by the marine Micrococcus sp., than the nutrient rich media with diluted marine broth as the best one. It was found that using diluted marine broth medium resulted in increase of the total carotenoids production rate by about 1.5 fold more than marine broth medium, 331.58 and 220.77 mg g L-1, respectively. However maximum cell biomass was found to be in culture using marine broth medium.
Effect of different sugars on the growth and production of carotenoid: The effect of addition different sugars to the diluted Marine broth media, triose, tetrose, pentose, monosaccharide, disaccharide and polysaccharides, was investigated. Glucose was found to enhance the cell
| Table 5: | Effect of different media on the cells dry weight, total carotenoid and carotenoid production rate by the marine Micrococcus sp. |
 | |
| MB: Marine broth media, MB ½: half strength Marine broth media, MB 1/10: 1/10 strength Marine broth media NB: Nutrient broth media, NB ½: half strength Nutrient broth media, NB1/10: 1/10 strength Nutrient broth media. XM: cell dry weight (g L-1); PM: Total carotenoid; YP/X: Carotenoid production rate. Standard deviation is 3-6% | |
| Table 6: | Effect of different sugars on the cells dry weight and carotenoid production rate by the marine Micrococcus sp. |
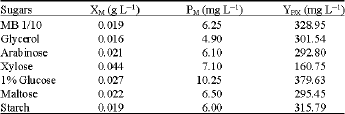 | |
| XM: cell dry weight (g L-1); PM: Total carotenoid; YP/X: Carotenoid production. Standard deviation is 2-6% | |
| Table 7: | Effect of different glucose concentrations on the cells dry weight and carotenoid production rate by the marine Micrococcus sp. |
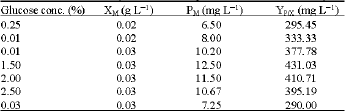 | |
| XM: cell dry weight (g L-1); PM: Total carotenoid; YP/X: Carotenoid production. Standard deviation is 2-4% | |
biomass, total carotenoids and carotenoids production rate (Table 6). It was found that the addition of xylose to the diluted marine broth medium reduce the carotenoid cell productivity to about 50% of the control. Addition of 1% glucose increased the total carotenoids slightly. The effect of different glucose concentration on the growth and carotenoid production was investigated (Table 7). Both cell biomass and carotenoids production was increased by increasing the glucose concentration up to 1.5% with cell biomass and carotenoid production rate of 0.029 g L-1 and 430 mg g L-1, respectively.
In summary, the carotenoids production rate by the marine Micrococcus sp. PAH83 was increased about five fold by varying the nutritional and incubation conditions 92.73 and 430 mg g L-1, respectively.
Marit H Stafsnes Reply
What kind of unit is mg g L-1? It should represent mg carotenoid per litre divided by g cell mass per litre and thereby mg/g, or?
I would have liked to see the MS data for the three peaks. Decaprenoxanthin has molecular mass 704, not 706 (in the text it is written decaproxanthin)
Micrococcus luteus does not produce decaprenoxanthin as written in the text, but this does rather Corynebacterium glutamicum,. The species has been identified to Micrococcus sp, another name of C.glutamicum is Micrococcus glutamicus (Kinoshita et al. 1958). Could it be a C.glutamicum?
The chapter in the introduction starting with From "The most important biological functions..." and ending with "The value in medical and pharmaceuticals has triggered..." is a direct copy of a paper by Wang et al, 2007 Progress in molecular breeding and metabolic engineering of biosynthesis pathways of C30, C35, C40, C45, C50 carotenoids. In Biotechnology Advances 25 (2007) 211 - 222
Auther Reply
Hi Marit
Micrococcus luteus is known to produce decaprenoxanthin and it has been published with its former name as Sarcina lutea, I will provide you with the reference soon, but if you make good search, you will find it easily
Regarding the introduction sentences, I think the auther used reference and it is something normal
Regarding the unit and calculation, it is right just read it carefully
Thanks for your interest
The Auther
Marit Reply
Hi Auther. M. luteus is known to produce sarcinaxanthin (from the former name Sarcina lutea)and is an isomer of decaprenoxanthin. The paper referred to (Sandmann, 2001)which is claimed to write about the decaprenoxanthin biosynthesis pathway by M luteus does not mention Micrococcus nor decaprenoxanthin. I am looking forward to receive the references you mention. Thank you.
Masoud Hamidi Reply
Hi Dear Abdelnasser Salah Shebl Ibrahim,
Is it possibel to provide the formula was used to calculate total carotenoid content?
Best regards
M.Hamidi