Research Article
Effect of Treated Drainage Water on the Mouse DNA of Primary Spleen Cell Cultures
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Saudi Arabia is a hot, dry country with rapid population growth, urbanization and development of industrial and agricultural activities which lead to increases water consumption while the resources limited. During the last twenty years most Arab countries invest heavily in projects related to wastewater collection and treatment. In Saudi Arabia, there are 30 major wastewater treatment plants treating around 1.25 million m3 per day. In Riyadh, the capital of Saudi Arabia there is 4 wastewater plants treating around 9 thousand m3 per day. Application of treated wastewater for irrigation of plants and crops is gradually becoming a common practice worldwide (Angelakis et al., 1999). Therefore, wastewater reuse in Saudi Arabia has effectively reduced environmental pollution associated with its treatment and disposal and provided a valuable source of water supply for landscape irrigation at a reduce cost.
However, municipal wastewater may contain a variety of conventional priority organic pollutants that exhibit toxic, carcinogenic or mutagenic properties (Ghosn and Al-Muzaini, 2004). Moreover, treated wastewater may also contain toxic heavy metals, which are hazardous to the environment and humans. These heavy metals may accumulate in the soil, contaminate ground water, or transfer directly to man via vegetables and crops or indirectly via animals eating herbage (Al-Enezi et al., 2004) The common metals found in wastewater are Cr, Pb, Zn, Cu, Ni, Hg, Co and Cd. Those elements are plant toxic, except the latter have minor effect on plant but may accumulate in edible parts and affect their consumers (Hasan and Tjell, 1983; Davis, 1984). In lower concentrations, those substances may have no detectable acute effects on living organisms, but may reduce their survival via long-term chronic effects. Such effects can be manifested by minor or major damage to somatic and germ cells and by the development of disorders such as cancer that requires long, latent periods before becoming clinically visible (White and Rasmussen, 1998; Avishai et al., 2002).
Therefore, it is very important to use a rapid, sensitive and reliable test for evaluation of genotoxic impact at low levels and short exposure time. Out of the available large number of short term tests, only few have been widely used for genotoxic evaluation. One of the sensitive short term assays for detecting DNA damage is the use of the single cell gel electrophoresis or the comet assay, developed by Singh et al. (1988).
The aim of this study was to introduce the comet assay as a monitoring tool for the detection of genotoxic materials in the Riyadh city treated wastewater, Saudi Arabia, year round, on the mouse DNA of primary spleen cell cultures.
Sampling procedures: Water samples were collected every odd month, six time throughout one year in 100 mL capacity polyethylene bottles. The water of each bottle was sterilized through 0.22 μm Millipore filter before checking the pH, salinity and level of heavy metals. This water was diluted by sterile balanced salt solution to correct the osmolarity to 220 mOsM .
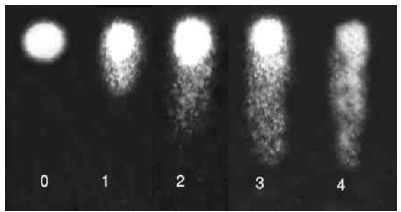 | |
| Fig. 1: | The different cell damage levels in the comet assay |
Isolation of mouse spleen lymphocytes: Using aseptic technique, the spleen of health mouse was removed and placed in a sterile 60 mm culture dish containing 5 mL of serum free medium. The spleen was gripped at one end with sterile fine forceps and using broad forceps, was squeezed progressively along its length, starting at the end held by the fine forceps pushing the spleen lymphocytes out into the medium solution. The cells were pipeted up and down with sterile pipette to desegregate any large clumps of cells and subsequently placed into a sterile tube and centrifuged for 5 min at 200 g. The supernatant was decanted and the pellet of cells was suspended in 5 mL of sterile ammonium chloride buffer (144 mM ammonium chloride, 17 mM Tris pH 7.2) and after 5 min at room temperature, the supernatant should turn a clear red following lysis of the erythrocytes. The volume was made up to 10 mL with serum free medium before centrifugation the mixture at 200 g for 5 min. The supernatant was decanted, the pellet of cells was suspended in 5 mL of medium, cell number was counted by haemocytometer and the cell count was adjusted to be 1x105 cells mL-1.
Cell culturing: To each of sterile 25 mm culture flask containing 10 mL complete medium, a 0.5 mL of cell suspension (1x105 cells mL-1) was added in order to give a final concentration 5x104 cells/dish. The culture flasks were incubated at 37 °C for 48 h. The medium was changed 2 h before exposing cells to treated wastewater. Experiments began by exposing cells in triplicate to 1:1, 1:3 and 1:6 treated wastewater : medium for 2 h at 37 °C. The medium of tissue culture flasks were then removed, the cells were washed with 1 mL cold calcium-magnesium free PBS, then trypsinized with 0.25% trypsin-EDTA solution. After trypsinization, cells were washed with cold PBS and suspended at 1x105 cells mL-1 in ice cold PBS. The cell viability was determined using trypan blue dye exclusion. Flasks representing cell survival >90 % were processed for the comet assay.
The comet assay: The comet assay was performed according to the method of Singh et al. (1998) with some slight modifications. In brief, 10 μL of cell suspension (1x105 cells mL-1) was combined with 100 μL of low-melting agarose at 37 °C and 75 μL immediately pipetted onto Trevigen comet slide. The slides were placed flat at 4 °C in the dark for 30 min until a 0.5 mm clear ring appeared at the edge of comet slide well. The cells were then lysed by immersing the comet slides overnight in lysis solution ( 2.5 M Sodium Chloride, 100 mM EDTA pH 10, 10 mM Tris base. 1% Sodium Lauryl Sarcosinate and 1% Triton X-100) at 4 °C. After lysis, the slides were washed in cold water and placed flat on a horizontal gel electrophoresis tray containing freshly prepared electrophoresis buffer (1 mM ETDA, 300 mM NaOH, pH 13 ) for 30 min to allow DNA unwinding. Electrophoresis was carried out at 20 V and at a starting current of 300 mA for 20 min. Thereafter, the slides were rinsed by dipping several times in dH2O, immersed in 70% ethanol for 5 min and finally air dried . The slides were stained with 60 μL of 20 μg mL-1 ethidium bromide solution and viewed under a Zeiss fluorescent microscope equipped with a 560 nm excitation filter and a 590 nm barrier filter.
Comet evaluation: Slides were examined at 250x magnification and 50 cells were analyzed per slide to evaluate DNA damage. The cells were assessed visually and received scores from 0 (undamaged) to 4 (maximally damaged) according to tail intensity (Fig. 1). Statistical differences between controls and treated samples were determined by student t-test.
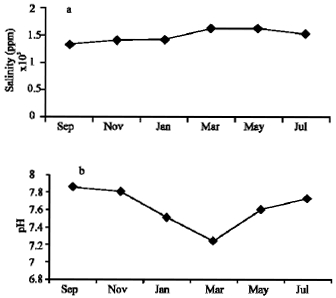 | |
| Fig. 2: | Sample characteristics levels of pH and salinity for years 2004/2005 |
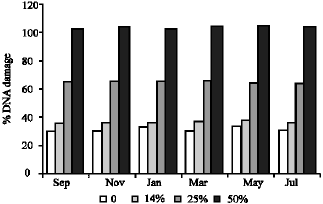 | |
| Fig. 3: | Average percentages of DNA damage at different concentrations for six months |
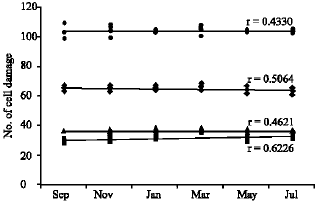 | |
| Fig. 4: | DNA cell damage at different concentration of treated wastewater for six months (� ) control, (▲) 14%, (♦) 25%, (�?) 50% |
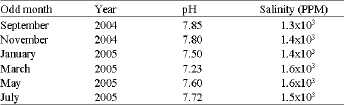
| Table 1: | Sample characteristics levels of salinity and pH |
 | |
RESULTS AND DISCUSSION
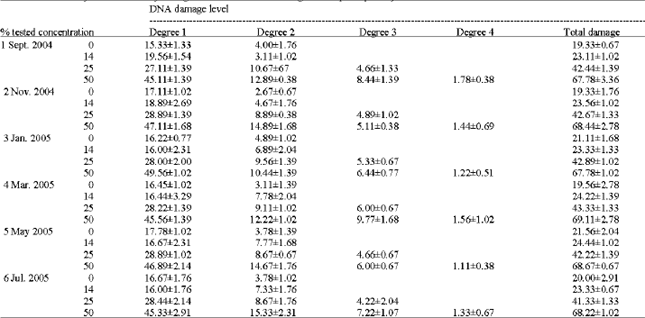
Table 1 shows the salinity and pH values of water samples collected every odd month year round. Values were found to be more or less the same in the case of salinity (Fig 2a and b). The pH values were found to be slightly varied (7.23-7.85) and in March samples the pH values decreased to 7.23. The percentage of DNA damage level and the total DNA damage from 150 cells per sample were given in Table 2 and plotted in Fig. 3. Increasing the percentage of treated wastewater in the growth medium expected to increases the level of DNA damage. No differences were found for DNA damage between 14% and the control cells, but significant increase of damaged cells (p≤ 0.001) were found for both the 25 and 50% when compared to controls. The highest percentage recorded is 43.33 and 69.11% from May samples for the concentration 25 and 50%, respectively. The highest mean value for the whole year is 68.33% for the 50% concentration test. It is clear that no significant differences between monthly samples for each concentration, therefore, the regression coefficient (r) is low for all samples tested (Fig. 4).
It can be expected such treated wastewater may contain a certain potent genotoxins such as N-nitro`s compounds, aromatic amines and heavy metals (Al-Enezi et al., 2004; White and Rasmussen, 1998; Lubello et al., 2004). Most of the domestic wastewater pollutant specially the heavy metals came from laundry detergents (Aonghusa and Gray, 2002). An evaluation of the wastewater treatment plants in Saudi Arabia has been carried out and the final report indicated that all the existing wastewater treatment plants (except one) in the major cities of Saudi Arabia are basically conventional biological treatment plants unable to meet the official stander for reuse (Abu-Rizaz et al., 1995; Abu-Rizaza, 1999). If this is so, then finding a genotoxic effect of wastewater on the primary culture of mouse spleen cells is expected. In conclusion, using the single cell gel electrophoresis or comet DNA assay not only is a rapid, simple, visual and sensitive technique but a cost-effective and reliable tool for genotoxicity screening and monitoring of wastewater and similar systems.
| Table 2: | Comet assay for Genotoxicity testing of treated wastewater using mouse spleen primary culture |
 | |