Research Article
Cerebrospinal Fluid Stem Cell Factor Concentrations in the Children with Meningitis
Department of Biology, The University of Guilan, Rasht, Iran
Zivar Salehi
Department of Biology, The University of Guilan, Rasht, Iran
Meningitis is the most common serious manifestation of infection of the Central Nervous System (CNS). Despite the advances in diagnosis and treatment of infectious diseases, meningitis and encephalitis are still considered to be important causes of mortality and morbidity (Taskin et al., 2004). Rapid diagnosis and appropriate treatment have a crucial influence on survival. The examination of CSF is a cornerstone in diagnostic procedure for patients with suspected meningitis (Kim et al., 2003; Ray et al., 2007).
CSF is secreted continuously by the choroid plexus, located in the lateral, third and fourth ventricles. The fluid flows through the ventricular system, passing over all regions of germinal activity. A number of studies have identified CSF as a carrier of important cytokines, such as transforming growth factor-β (TGF-β), nerve growth factor (NGF), Brain Derived Neurotrophic Factor (BDNF), Neurotrophin-3 (NT-3) and Insulin like Growth Factor (IGF) which are present at specific times during development and under specific physiological conditions (Mogi et al., 1996; Ikeda et al., 1999; Van Setten et al., 1999; Grouzmann et al., 2000). In recent years brain specific proteins as markers for structural brain damage have been widely investigated. There seems to be a close relation between the concentrations of these proteins in the CSF and the severity of brain damage (Dang et al., 2005; Lai et al., 2006; Kirchhoff et al., 2006). Elevated CSF levels of Glial Fibillary Acidic Protein (GFAP) have been found in normal pressure hydrocephalus (Tullberg et al., 1998).
We have previously shown that CSF nerve growth factor level is increased in the patients with congenital hydrocephalus (Mashayekhi and Salehi, 2005) and Alzheimer's disease (Mashayekhi and Salehi, 2006) when compared to the normal group. Since the CSF is in contact with the extracellular space of the brain, biochemical brain modifications could be reflected in the CSF and measurement of intrathecal peptides and amino acids might identify biomarkers of meningitis. Researchers have sought to highlight the correlation between the meningitis process and potential biochemical markers in the CSF (Kahle et al., 2000). Thus, it is important to analyze CSF biochemistry to find a reliable biomarker.
It was shown that SCF and its receptor, c-kit, are highly expressed in cells of the nervous system during development and in adulthood (Zhang and Fedoroff, 1999). SCF and c-kit mediated signaling plays an important role in maintenance of neural stem cells by regulating both the cell proliferation and differentiation (Ani et al., 2004). SCF can appear in two distinct forms; one is soluble and the other is membrane bound (Cheng and Flanagan, 1994). The production of different SCF isotypes seems to be regulated in a tissue-specific manner (Huang et al., 1992). It has been reported that in the brain, most of the SCF produced is in the soluble form and that in the normal adult brain SCF is produced predominantly by neurons (Zhang and Fedoroff, 1997; Ani et al., 2004). SCF is a survival factor for neural stem cells in vitro (Toda et al., 2003; Erlandsson et al., 2004; Dhandapani et al., 2005). It stimulates neurogenesis in vitro and in vivo (Jin et al., 2002).
In this study, the presence and level of SCF in the CSF from acute bacterial and viral meningitis was measured by Western blotting and ELISA.
Patients: We conducted this study in genetics and developmental biology laboratories of the Guilan University, Iran between October 2005 and September 2006.
A total of 48 children with meningitis were followed in this study. The mean age was 82 months and the age range was 18-144 months. Twenty-four children were diagnosed as bacterial meningitis and the other 24 children as viral meningitis. The normal control group was revealed by 24 children whose age range was between 16-133 months.
Meningitis was diagnosed according to evaluation of history, physical examination, CSF laboratory findings, identification of bacterial agents in CSF Gram staining and culture. The control group was defined by an absence of inflammatory cells in CSF. CSF was obtained by lumbar puncture. Samples were taken from both male and female patients. For the lumbar puncture the skin were cleaned with 70% alcohol. CSF of 1 mL was collected and used for this study. After centrifugation for 15 min at 21920xg, the supernatant frozen immediately and stored until analysis. Three independent repeats of each analysis were carried out on each sample.
Antibodies and reagents: Anti-SCF polyclonal antibody was purchased from Abcam plc, Cambridge, UK, whereas Avidin Biotin complex kit was purchased from Vectorlab, Peterbrough, UK. HRP-conjugated anti-mouse antibody was obtained from Amersham, Aylesbury, UK.
Western blotting: For western blot experiments, aliquots of CSF were denatured in a buffer containing 50 mM Tris/HCl (pH 6.8), 2% SDS, 10% glycerol, 100 mM DTT and 0.01% bromophenol blue. Samples were applied to a 5-20% gradient SDS-PAGE gel (Bio-Rad, Milan, Italy) according to the manufacturer’s instructions and the proteins obtained were transferred to nitrocellulose sheets (Bio-Rad-Italy). For the Western blot experiments, the filters were blocked in 5% dried nonfat milk in TBS solution and incubated with appropriate dilutions of the anti-SCF polyclonal antibody (Abcam plc, Cambridge, UK). The excess antibody was removed by sequentially washing the membranes in TBS-T, then a 1/5000 dilution of HRP-conjugated anti-mouse antibody (Amersham, Aylesbury, UK) was added to the filters for 2 h. The peroxidase activity was revealed with diaminobenzidine (0.5 mg mL-1 in PBS with 0.02% hydrogen peroxide). Differences in SCF protein expression on western blots were quantified using Metaview Software.
Enzyme- linked Immunosorbent Assay (ELISA): The SCF level was measured by means of two-site enzyme-linked immunosorbent assay. Microplates were coated with 100 ng/well of anti-SCF polyclonal antibody (Abcam plc, Cambridge, UK). After overnight incubation, the plates were blocked with EIA buffer (50 mM Tris, pH 7.5, 0.3 M NaCl, 0.1% Triton X-100, 1% BSA and 1% Gelatine). The samples and standards were placed in triplicate wells and incubated overnight at room temperature. After washing a biotinylated secondary antibody (10 ng mL-1) was added to each well and the incubation was carried out for two h at room temperature with shaking. β-Galactosidase coupled to avidin was then added for 2 h followed by washing. Finally, 200 lM 4- methylumbelliferyl-b-galactoside (Sigma, Poole, UK) in 50 mM sodium phosphate and 10 mM MgCl2 buffer was added and the amount of fluorescence was measured after 40 min incubation at 37°C. Measurements were performed at least in duplicate. The minimum detection levels were 9.4 pg mL-1 for SCF.
All values are expressed as mean±SEM. Statistical analysis was performed using Student’s t-test. Significance was assumed for p<0.05 in all statistical tests used.
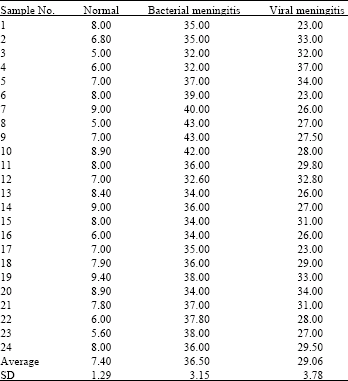
SCF concentration was significantly elevated in acute bacterial meningitis samples, as compared to acute viral meningitis (p<0.001) (Table 1). Median SCF concentrations were 34.25±4.07 pg mL-1 in acute bacterial meningitis samples when compared to acute viral meningitis which was 29.5±3.00 pg mL-1 (p<0.001). The concentration of CSF SCF in normal subjects was 22.1±2.50 pg mL-1. We also analyzed CSF from acute and viral meningitis and normal using SDS-PAGE. A difference on the gel was the presence of a low molecular weight protein (approximately 30 kDa) in the CSF from acute bacterial meningitis, which was weaker in the CSF from acute viral meningitis children.
| Table 1: | CSF SCF concentration in normal, bacterial and viral meningitis (pg mL-1). In each of the experimental groups the No. of samples investigated was n = 24 |
 | |
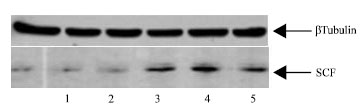 | |
| Fig. 1: | Western blot for SCF protein. Immunoblot analysis Of CSF obtained in normal (Lanes 1, 2), bacterial meningitis (Lane 3 and 4) and viral meningitis (Lane 5). The blot was probed with an anti-SCF polyclonal antibody, as described in materials and methods |
A western blot analysis using anti-SCF antibody as a probe confirmed the presence of SCF (Fig. 1). In order to obtain semi-quantitative estimates of the relative amounts of 30 kDa protein, an image analyzer was used to determine the intensities of the band in the respective lanes. Quantification of the Western blot gels from repeated experiments showed that the amount of 30 kDa was clearly increased in the acute bacterial meningitis when compared with the acute viral meningitis CSF (Table 2). The data from this study has shown that SCF is present in human cerebrospinal fluid. The level of CSF SCF in acute bacterial meningitis is more than that in acute viral meningitis. Furthermore, as SCF was detected in all meningitis and normal samples analyzed in this study, SCF appears to be a constant component of the CSF.
| Table 2: | Relative expression of SCF in normal, bacterial and viral meningitis. An image analyzer was used to determine the intensities of the band in the respective lanes. In each of the experimental groups the No. of samples investigated was n = 24 |
 | |
The present study demonstrates that there are increased levels of the SCF in the CSF of children with acute bacterial meningitis when compared to acute viral meningitis (Table 1 and 2, Fig. 1). We investigated SCF since it is one of the most factors that is important in neurogenesis (Jin et al., 2002) and neural cell survival (Erlandsson et al., 2004; Hirata et al., 1993; Dhandapani et al., 2005).
The c-kit receptor tyrosin kinase is encoded by the W locus in mice (Silva et al., 2006). The ligand for c-kit receptor has been designated as SCF and is encoded by sl locus in mice. In situ hybridization studies revealed that the c-kit receptor and SCF are highly expressed by some neurons (Sun et al., 2006).
Understanding the signals that trigger neuronal proliferation and survival in the brain in vivo could assist the development of cell-replacement therapy for neurological disorders such as meningitis. Efforts to identify these signals have been aided by the ability to grow neuronal precursor cells in vitro. Several factors can stimulate neurogenesis in such systems, including Epithelial Growth Factor (EGF), fibroblast growth factor-2 (FGF-2), BDNF and SCF (Holm et al., 2006; Rossi et al., 2006). We have previously shown that CSF NGF is important in neural cell proliferation and survival (Mashayekhi and Salehi, 2007).
SCF is important in neural cell survival (Zhang and Fedoroff, 1997; Erlandsson et al., 2004), neural stem cell migration and neurogenesis (Jin et al., 2002). Both SCF and its receptor are expressed by some neurons. Increased expression of SCF has been found in neural cells after brain injury (Zhang and Fedoroff, 1997). As SCF is known to be involved in the regulation of survival and differentiation of neurons, it may play a role for the recovery of damaged nerve cells in children with meningitis.
Considering the major roles of SCF in the peripheral nervous system (Dang et al., 2005), it is possible that a high concentration of SCF in CSF in the patients with bacterial and viral meningitis may partially derive from peripheral circulation. The fact that there is a high penetration rate of injected protein into brain supports this explanation. The comparison between plasma/serum and CSF levels should be included in the future studies to better delineate the site of SCF synthesis.
In summary, the CSF SCF concentration is increased in the children with acute bacterial meningitis when compared to acute viral meningitis, which suggests that it is involved in the pathophysiology of meningitis. The data from this study supports the association between SCF expression and meningitis. SCF may be important in neurogenesis and neural cell survival in meningitis and provide a basis for future studies related to neuroprotective mechanisms exerted by SCF in meningitis. In conclusion, CSF SCF levels can be used in the early diagnosis of bacterial meningitis similarly, they may be useful adjuncts in differential diagnosis of bacterial and viral meningitis.
The authors thank all people in the Genetics and Developmental Biology laboratories, Department of Biology, Faculty of Sciences, Guilan University, for their technical assistance. The authors thank Dr. N. Nouri, Shahid Rajaei Hospital, Iran that kindly supplied the CSF samples for analysis.