Review Article
G-protein Coupled Receptors: A Potential Candidate for Drug Discovery
Department of Zoology, Mahatma Gandhi Central University MGCUB, Motihari 845401 (Bihar), India
LiveDNA: 91.15888
Surface plasmon resonance
Membrane protein interaction studies: Protein-protein interactions are fundamental to most of the physiological processes. Understanding the nature and mechanism of such interactions is crucial to biological research. Membrane proteins are the major molecular targets for most of the validated drugs and also the upcoming drugs. In this respect, development of surface chemistry enabling the immobilization of lipid bilayers and determination of protein-membrane interactions in real time has made Surface Plasmon Resonance (SPR) an invaluable tool. Surface analytical tools like SPR is a preferred method of choice for label free analysis for kinetics and affinity of bimolecular binding (protein-ligand) events in real time. It offers high detection sensitivity of bimolecular interactions compared to traditional biomedical methods. Most important step in SPR mediated study involves immobilization of reactants on the chip surface, which becomes a limitation for transmembrane proteins due to their susceptibility to the surroundings. The SPR spectroscopy is a powerful technique for the study of ligand binding interactions with membrane proteins. To achieve this, transmembrane proteins need to be incorporated in a structure where it can reside in its native like environment. Multilayered supported liposomes offer such environment and are used to immobilize transmembrane proteins onto the chip surface to facilitate SPR based detection. Recently, biomimetic membrane model systems have been developed to study protein-membrane interactions as well1.
With the introduction of new improvised instruments and modified sensor chip chemistries, SPR is finding new applications for molecular interaction studies on larger scale. Availability of kinetic and thermodynamic data of bimolecular interaction helps in elucidating macromolecular binding events and mechanism of molecular recognition. The existing optical biosensing platforms like SPR can be integrated with other assays thus ensuring portability and enhanced functional efficiency2. The SPR since its development is consistently improvised and modified for its widespread application that ranges from ligand screening, immunology, cell biology, signal transduction, nucleotide-nucleotide, nucleotide-protein, protein-protein and protein-lipid interactions3.
Further, advent of protocols to synthesize stable membrane like surfaces coupled with commercialization of sensor chips has facilitated the study of protein-membrane interactions via SPR. L1 sensor chip from Biacore is particularly important owing to its ability to capture intact liposomes or subcellular preparations. This allows analyzing the lipid specificity of protein-membrane interactions by manipulating the lipid composition of the immobilized membrane. This opens up the enormous future possibility for studies involving domains or proteins that participate in cell signalling, pore-forming proteins, membrane-interacting peptides, coagulation factors, enzymes, amyloidogenic proteins and prions, etc4-6.
This review analyzes the protocols and associated constraints related to the synthesis and study of membrane proteins like GPCRs whose interactions with specific ligands have been characterized already. It also describes the latest advances and improvisations in SPRs in relation to membrane protein-ligand interactions and future trajectory leading to the discovery of new drugs.
G-PROTEIN COUPLED RECEPTORS (GPCRs)
Biosensing based on plasmon resonance have opened a flood gate for the investigation of G-Protein Coupled Receptors (GPCRs) binding with its ligands. The GPCR superfamily constitutes one of the largest protein family in mammals with more than 800 coding sequences reported in the human genome1 (Fig. 1). Over 50 % of marketed drugs either activates or inhibits these proteins. Therefore, it becomes imperative to understand mechanism behind signal transduction and protein-ligand interactions as it will have direct bearing to the different aspects of health and disease. However, the information available for the same is grossly inadequate citing their perceived importance in human health and disease. This is partly due to the limitations in getting their high resolution structure, low abundance naturally coupled with difficulty in their purification in significant amount from tissue derived or recombinant protein derived vector and crystallization are some of the serious roadblocks in achieving their full potential for human benefits. This is where plasmon resonance based techniques, which are highly sensitive, non-invasive, require very little amount of protein becomes all the more important for studies related to the membrane protein-ligand interactions.
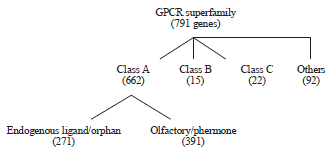 | |
| Fig. 1: | Schematics of GPCRs classification (Bjarnadottir et al.106) |
 | |
| Fig. 2: | Alpha helical structure of GPCR (Retrieved from open source of Wikipedia) |
Plasmon resonance based techniques also offer the advantage of test protein or analyte to be in lipid bilayers. This mimics the native membrane environment and provide the most ideal setup for the kinetics and equilibrium analysis of protein-ligand, protein-protein and protein-lipid interactions1.
PROTOCOLS: MEMBRANE PROTEIN INTERACTIONS
Membrane immobilization
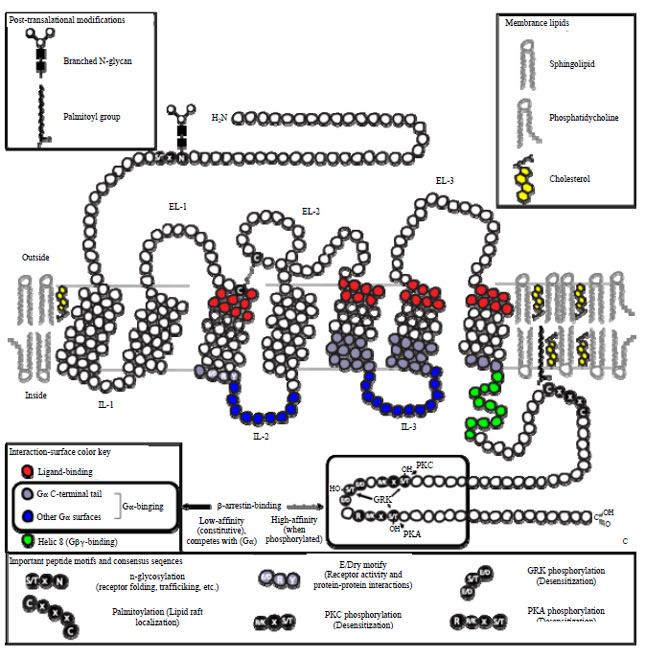
Biomimetic membranes constitution on the sensor surface: Knowledge about the nature and mechanism of interactions occurring with membrane bound receptors like ligand-gated ion channels, GPCRs, antibody receptors and cytokine receptors are important for drug discovery process (Fig. 2, 3). Among these, GPCRs are very crucial as majority of the drugs target them. To understand the mechanism of these interactions, these must be probed in vivo or in the facile biomimetic membrane system7,8. Currently available techniques rely on the soluble form of the receptor in solution phase for kinetic analysis of molecular recognition event, which is more suitable for receptors with single transmembrane domain. This is a big limitation as membrane receptors have substantial hydrophobic domain, different tertiary structures and binding affinities in solution. Other limitations include inability to analyze ligand induced signal cascades and complex membrane proteins. To circumvent these roadblocks, techniques were required that allow membrane-ligand interactions analysis in their near native membrane environment. Many biophysical techniques like patch clamping, magic-angle-spinning NMR, fluorescence-correlation spectroscopy, Fluorescence Resonance Energy Transfer (FRET) and analytical ultracentrifugation was applied to the study of binding to whole cells, membrane protoplasts and proteoliposomes. However, surface-related processes like interactions with membrane receptors are difficult to study with bulk techniques. Also, these techniques require high concentration of protein which is difficult to obtain. Therefore, the need of the hour is to devise a protocol that enables quantitative analysis of affinity and kinetics of interactions by recreating or immobilizing biomimetic membranes on the sensor surface.
 | |
| Fig. 3: | Schematic illustration depicting structural features important to the biological functions of GPCRs (Retrieved from open source of Wikipedia) |
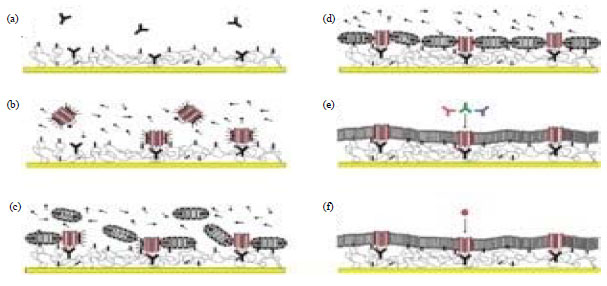
Supported lipid monolayers/bilayers: This simplest protocol for immobilization of membrane on the sensor surface involves adsorption of lipid onto a hydrophobic surface. This leads to the formation of supported monolayer with hydrophobic acyl chains contacting the hydrophobic surface and polar head groups of lipid facing the solution9. These hybrid bilayers formation involves modification of surface chemistry through physisorbed monolayer of fatty acid salts or a chemisorbed monolayer of alkyl chains linked to the support via functional groups or a hydrophobic hairy-rod polymer (Fig. 4, 5). However, this protocol is confined to the receptors attached to the outer leaflet of native membrane and to the cases where analyte binds to the lipid. The hybrid bilayer membranes composed of SAM and lipid monolayer provide excellent system to study the molecular interactions on the membrane surface. However, these cannot be use to insert transmembrane domain in active form. Reason could be inherent rigidity of layers having high proportion of hydrophobic and polar regions. Attachment to the solid surface leaves no space for water that is required for structure and function of membrane proteins. Another limitation is the inability to hold the transmembrane domain with significant cytosolic or extracellular domains. Vogel and group have pioneered the study analyzing the interaction of cholera toxin B subunit with the cell-surface ganglioside GM110 using optical biosensors10,11.
Surface of mammalian cell is studded with membrane bound receptor molecules. These naturally occurring receptors provides an insight into exploring the protocols and methodologies to transport polar drug candidates across membrane bilayers, a process of huge significance in medicinal chemistry12,13. In such scenario, artificial membrane receptors are composed of covalent attachment of the recognition motif to a lipid or steroid derivative that is incorporated in a synthetic membrane.
 | |
| Fig. 4: | Schematic of capture and reconstitution of GPCRs on a sensor chip surface. Reproduced with permission from Patching3 |
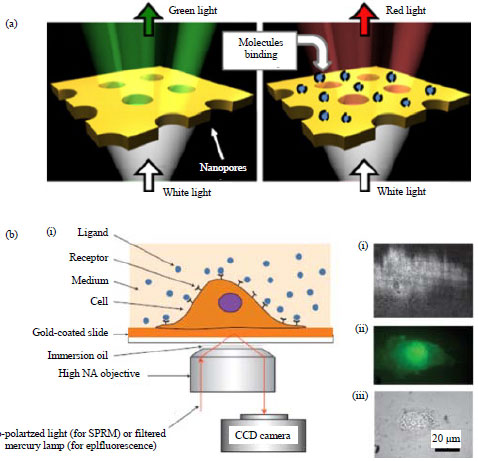 | |
| Fig. 5: | SPR setup for quantifying membrane protein-ligand binding (a) Nanopores in a gold film and (b) Surface plasmon resonance microscopy with intact living cells |
The recognition motif is displayed above the membrane surface to bind to the target14-16. Another alternative could be the provision of binding pocket inside the membrane layer, a strategy utilized by transmembrane pore forming peptide. However, synthetic pocket inside the membrane bilayer poses problem for natural system including poor solubility, non-specific localization in membrane bilayer or reduced affinity for target17-20.
Deep cavitands: Deep cavitands are mimetic proteins that provide a cavity for selective and specific recognition of target molecules based on similar charge distributions. Diffusion NMR and 1D NMR have been employed to study the host: Guest properties of cavitands. Results show the specific binding in the presence of lipids above the critical micelle concentration21-27. Prominent technique for such studies are confined to fluorescence microscopy but the advent of SPR, a label free, simple protocol to monitor the events on membrane in real time has greatly revolutionized such type of studies. Availability of calcinated nanoglassified gold surface has facilitated the direct assembly of supported membranes on the sensor surface enabling the relevant analysis and measurement thereof. Binding interactions of lectins and bacterial toxins were analyzed with calcinated gold chips28. However, measuring small molecules via SPR in supported membrane is plagued with lower sensitivity as low molecular weight compounds are small enough to generate quantifiable refractive index variations. Also, the recognition event might get displaced from the surface and affinity comes in millimolar range than micro- or nanomolar range29-32.
In a study by Liu et al.33, binding interactions of small molecules with cavitand incorporated in biomimetic supported lipid bilayer membrane were analyzed by SPR spectroscopy and fluorescence microscopy. Results show that cavitand has retained its host properties and show selectivity for guest/substrate (choline) and no competitive inclusion of substrate in membrane bilayer is observed with binding affinity in millimolar range. Therefore, membrane-cavitand-guest system can be used to sense proteins on the membrane surface.
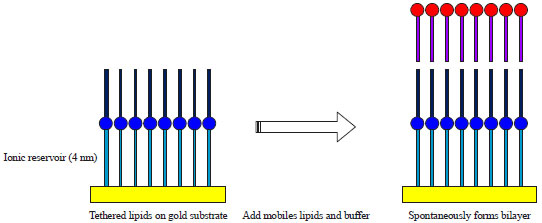 | |
| Fig. 6: | Schematic illustration of supported bilayers (Retrieved from open source of Wikipedia) |
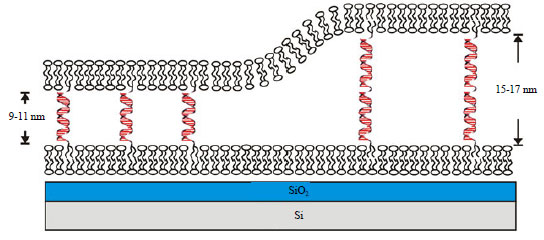 | |
| Fig. 7: | Schematic illustration of a DNA tethered lipid bilayers. Chung et al.109 (Retrieved from open source of Wikipedia) |
Lipid transfer proteins: Lipid transfer proteins plays important role in plasma where they are associated with lipoprotein differentiation and maturation and also lipid metabolism and membrane biosynthesis. The SPR can measure the binding interactions of unmodified natural lipids with lipid transfer protein and provide the quantitative data on kinetics and affinity of interactions. In a study, biotinylated sterol career protein, a lipid transfer protein was genetically engineered with C-terminal biotinylated tag for expression. This was done to facilitate binding with streptavidin coated SPR chip. Biotinylated sterol career protein was immobilized on the streptavidin SPR sensor chip. This was followed by kinetics and affinity measurements for the analytes oleic acid, linoleic acid, cholesterol and fluorophore (NBD)-derivatized cholesterol. This standard protocol can be applied to other transfer proteins as well after appropriate protein engineering34-38.
Proteoliposomes: The SPR coupled with proteoliposomes as affinity elements has provided a powerful and highly sensitive alternative for multiple analyte detection. One crucial issue in utilizing such platform is to maintain the functionality of the immobilized protein on the sensor surface that is determined by the appropriate choice of lipids and surfectants involved in the membrane reconstitution. In the previous study Olguin’s group has used TLR5-proteoliposomes as screening affinity elements of bacterial flagellin. In the later part of study they had altered the immobilization condition of TLR5 to assess the effect of fluidity of membrane and size of the liposomes for their use as affinity elements for novel biosensing applications. Here, this group has used preformed liposomes with lipid composition of POPC, POPC-DMPC and POPC-POPE and surfactants OG, Triton X100 and DDM. The kinetic and affinity analysis by SPR has revealed that fluidity and size of liposomes are important determinants for affinity measurements39.
Tethered lipid membranes: This method involves covalent attachment of membranes to solid support via lipid polar head groups or amphiphilic copolymers at some distance from the surface (Fig. 6, 7). Lang et al.40 have developed tethered lipid membranes on the gold surface utilizing a thiolipid with a hydrophilic oligoethoxy spacer between the lipid head group and the anchoring sulphur groups. Bilayers were formed from the thiolipid monolayers through the self assembly of phospholipids using lipid vesicles or mixed micelle dilution with the advantages of robustness and longevity. Besides, spacer acts as elastic buffer decoupling the lipid layer from the solid support. Hydrophilicity of spacer allows the interface of water film between spacer and solid support essential for maintaining the functionality of GPCR inserted into these layers. These tethered bilayers were studied by SPR, Surface Plasmon Fluorescence Spectroscopy (SPFS), resonant-mirror, waveguide techniques and electrical measurements for the conducting surface like metals, Indium-tin oxide and conducting polymers. This system was utilized for the reconstitution of membrane proteins like rhodopsin to study receptor and transducin interaction41.
Naumann et al.42, have used peptide spacer for bilayer deposition and incorporation of F0F1-ATPases. Cornell et al.43 have included membrane-spanning thiophospholipid to increase stability and analyze channel conductance. Similarly, SPR was utilized to monitor the incorporation of Torpedo californica acetylcholine receptor into a thiopeptide supported lipid bilayer by fusion of receptor proteoliposomes with a gold-supported thiopeptide lipid monolayer. Williams et al.44 introduced one step process of bilayer formation as well as anchoring by spreading phospholipid vesicles on gold surfaces having immobilized cholesterol derivatives in variable densities44. This involved incubating the surface with mixed lipid vesicles consisting of fraction of lipids with surface-active head groups. This was reported for mixed thiolipid/phospholipid vesicles on bare gold and mixed vesicles from succinimidyl-conjugated dialkylamine and phospholipid on cysteinamine-covered gold. Recently, His-tagged lipids as anchor lipids were introduced for reversible coupling to metal-chelating surfaces45.
Self assembled solid-supported lipid membrane: This system uses a teflon spacer to support bilayers over a prism surface, of a SPR/PWR sensor, where GPCRs are inserted and signaling is investigated46. The generation of a self-assembled solid-supported lipid membrane follows Montal-Mueller-Rudin protocol47. This protocol involves spreading a lipid bilayer across Teflon orifice separating the silica waveguide surface from the aqueous phase in the PWR cell. The hydrophilic SiO2 surface with thin water layer47 attracts the polar groups of the lipid molecules with the hydrocarbon chains oriented towards the bulk lipid phase leading to the orientation of lipid molecules. Addition of buffer into the sample compartment of PWR cell induces the formation of plateau-Gibbs border that anchors the membrane to the Teflon spacer allowing membrane flexibility.
The SPR and PWR can be used to monitor the conformational changes of GPCRs such as rhodopsin48,49, the Human Delta Opioid Receptor (hDOR)48-52 and the β-adrenergic receptor in real time fashion53 followed by insertion into the bilayer and ligand binding. SPR and PWR were also utilized to monitor the binding interactions of phosphatidylserine (PS) synthase52,53 and penetratin54 with lipid membranes.
Tanvir et al.55 have done the characterization of thin films on solid supports like gold mica, macroporous aluminium oxide using SPR, Atomic Force Microscopy (AFM) and Environmental Scanning Electron Microscopy (ESEM). Commercially available macroporous aluminium oxide membranes with a high surface area permits the retention of a high amount of microsomal membranes. Functionality of microsomal film was tested by analyzing the activities of phases I and II enzymes. Microsomals are reusable for the same or different substrate after washing with appropriate buffer55.
CELL SIGNALLING
Signal transduction studies via SPR: Rhodopsin (a GPCR) is widely used in plasmon resonance studies owing to its biophysically and chemically well characterized signal cascade and availability of purified components in large amount. Tollin’s group56 have initiated the SPR based investigation of rhodopsin. These studies involved octyglucoside-solubilized receptor incorporated into a solid-supported lipid bilayer. Variation in SPR signal was found to correlate with the variation in pH leading to the formation of light induced metarhodopsin II. Transducin binding to rhodopsin was monitored and results show tight binding in positively cooperative manner57,58.
Solid-supported lipid bilayers to study GPCRs signal transduction pathways: Solid-supported lipid bilayers constitute a good model system for surface plasmon studies of GPCRs signal transduction pathways. Such model system provides a fluid environment for inward and outward movement of lipids. The lipid membrane systems are simple to prepare and remain stable. This technique can be extended to the lipid-protein interaction studies. Protein can be inserted into the bilayer via N or C terminus and do not require chemical modification. Bilayer can be easily removed from the surface and surface can be reused many times followed by washing with appropriate detergent48,49. However, the main disadvantage with the system is the possibility of denaturation of membrane protein thus cessation of mobility due to paucity of space and surface irregularity of the silica surface.
Rhodopsin (GPCR):
A case study: Heyse and group have devised novel strategy for the immobilization of rhodopsin, a GPCR. Here, bilayer of lipid on the gold sensor surface separated by phospholipid monolayer provides a suitable matrix to reconstitute a transmembrane protein in functionally active form. This results in rhodopsin distribution in phospholipid bilayer while transducin (a G protein) confined to both mono and bilayer. Binding interactions of rhodopsin with transducin resembles with those of native system indicating restoration of functionality of rhodopsin in supported membranes. Diffusion of transducin follows a gradient from monolayer to bilayer resulting in displacement of mass and protein monitored by one dimensional SPR imaging (SPRi). This methodology could be applied to other GPCRs as well.
In another study, rhodopsin was immobilized on a biotinylated sensor surface covered with SAM. This resulted in a uniform orientation with the intracellular domain pointing away from the surface thus, optimizing the interaction with G protein. Micropattern of the receptor was created using microcontact printing8,40,59 to pattern the SAM with respect to the biotinylated thiols resulting in micropattern of streptavidin. After immobilization of the receptor and washing with detergent, a Supported Lipid Bilayer (SLB)8 was formed by micellar dilution40 followed by addition of G-protein. The activity of the receptor was detected through SPR signal, observed upon addition of GTP to the proteolipid film. This was consistent with the dissociation of the G-protein followed by a reassociation signal with G-protein relaxing to its resting state by hydrolysis of the bound GTP. In these studies the capability of rhodopsin to bind its ligand, retinal was also investigated in patterned (receptor free zones for enhanced sensitivity), oriented and functional manner that facilitates surface-sensitive detection. Micropatterning was done utilizing microc,40. The sensor chip was composed of SAM having biotinylated thiols and hydroxy-undecanethiol (HTA) for streptavidin binding8,40,59. Receptor immobilization was followed by washing with detergent and supportedontact printing to pattern the SAM related to biotinylated thiols. The glycosylation site of rhodopsin was utilized for biotinylation. This confines the biotin tag to extracellular receptors resulting in the uniform orientation8 lipid bilayer formation induced by micellar dilution8 and finally the addition of G protein8,40. The SPR was employed to analyze the binding of receptor after addition of GTP to the proteolipid film. This was consistent with the disassociation of G protein followed by reassociation with G protein in its resting state on hydrolysis of GTP. The SPR was also utilized to analyze the binding of rhodopsin with retinal, which involved conversion of rhodopsin to opsin by repeated flashes followed by the addition of 11-cis- or 9-cis-retinal for binding reaction. The reconstituted receptor G protein show good binding activity and stability thus validating the utility of the system8,40.
GPCR immobilization on CM5 sensor chip: Karlsson and Lofas60 provided a simple protocol for immobilization and reconstitution of GPCRs on CM5 sensor chip surface. Detergent soluble receptor was coupled to the chip surface via amine coupling by the injection of lipid/detergent-protein micelles over the chip surface indicating an optimal ratio of lipid to detergent for optimal protein capture. This was followed by the removal of detergent through flow of detergent free water. This methodology is simple and rapid and suitable for rapid screen or array assays. The only disadvantage is the formation of mixed population of receptors due to amine coupling. Stenlund et al.61 have successfully immobilized chemokine receptors CXR4 and CCR5 with a C-terminal C9 tag on CM5 chip modified with hydrophobic alkane groups and the monoclonal antibody ID4 (binds specifically C9 tag). Detergent solublized receptors were immobilized on the chip surface followed by reconstitution in lipid environment. Binding with conformational dependent antibodies and binding with chemokine revealed the native conformation of the receptor.
SPR spectroscopy to monitor receptor assembly: The SPR spectroscopy was used to monitor the fusion of nicotinic acetyl-choline receptor from the electric organ of Torpedo californica incorporated into a thiopeptide supported lipid bilayer invloving fusion of liposomes with reconstituted receptor onto a gold-supported thiopeptide lipid monolayer62. SPR was also utilized to monitor the binding with the antagonist α-bungarotoxin. Surface plasmon fluorescence spectroscopy was employed to monitor the binding of the monoclonal and fluorescent labeled polyclonal antibodies. These binding assays reveal successful incorporation of receptor in the lipid bilayer62.
Yildiz et al.63 have described the protocol for superficial assembly of biomimetic membranes for expression and insertion of membrane proteins. This was achieved in a 3-step protocol, i.e., printing of alpha-laminin peptide on gold; covalent coupling of lipid anchors to the peptide and spread of lipid vesicle to form a complete bilayer. This system was characterized by Contact Angle (CA) goniometry, SPR spectroscopy and SPRi. Functionality of proposed system was analyzed by incorporation of Cytochrome bo(3) ubiquinol oxidase (Cyt-bo(3)) into biomimetic membrane by in vitro expression technique monitored by Surface Plasmon Enhanced Fluorescence Spectroscopy (SPFS). Such system provides an attractive alternative for membrane protein insertion and characterization supported by planar membranes tethered by α-laminin peptide cushion layer63.
Intact cell based SPR: Intact cell based SPR provides a powerful tool for the discovery of drugs against membrane receptors on pathological cells. This involves a simple chip chemistry having epidermal carcinoma as an analyte against Epidermal Growth Factor (EGF) immobilized on the sensor chip generating a SPR signal. Analyte (epidermal carcinoma) with suspension of EGF has competitively reduced the SPR signals indicating specific interactions of immobilized EGF with the extracellular region of its receptor, which is highly expressed on the surface of the A431 cells64.
EEM+PCA coupled interaction analysis: A combine of fluorescence-based Excitation-Emission Matrix (EEM) and Principal Component Analysis (PCA) was utilized to study the interaction between natural colloidal/particulate and proteins. The SPR was used to validate the approach based on fibre optic based surface fluorescence measurements. This has the potential of elucidating variety of physiochemical phenomena like membrane fouling, adsorption of bacteria onto surfaces and various applications of nanoparticles in nanomedicine and nanotoxicology65.
Receptor-mediated signaling recruits activated intracellular signalling molecules onto Receptor-Induced Signaling Complexes (RISC) at the cytoplasmic surface of the cell membrane. This process is governed by protein-protein, protein-lipid interactions and protein acylation66. Mozsolits et al.67 have described the use of SPR for such studies.
Supported Bilayer Membranes (SBMs)
A cell mimics system: Supported Bilayer Membranes (SBMs) formed on glass substrates provides an ideal cell mimicking model system useful for biosensing applications. Stability of such system against the damage by air or chemical is crucial in determining its biosensing application. Han et al.68 have analyzed the stability criteria of seven SBMs against air or chemical damage (surfactants, organic solvents and dehydration) using SPR spectroscopy on nano-glass substrates in real time. These SBMs were composed of phosphatidylcholine and DOPC+ including single-component, mixed, protein-reinforced SBMs (rSBMs) and protein-tethered bilayer membranes (ptBLMs). Results show that PC membranes can be removed by low concentrations of surfactants and also dehydration while DOPC+ is relatively stable towards air damage and nonionic surfactant owing to favorable electrostatic interaction between the silicate surface and the lipid head group. Cholesterol doping of PC's improves the lipid membrane packing thus providing protection against dehydration damage. Similarly, rSBMs and ptBLMs were found to be susceptible to damage by low concentrations of ionic and nonionic surfactants though these demonstrated improved stability against air damage. Nanoglass SPR has shown promise for its future biosensing applications68.
Supported membrane array lipid-protein interactions: Wang et al.69 have devised supported membrane array using photolithography for label-free detection of lipid-protein interactions. The SPRi in conjunction with lipid microarray were utilized to study the binding of phosphoinositides and their phosphorylated derivatives (PIPs) with proteins. A number of lipid compositions have been tested for the robustness of resulting membranes when undergoing dehydration and rehydration procedures. DOPC+/PEG-PE system gave the best results in terms of sensitivity, suitability and recovery as depicted by SPR sensograms. The air-stable array devised by Wang et al.69 provides a simple and effective protocol for the construction of functional membrane surfaces for screening applications and possibility of label free study of membrane proteins and lipid-membrane interactions69.
Polymer-supported bilayers: The protocol utilizing polymer supported bilayers involves coating of the surface with a flexible polymer to circumvent roughness of the surface thus providing a cushion between surface and the membrane. This resembles a cytoskeleton that anchors plasma membrane of the cell. The stability of such system can be enhanced by covalently linking the polymer support to the substarte70. This involves chemical grafting to a surface of carboxymethyl dextran that is modified with alkyl chains71, coupling of lipopolymers having functional hydrophobic head groups to the surface; and copolymers having disulphide to anchor the polymer to a gold surface62 Such membrane surface can be used for extremely rapid functional reconstitution of membrane fragments and detergent-solubilized on-a-chip GPCRs7,60.
Membrane arrays: Hovis and Boxer72 have pioneered the concept of membrane arrays using the microcontact printing process to create interface between PDMS and planar supported lipid bilayer. This replaces the adsorbed lipid at the interface area used to fill with blocking agent like Bovine Serum Albumin (BSA). Boden et al.73 have synthesized SAM of hydroxyl- and cholesterol terminating thiols to capture lipid bilayers that can be arrayed using microcontact printing.
Integral Membrane Proteins (IMP): Integral Membrane Proteins (IMP) are pharmacologically important biomolecules. About 25% of human genes encode these proteins and one half of pharmaceutical drugs target membrane proteins. Despite their perceived importance not much have been done to reveal their functional and structural studies. This is partly due to the requirement of these proteins in highly purified form and yield. Also, the requirement of a biological membrane or biomimetic medium to support their structure and function hinders their structural and functional studies. Requirement of IMP in high yield can be met by large scale production in misfolded form followed by in vitro folding. However, folding is not easy to achieve as it involves folding of extra-membrane domain and a transmembrane domain, latter being most difficult. Study by Shenkarev et al.74 have shown the potential of alternative membrane mimicking medium, i.e., Lipid Protein Nanodiscs (LPN) to achieve the folding of recombinant IMPs. The LPN contains a lipid bilayer and two copies of apolipoproteins and Membrane Scaffold Proteins (MSP). The IMPs obtained by the membrane-targeted expression in a folded state and extracted under non-denaturing conditions by mild detergents can be reconstructed into the LPNs without loss in the functional activity75,76. The IMP/LPN complexes ensures long term stability for the enclosed proteins75,76. These results establish beyond doubt that lipid composition of LPNs are key to determine efficient folding of IMPs74.
Peptide membrane interaction: The interactions between peptides and membranes form a key mechanism of cellular events such as protein trafficking, cellular signaling and ion-channel formation. Biophysical techniques are available to characterize peptide membrane interaction but they lack in depicting the affinity of peptides to the variety of membrane systems. The advent of SPR spectroscopy has largely circumvented this issue and is recently applied to the study of biomembrane systems using single and bilayered liposome chemistries thus providing an invaluable tool for increasing our understanding of membrane-peptide structure and function67.
Antimicrobial peptides: Antimicrobial peptides can be a crucial candidate for its use as antibacterial drug/therapy. This is particularly important in wake of increasing drug/antibiotic resistance escalating into ‘superbug’. However, analyzing interaction is not easy due to complex nature of interaction of antimicrobial peptides and cell membrane and also the variability in the mechanism of interaction. Hall and Aguilar77 have described the SPR based study of Melittin, an antimicrobial peptide.
Most Antimicrobial Peptides (AMPs) usually act by disrupting the lipid bilayers. In a study by Nascimento et al.78 Magainin I (MagI) was studied with respect to its interaction with phospholipid layers coated or uncoated with synperonic (Synp) via the combine of Cyclic Voltammetry (CV), Electrochemical Impedance Spectroscopy (EIS) and SPR. Results show that Synp plays the role of sealant after the disruption of lipid membrane with MagI. This study has demonstrated the significance of triblock copolymer to stabilize liposomes to act as drug delivery vehicle for MagI78.
Wilkop et al.79 have studied the interactions of bacterial toxin streptolysin O (SLO) and cholesterol-containing membranes using Electrochemical Impedance (EI) and SPR spectroscopy. The EI monitored the fusion of cholesterol to gold sensor surface modified with Hexyl Thioctate (HT) and charge transfer of resulting lipid membrane, while SPR spectroscopy characterized the bilayer membrane, i.e., monitoring the lipid deposition, incubation with SLO. Results showed that hexyl acetate surface chemistry allows partial insertion of toxins into membrane. This study has valuable applications in nanotransport channel construction for biosensing applications79.
Tethered membrane array: A combination of tethered membrane array technology with SPRi offers an attractive solution to study membrane protein for rapid drug screening. Microfabrication approach was utilized to synthesize lipid membrane array in PDMS microchip to facilitate label free analysis of lipid-protein interactions in conjunction with SPRi. This microchannel equipped platform allowed multiplexed detection, i.e., carrying out multiple measurements simultaneously with high sensing interface reproducibility. This study has utilized lipid-conjugated receptors as model systems for protein binding analysis and interaction of phosphatidylinositol (PIP) with biotin or avidin for array measurements. The results show that the lipid membrane can be removed effectively by non-ionic surfactant like Triton X-100 while the SPR detection signal was enhanced (400%) by layer-by-layer amplification strategy utilizing biotinylated antibody, NeutrAvidin and biotinylated anti-avidin and the signal for protein binding on the membrane80 (Fig. 7).
Single domain antibodies (sdAbs): Single domain antibodies (sdAbs) are crucial for therapeutic and diagnostic applications owing to its small size and low structural complexity when compared to conventional antibodies. This helps in their production as recombinant fusion proteins in range of forms. In a study by Walper et al.81, C-terminal fusions and immobilization strategies (direct covalent attachment and affinity immobilization) for two sdAbs (standard sdAb monomers, biotinylated monomers and fusion constructs of alkaline phosphatase dimers and streptavidin core tetramers) were compared using SPR. Results show that sdAb-streptavidin core tetramer provided the highest density of active molecules followed by the sdAb-alkaline phosphatase followed by site-specific biotinylated monomer81.
Exosomes
Diagnostic markers for health disorders: The SPR can also be utilized for determining the concentration of exosomes in biological fluids. Exosomes are cell secreted, nanometer-sized extracellular vesicles crucial for intercellular communication, important diagnostic markers for health disorders and could have potential application in drug delivery and gene therapy. In a study by Rupert et al.82, concentration of human mast cell secreted exosomes carrying the tetraspanin membrane protein CD63 were determined by measuring their diffusion-limited binding rate to an anti-CD63 antibodies immobilized on SPR sensor surface. Actual concentration was determined by transforming the SPR signal into surface-bound mass, where increase in mass uptake over time was related to the exosome concentration in solution. Accuracy of the concentration was better than ±50%82.
Nanodiscs and Liposomes as membrane mimics: Proverbio et al.83 have characterized the binding of the human endothelin receptors to ligands in different membrane environments and lipid types like nanodiscs and liposomes (Fig. 5). The binding interaction was analyzed through SPR. The highest value was obtained in association with nanodiscs. This setup could be utilized for rapid in vitro screening83.
SPR for toxicological assays: The SPR was applied to monitor the biological activity of Botulinum neurotoxins A and B (BoNT/A and B) to screen for potential inhibitors. The BoNT/A and B are neurotoxins and highly pathogenic, have perceived potential to act as a therapeutic agents. SPR was used to measure membrane vesicle capture by antibodies against SNAP-25 and VAMP-2, an intracellular presynaptic protein located in synaptic vesicle. The BoNT/A and B neurotoxin cleaves these proteins to block the neurotransmission. The SPR based analysis revealed the toxin activity of BoNT/A and B and its activity by using MAb specific to the epitopes on the toxin generated by proteolytic cleavage. Results obtained were powerful enough to establish this technique as a standard procedure and could replace the in vivo mouse assay. The SPR has potential to replace animal models as a worthy alternative for toxicological assays84.
DNA protein interaction: The SPR can also be utilized to study the qualitative and quantitative interactions of single-stranded DNA (ssDNA) binding protein, (SSB)-heterologous protein, providing details of equilibrium and kinetic parameters of the interaction. In one such study, biotinylated ssDNA was immobilized onto the chip surface bound by SSB before analyte was added to the SSB coated sensor chip85. Spin coating protocol was applied for coating very thin film on prism for SPRi application. Results have validated the cost benefit of the protocol with the advantages of rapid film formation at low temperature without requiring a vacuum system86.
SPR and SPR-SPFS to monitor lipid membrane assembly: The SPR and SPR-SPFS were used to monitor the adsorption and reorganization of phospholipid vesicles / lipid membrane assembly on solid substrates. The distance dependency between gold surface and fluorophore due to excitation energy transfer was utilized to obtain information about physiochemical properties of adsorbed membrane and topography. Thus, the combine of SPR and SPR-SPFS provides invaluable information to monitor the lipid membrane assembly on solid substrates. In this combine SPR, SPR-SPFS, SPR provides the information about the membrane thickness adsorbed on the solid surface down to subnanometer range while SPFS provides the information about the topography and physiochemical properties and also the lateral mobility of the membrane associated molecules of adsorbed membrane (Fig. 8). Different fluorescence based techniques like FRAP can be readily combined with SPR and SPFS. This combined platform can be integrated with microscopic imaging system87.
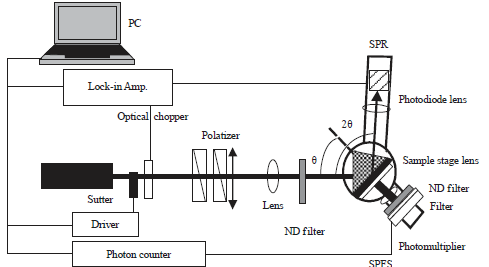 | |
| Fig. 8: | Instrumental setups of SPR-SPFS combine (Reproduced from Tawa and Morigaki107) |
In this study, substrate supported planar lipid bilayers forms the universal system for the fundamental studies related to biological membrane like cell-cell recognition in immune system88; cell adhesion89 and reconstitution of membrane proteins90-92. However, the formation of planar bilayer by vesicle fusion process is complicated and a reliable tools or technique is required to monitor the process. Currently available tool for such type of study like impedance spectroscopy, ellipsometry93, quartz crystal microbalance with dissipation monitoring (QCM-D)94,95 and atomic force microscopy (AFM)95-98 have enhanced our understanding for the process but the complex interplay of various factors that leads to the self-assembly process and the physiochemical attributes of membrane are poorly understood. However, SPR and SPFS combine has significantly improved our understanding about the process coupled with higher sensitivity and detection limit99,100. The distance dependency between gold surface and fluorophore due to excitation energy transfer could be utilized to obtain information about physiochemical properties of adsorbed membrane and topography.
CONSTRAINTS FOR MEMBRANE PROTEIN-LIGAND STUDIES USING SURFACE PLASMON
Effects of the immobilization of reactants on sensor chip: The immobilization of reactants on the sensor chip surface may alter or interfere with the binding characteristics like conformational changes in the binding pocket or steric obstruction of the binding partner. There might be a possibility of macromolecular oligomerization upon immobilization. Non specific immobilization3-5,42 method may lead to the mixed population with variable orientations and affinity, thus broadening the binding isotherm in equilibrium data. This will lead to erroneous characterization of binding kinetics as multi-exponential binding-progress curves.Steric clashes: Increase in the concentration of immobilized partner at the sensor surface will lead to the steric hindrance in binding to neighboring binding site and excluded volume effect. This will broaden the binding isotherm curve of the equilibrium data. However, altering the density of immobilized binding site can do away with the effects of steric hindrance55,61,67.
Mass transport and bulk effects: Mass-transport-limited binding that occurs due to binding between the analyte in the solution and receptor/ligand on the sensor surface is an important cause of concern. This usually happens when the binding of analyte to the ligand happens relatively faster than the diffusion of analyte from the solution. This will also lead to the enhanced rebinding of analyte in disassociation phase due to analyte binding to free receptor before going back to the solution. This phenomenon is integral to the analyte with faster diffusion rate and faster association rate. This could be minimized by immobilizing low level of receptors and using fast flow rates. By including mass transport rate constant into fitness algorithm, binding data can be correctly analyzed excluding mass transport limited binding. Carboxymethyl-dextran hydrogel can significantly retard the diffusion of analyte to the sensor surface. However, in aqueous media carboxymethyl-dextran hydrogel may inflate the hydrogel altering the mass distribution in the evanescent field near the surface55,61,67.
PLASMON-WAVEGUIDE RESONANCE (PWR) SPECTROSCOPY
Plasmon-Waveguide Resonance (PWR) spectroscopy is similar to SPR and utilizes refractive index anisotropy that indicates variation in mass distribution and hence changes in molecular orientation and conformation. This involves overlaying dielectric (silica) on the silver surface thereby narrowing surface plasmon and waveguide excitation modes coupled with line widths. This produces resonances from p and s-polarized excitations permitting direct measurements of anisotropy. The dielectric layer protects the metal layer from chemical and mechanical effects. Optical properties of dielectric layer can be altered to allow the alteration in the relative bandwidths of resonances from p and s-polarized light and the sensitivity of the measurement46.
Integral membrane proteins such as GPCRs, growth factor receptors, ion channels are anisotropic structures and so difficult to study the conformational changes and other dynamic properties in response to ligand binding and signal transduction. Plasmon-Waveguide Resonance (PWR) spectroscopy, a variant of SPR spectrometry facilitates label free kinetics and affinity analysis of integral membrane proteins incorporated into a supported lipid bilayer with high sensitivity and dynamic range while maintaining the receptors in membrane environment. This throws light on the kinetics and thermodynamics of the conformational changes associated with ligand binding to GPCRs, G proteins and other downstream effectors in signal transduction with a high potential for studying membrane signaling and drug development101. In a study by Tollin’s group101, cloned delta-opioid receptor from human brain was incorporated into a lipid bilayer to determine the affinity. The affinity determined using PWR was found to be consistent with those determined by standard binding procedures using membranes or whole cells containing the receptors. This study has also demonstrated the dynamics of GTP/GDP exchange coupled with type of ligand pre bound to the receptor and thus affecting the G protein binding affinity101-103.
The PWR was recently applied to monitor the effects of lipid composition on light induced conformational changes of rhodopsin due to binding and activation of transducin (Gt), simultaneously measuring the exchange of GDP to GTP48,49. This enables the simultaneous measurement of mass density and conformational changes associated with lipid modulation following GPCR activation and signal transduction events. This is particularly important for GPCRs for which the technique of flash photolysis cannot be applied for similar measurements. Also, the lipid composition of bilayer dependent activation of GPCR for binding to G protein has important implications for receptor activity57,104.
The PWR spectroscopy was also utilized to elucidate the mechanisms of signal transduction pathways for the human delta opioid (hDOR) receptor. The study involved purified and detergent-solubilized receptor incorporated onto a solid-supported lipid bilayer thus activating the receptor, which recognizes the different class of ligands with varying affinity providing additional information on the conformational changes of the proteolipid system upon binding to different class of ligands. These informations are possible due to the ability of PWR to differentiate between mass and conformational changes in anisotropic films due to doping of silica on the sensor surface46. Different conformational states observed upon ligand binding were further analyzed by extrapolation of optical parameters and fitting of PWR spectra to the theoretical curves. Results show that agonist binding causes increase in thickness and molecular packing density of the membrane due to the mass movement occurring perpendicular to the plane of bilayer48,49,54,105.
Interaction of G protein with hDOR bound to different types of ligands was monitored using similar set up as above52. PWR revealed the interaction of different G protein subtypes with hDOR thus providing information on the relative contribution of these in the signal transduction pathway. This has shown significant diversity and specificity with respect to the binding of ligand activated receptors and different G protein subtypes and rule out any possible correlation between G protein affinity to the ligand receptor and its ability for GDP/GDT exchange. PWR also elucidated the mechanism leading to side effects of drugs. For instance, some ligands may choose specific G protein subtype over others leading to altered signaling pathways and hence, unwanted side effects. However, by designing ligands specific to desired pathways this can be avoided. The PWR therefore, is integral to the drug design and screening protocol giving new insights into the way physiological and pharmacological effects are exerted by drug.
In a similar study, interactions of the α subunit and βγ dimer with hDOR was investigated. Results show that presence of α subunit increases the affinity of βγ dimer for hDOR in cooperative manner and vice versa48,49. This indicates the important biological role each subunit has to play in specific signaling pathway producing distinct metabolic effects. These informations have important implications48,49 in thwarting the potential side effects of drugs under trial. This may also elucidate the mechanism of drug action and its effects on human physiological system.
Plasmon resonance based protocols have revolutionized the research involving membrane protein-ligand interaction analysis particularly those involving GPCRs, which constitute a very important superfamily of proteins. This was aptly complimented by the advantages of techniques including creation of native membrane environment on the sensor surface, higher sensitivity, low protein requirement, non-invasive procedure involving label free kinetics and equilibrium analysis of interactions thus providing information on the affinity of peptides with different membrane proteins, a distinct advantage over other biophysical techniques. Integration of Membrane array technology with SPR imaging has taken the rapid drug screening to a new level. The SPR has also emerged as a preferred alternative for animal models required for toxicological assays. In backdrop of innumerable advantages, SPR based protocols have its share of pitfalls like conformational changes, steric hindrance, macromolecular oligomerization upon immobilization to the sensor surface. In addition, there are issues related to mass-transport and bulk effects. SPR based protocols need to be improvised to plug these loopholes to realized the full potential of this technique.
| • | This review reports the significance of protein interactions to understand the physiological processes |
| • | Interactions involving G-Protein Coupled Receptors (GPCRs) are important because these are target for several drugs and drug candidates in clinical trials |
| • | GPCRs however, are difficult to purify and study in their native form thus hampering their study |
| • | Surface Plasmon Resonance (SPR) with the advantage of requiring minimal amount of protein, facility to re-create membrane mimetic environment provides the added advantages that facilitates their study |
| • | This review elucidates the standardized protocols for GPCR study, interactions with native binding partners, limitations and advantages of each protocol and future of SPR mediated GPCRs study for their role as potential drug candidates. |
Author acknowledges the support from his organization and inputs from different sources.
Azza Ali Said Reply
This search is very interesting