Research Article
Growth Features and Structure of a Living Cotton Hairs
Arifov Institute of Electronics, 100125 Tashkent, Uzbekistan
Viktor A. Krakhmalev
Arifov Institute of Electronics, 100125 Tashkent, Uzbekistan
There are a great number of papers (Bowman, 1882; Bolls, 1938; Wyssling, 1962; Wareing and Fhillips, 1977; Applequist et al., 2001; Krakhmalev and Paiziev, 2006, 2007) devoted to the structure of an ovary seed, seed cuticle and especially to the morphological development of trichomes and the cell wall pattern of cotton hairs. For example it is shown that approximately one of four epidermal cells on each ovule differentiates into a single-cell trichome (Stewart, 1975) and fibre initials appear on the day of anthesis as spherical protrusions on the ovular surface (Ryser, 1999). Differentiation begins near the chalazal end of the ovule, progressing later toward the micropylar end (Stewart, 1975). On the first and second Days Post Anthesis (DPA), the spherical fibre initials begin to expand lengthwise, bending and growing toward the micropyle. Then the rate of fibre elongation increases (Hof, 1998) and reaching a maximum around 15 DPA (Schubert et al., 1973) and at that the length of the elongation period is highly dependent upon environment conditions (Quisenberry and Kohel, 1975). Regard to secondary cell wall deposition the transmission electron microscopy have demonstrated that the secondary cell-wall thickening phase overlaps with the elongation phase by up to 10 days (Quisenberry and Kohel, 1975).
Despite of more than a centenary period of studying the processes of the cotton fiber evolution (Bowman, 1882) till now it is not quite clear why some cells of outer epidermis of an ovary seed are differentiated to hairs; what differs principally this cell from the other epidermis cells? In fact, the direct observation could not establish the initial stages of the formation and following lengthening of the hairs in a closed unripe cotton boll. In the biology of the cotton growth there are other important, but not studied yet, questions, for example, what is the mechanism of cellulose synthesis, what is mechanic and regularity of cotton hairs arranging in cotton lobule, why ripe cotton hairs have a screw-like twisted form etc. For answering to this important questions we need to draw attention of cotton plant physiologists to new in vivo observing methods in cotton cell biology based on direct observation living cotton cell-hairs on early growth stage without any physical and chemical treatments. But current preparing techniques of cotton seed single-cell trichomes usually based on detaching of seeds from cotton boll, their chemical treatment, liquid-nitrogen refrigeration, metal sputtering before electron microscope investigation and other. Such approach modify surface state of cotton fibre and do not permit to watch some native morphological features of living cotton trichomes.
The aim of this study is to study the in vivo growth and structure of hairs on a cotton seed ovary in the early period of their growth.
The surface of the in vivo ovary seeds and the cells-hairs on them are studied for the early stages of the evolution (from 1 to 15 DPA). The cotton varieties Tashkent-1, 108-F G. hirsutum L., C-6030, C-6524 G. barbadense L. and Turfan gusa G. herbaccum were studied. The cotton bolls with considerable part of plant stem has been harvested during season of 2004-2005. The open flowers were labeled each day at the same time (about 1100 in the morning) and studied by using the universal optical microscope Neophot-2 (Carl Zejss, Jena) in reflected light with magnification up to 4700 time. In all cases the labeled unripe cotton boxes were cut with a considerable part of branch. A small hole (~1 mm2) was made in a segment of the unripe cotton box through which the in vivo observations were performed. The microphotography of the growth of fiber-forming cells of epidermis and the structure of their surface was taken using a highly sensitive negative film of 9x12 cm in size.
The fine structure of the primary cell wall surface of a cotton hairs and adjoining plasma membrane were studied by the method of replica and prints using optical and electronic microscopes. The replica-prints were obtained as follows. A thin non-destructive polymer film was deposited (e.g., by spraying a water solution of gelatin), after drying this film was removed from the plant and studied under optical microscope. The replica-prints from the cross sections of the ripe cotton hairs were obtained by the same method. They were obtained by the method of micro-sections (Krakhmalev et al., 1978) and then studied by an electron microscope to reveal the undistorted forms and define the sizes of supra-molecular patterns appearing in the cell wall of the hairs during cellulose biosynthesis.
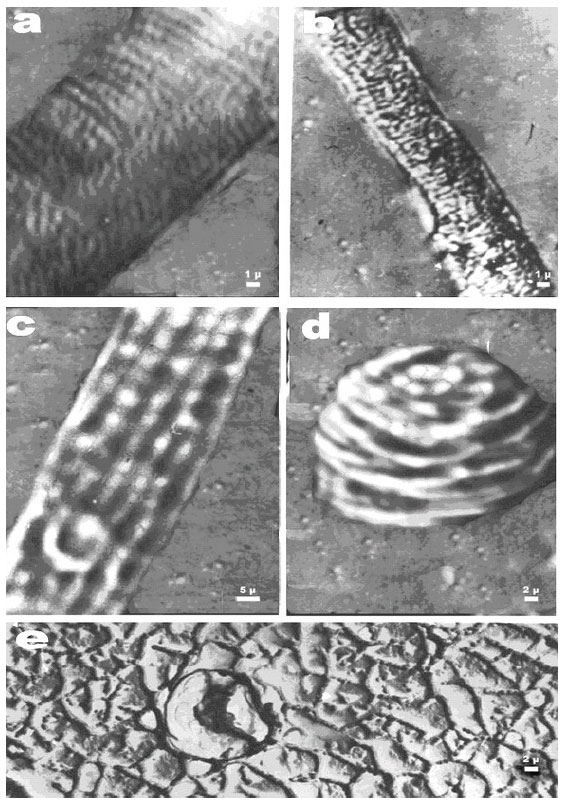
According to Vlasova (1974), on a ovary seed epidermis of different cotton varieties (G. herbaceum, G. hersutum and G. barbadence) at the one day before flowering and at the day of flowering, the pattern of all epidermal cells are similar and there is not any indications in favor of the fiber formation. The surface of the ovary seeds, at this time, is lusterless and smooth, the outlines of the epidermal cells are hardly visible and the orifices are very distinctly (Fig. 1a). The orifice distribution on the ovary seed surface is not homogeneous. In the chalasal part of the ovary seed there are many orifices, in the middle separately located ones and in the micropile part no orifices. The calculations show that on the chalasal part of the ovary seed 300-400 orifices per 1 mm2 are sometimes observed, that is, on the average, 25% of the total number of the epidermis cells per this area. The availability of these orifice fields, evidently, greatly influences the beginning and intensity of the fiber formation along the ovary seed. In fact, the observation shows that the first hairs start to appear on the day of flowering on the ovary seed areas close to the chalasal part where the central beam of the seed leg is branched out.
The following regularity is revealed. Before appearing of the hairs, the epidermal cells become convex. On some of them, the microscopic areas of protrusions with the size of ~ 1 mm2 appear. At the moment of flowering and for the following hours the protrusions are transformed into semi spheres and then into balls of 18-22 μm in diameter. Each cell-hair, in this stage, has a short pedestal connected with the initial epidermal cell. The nuclei of the epidermal cells move into the formed hairs. These nuclei rotating slowly either approach to the geometrical center of the ball or move away from it. No circular motion of cytoplasm in hairs is observed in this period.
On the next day after flowering the number of hairs on the chalasal part increases as avalanche and they begin to appear on the middle part of the ovary seed. The density of the chaotically appearing hairs becomes so high (~2000-2200 mm2) that they come into contact with each other (Fig. 1b) forming the well-known hexagonal picture of densely-packed balls.
In the process of the growth of the epidermal cells into the ball-like hairs (especially at the moment of reaching the limit sizes of 18-22 μm), the microscopic areas of increased plasticity again appear on their top. Under the turgor pressure these areas rhythmically grow into micro protrusions (Fig. 1c) enlarging at their base up to a constant diameter of the hair. The pulsed rhythmic process of the apical part growth of the hairs occurs continuously until the cotton cells reach their limit length.
This growth mechanism can be well seen not only for the separate hairs of the early stages of their evolution but also for those of the late ones, e.g., for the hairs of from 20 to 30-35 days from the flowering day. To prove this, it is sufficient to study the in vivo lobules of the unripe cotton boll using a microscope (Vlasova and Krakhmalev, 1984a, 1986).
It is very interesting to find, in the growing tips of the in vivo hairs, the specific structural elements: the longitudinal beams of the cellulose fibrils that extend beginning with the apical part point of the growth (Krakhmalev, 1991) like the shroud lines of a parachute. The analysis of their disposition along the cotton cell evidences that the apical part rotates about the conventional axis of the hair growth (Fig. 1e). While rotating, the longitudinal filaments, located at the equal distance of about 2 μm from each other, form the flat spirals on the hair surface at an angle of from 3 to 8° with the generatrix of the cylindrical surface of the cell. It is observed that the tip of the growing hair periodically changes the direction of its rotation, clockwise and vice verse. The rotation of the cotton cell core begins from the first moment of its appearing (Fig. 1e) and continues until the limit length of the hair. In this case the apical part of cotton fiber can serve as a classical example of the non-muscular motion of a plant.
 | |
| Fig. 1: | Peculiarities of the surface structure of in vivo ovary seeds and cotton hairs while growing. (a) The structure of the chalasal surface of cotton ovary seeds for a variety Tashkent 1 several hours before the beginning of flowering, (b) The chalasal part of cotton ovary seeds for a variety C-6524 one day after flowering, (c) Some part of the surface of cotton ovary seeds for a variety 108-F on the day of flowering, (d) Longitudinal beams of cellulose fibrils near the growth tip of the 3-days-old hairs of cotton ovary seeds for C-6030, (e) Rotation along the screw-like line of the appearing cell-hair on the surface of cotton ovary seeds for C-6024 on the day of flowering and (f) Periodical protrusions of the plasmalemma surface of in vivo hairs on the cotton ovary seeds that move like a wave to the apical part of the cotton cell growth |
The screw-like spinning of the fibril core of the apical part of the hairs, the periodical alteration of the direction of their motion influence the formation of the so-called twists on the ripe cotton cells while opening the cotton boxes and, as known, making a great impact on the technological properties of cotton fiber (Popova, 1975).
The nature of the apical part rotation of the cotton hair is not clear until now but evidently this nature is connected phylogenetically with the rudiments of the motional process of the simplest organisms (Apponet, 1978). The similar screw-like growth is characteristic of the simplest plants, exclusively of fungus Phycomyces sp. (Razdorsky, 1949).
In the apical part of the growing hair, not only the longitudinal beams of fibrils are formed but also rings and spirals from them. Earlier, by using copper-ammonium solutions it was found that the primary thickening of the cell wall as fibril rings and spirals is observed in this part of the hair after 5, 10 and 45 DPA (Vlasova and Krakhmalev, 1984b).
It is naturally expected that by studying the surface structure of cotton fiber with an electronic microscope the fibrile filaments be observed along the whole length. However, this is not confirmed. Evidently, being formed during the early stages of the growth, the fibrile beams are not stable and in the process of fiber ripening they are subject to change or decayed. The longitudinal fibrile beams are found only on the apical parts in the form of short lines. This evidences that the formation of the longitudinal cellulose filaments occurs only on the apical part of the cell. These beams degrade with the increase in the distance from the growth point of the hair.
In the growing in vivo hair not only the longitudinal filaments of fibriles going from the apical part point of the growth but also a characteristic cross structure are well seen. For the first time this structure was found for 3-days-old cotton fiber by a polarization microscope (Usmanov and Nikonovich, 1962). The cross structures in the in vivo cotton hairs in the early stages of their growth (1-15 days DPA) when they are transparent are well seen in reflected light (Fig. 1f).
The analysis shows that the light and dark cross areas in the in vivo hairs are the successively located protrusions and hollows of the plasma membrane- 2.5-2.6 μm in width. The study of the hairs close to the point of their growth by the method of replications-prints shows that the cross structure has a fine structure; in fact, large protrusions and deflections are a set of smaller ones (from 1.25 to 1.37 μm). With the increase in the age, observed with an optical microscope, the cross structure of the hair becomes rougher, the separate narrow corrugated bands grouped by 4-6 pieces in a set sag into cytoplasm.
Not the cross structure in itself is surprising and unusual but the fact that plasma lemma, its surface in the in vivo hair is in the continuous oscillation motion. This is well seen for the young hairs because they are quite transparent, as mentioned. It is seen with a microscope that the protrusions and deflections of the plasma membrane move along the in vivo hair to the point of growth owing to the wave-like oscillations of the plasma membrane surface. These observations make it possible to assume that the similar wave-like oscillations of the plasma membrane surface take place, evidently, in all in vivo cells of various organisms, including single-cell ones. It is not excluded that the auto-oscillations of the charged plasma membrane surface in the cotton hair generate electromagnetic phenomena on its surface, making the substance particles from the bulk of the cell move to or along its surface. From this point of view, the single cotton cell is an ideal oscillator of electromagnetic fields. Owing to the active mechanic mobility of plasma membrane, the hair cytoplasm makes the directed motion. The reverse flow of the cytoplasm in the hairs is realized according to the reverse-fountain principle described in detail for the plant cells (Kamiya, 1962). The transparency and small thickness of the cell wall of the young hairs make it possible to study the outer surface of plasma membrane by the method of replications-prints (Fig. 2a). It is seen that the plasma membrane has, in the stationary state, the form of corrugation the folders of which are almost perpendicular to the axis of the hair growth. On the upper part of the folders there are round formations creating the symmetrical images along both the corrugation folders and the growing hair. The round formations are always connected with the outer surface of plasma membrane.
We assume that the round formations are the associates from the so-called terminal complexes what was observed in the cells of higher plants by the method of freezing and chips (Mantezinos and Brown, 1978). The ranges of such globular particles connected with the plasma membrane are found not only by prints but also by cutting the cell walls (Fig. 2b).
Visible through the first wall, the diameter of the associates of the terminal complexes (without the thickness of the cell wall) is approximately 0.2-0.3 μm. The distances between the ranges of the round formations visible on the folders of corrugation are 1.22 μm. The linear density of the globular associates along the ranges is 820 pieces mm-1 and the surface density (taking into account the wave-like form of the plasma membrane) is approximately 106 per 1 mm2.
The studies show that the disposition of the ranges of the globular particles on the plasma membrane surface of the hairs depends on the hair age. Three days from the flowering, the associates of the terminal complexes are disposed on the plasma membrane as chains oriented along the growth axis (Fig. 2c). After that the globules are redisposed first diagonally to the growth axis, then rearranged to the almost cross ring structures.
 | |
| Fig. 2: | Peculiarities of the plasma lemma structure of in vivo hairs of cotton ovary seeds (a) Rows of the round formations-associates of the terminal complexes on the surface of the protrusions and hollows of hair plasma lemma (b) Rows of the spherical particles on the plasma lemma surface of the hairs visible after the detachment of the primary cell wall (c) Rows of globules on the plasma lemma surface of the 3-day-old hair (d) The location of the terminal complexes or their associates on the apical point of the growing hair and (e) The shape of the cellulose fibril beams in the cross section of the native ripe hairs of cotton |
Since the terminal complexes are molecular machines (Giddings et al., 1980) producing the cellulose micro fibrils from cytoplasm polysaccharides, their reorientation must make an impact on the character of microfibrile arranging in the layers of the cell wall. In fact, in the cotton hair of the 1-3 day growth, the fibrile beams, according to the electronic microscopy data (Roelofson, 1951), are oriented parallel to the axis of the cell growth on the outer surface of the primary wall and diagonally on the inner one. This coincides with the orientation of the globular particles in the in vivo hairs for each moment of fibrilogenesis. The periodical alterations of the direction of the screw-like motion of the growth tips are the reasons for the motion of cellulose spirals of the second wall (Usmanov and Nikonovich, 1962) and for the formation of twists on the fiber surface (Warwicker et al., 1970). The terminal complexes or their associates located on the most active growing point of the apical part are of special interest. The observations of this point show that the terminal complexes (or their structural combinations) continuously move on the plasmamembrane surface, forming for 1-2 s the different geometrical patterns that are smoothly transformed into each other (Fig. 2d). In the center of these patterns there is often only one associate of the terminal complexes, seldom a group of three ones. In the sub-apical part, the ranges of the globular particles do not move.
The observations of the apical parts of the growing hairs on some ovary seeds, as well as the direct measurement of the rates of their growth on the cotton segments show that the process of the cell-hair growth has an oscillatory character and depends on the location of the hair on the ovary seed epidermis, on curvature of the apical part surface and the hair age. The growth rates are minimal (up to 0.13 μm min-1) in the first days after flowering and maximal (from 0.95 to 1.2 μm min-1) for the period of 6-20 days from flowering. After 20-23 days of the growth, the growth rates decrease considerably and by 30-35 days from flowering close to zero (less than 0.04 μm min-1). The time spent by the ovary seed for one act of protrusions-hair growth is 16.3, 4.3 and 3.2 min for the 3, 10 and 20-day-old hair, respectively. For the 30-day-old hair this time is 90 min. The growth rate of cotton 108F periodically increases and decreases. The growth rates are within some interval of values. The oscillation period of the rate of the in vivo hairs is close to 4.5-5.0 min. This is quite characteristic of the rhythm of some physiological processes in animals and plants (Polevoy, 1989). It is quite possible that the oscillation character of the hair growth is due to the oscillatory processes occurring inside the nucleus of the cotton cell.
It is interesting to clarify the question about the real arranging of cellulose microfibrils in the cell wall of the native hairs after the processes of their lengthening and complete ripening are over, what independent structural elements they form. This is principally significant for the biology of the cotton hair evolution.
For this purpose, the replications from the cross sections of the hairs obtained by the method of micro-sections (Krakhmalev et al., 1978) were studied with an electronic microscope. The results are surprising. In the cell wall of the native hairs, the cellulose microfibrils are assembled in beams but with no grouping in layers, as observed for chemical treatment leading to swelling (Wareing and Fhillips, 1977; Usmanov and Razikov, 1974). Moreover, the cross section of the cellulose fibril beams is not round, as assumed in different hypothetical schemes of their construction (Usmanov and Nikonovich, 1962), but polygonal (Krakhmalev et al., 1984). The typical formations from the cellulose microfibrils (Fig. 2e) with the cross section of 0.1 μm2 have the shape of rectangle, triangle, hexagon and others located relative to each other at an angle of 90 and 120°. This hair structure in the cross section looks like a mosaic structure of metallic monocrystals (Krakhmalev and Kleyn, 1972) grown by electron-beam zone melting.
Thus, the studies of the in vivo cotton hairs show that the available orifices influence greatly the fiber formation and the differentiation of the outer epidermal cells of the ovary seeds into hairs. The processes of the hair appearing from the epidermal cells are fortuitous and begin with the arising of microscopic protrusions. Under the pulsed inner-cellular pressure the protrusions are elongated rhythmically and simultaneously enlarged at their base up to the limit diameter of the hair. This hair makes the screw-like motion about the axis of the cotton cell growth. The screw-like rotation of the apical part is accompanied simultaneously by continuous wave-like oscillations of the plasma membrane surface in the hair. In the stationary state the plasmic membrane is assembled from protrusions and hollows on the surface of which the ranges of the round associates of the terminal complexes are located. The orientation of these ranges on the plasma membrane surface depends on the hair age and changes from longitudinal to cross. As a result of this rearranging, as well as of the screw-like rotation of the apical part, the cotton hair has the specific corkscrew-like form.