Research Article
Bioleaching of Copper from Saindak Ore in Columns
PCSIR Laboratories, P.O. Box 387, Mastung Road, Quetta
Shereen Gul
PCSIR Laboratories, P.O. Box 387, Mastung Road, Quetta
Bioleaching processes are based on the bacterial oxidation of sulfide minerals involving ferrous iron and various organic compounds as the electron donor (Tuovinen et al., 1991; Kelly and Tuovinen, 1988). In bioleaching the Fe++ iron is first oxidized to Fe+++ iron in acidic medium. This Fe+++ iron attack the ore and releases the copper in the medium in the form of copper sulfate.
![]()
Commercially the bioleaching processes are used for the dump recovery of copper (Rossi, 1990) and in tank leaching for the pretreatment of refractory pyrite and arsenopyrite containing gold ores (Lindstrom et al., 1992). The bioleaching of other mineral such as Gold, Zinc, Uranium and Manganese, has reported by Krafft and Hallberg (1993), Munoz et al. (1993). Toro et al. (1993) and Chapman et al. (1993), Ahonen and Tuovinen (1991) reported the column leaching of ores, which lasts for more than one year. The environment temperature effects the bioleaching of different ores (Ahonen and Tuovinen, 1992). The direct bioleaching processes are very slow and takes very long time for the leaching of minerals in appreciable amounts. Ahonen and Tuovinen (1991) reported bioleaching of copper from chalcopyrite at 28 percent per year and zinc from sphalerite at 27 percent per month at 37°C. The present study were carried out on the bioleaching of Saindak ore for the efficient and low cost recovery of copper.
The Saindak ore is a composite of Chalcopyrite, Pyrite, Magnetite, Gold and Silver. The ore used in the studies contain 0.439 percent copper including 0.038 percent copper as copper oxide.
A culture of Thiobacillus ferrooxidan was isolated from the Soar Range coal mine water on 9K medium (Silverman and Lundgren, 1959). The culture was maintained in mineral salt solution (g/lit) ammonium sulfate 3.0, potassium chloride 0.1, Dipotassium hydrogen phosphate 0.5, magnesium sulfate 0.5 and calcium nitrate 0.01, pH 2.0 supplemented with 10 percent w/v Saindak ore.
Inoculum preparation: The culture was grown in 9K medium for 15 days at 35±1°C. The culture was harvested by centrifugation and resuspended in 0.005 M H2SO4. The inoculum contained — 5X105 bacteria per mL.
Preparation of acidic ferric sulfate solution: The acidic ferric sulfate solution was prepared by using mineral salt solution (g/lit) ammonium sulfate 3.0, potassium chloride 0.1, magnesium sulfate 0.5 and ferrous sulfate 20.0. The pH of the solution was set at 2.0 by 10 N sulfuric acid. 60 liters of the mineral salt solution was inoculated by 10 percent v/v inoculum containing ~5X105 bacteria per mL and incubated at 35±1°C. The aeration of the medium was started after 24 hrs and continued up to 240 hours of inoculation. After than the medium was kept standing for further 120 hrs.
Leaching of copper in small columns: The column (dia 2.5", length 48" ) was charged with 750 g of 18-8# size ore and was held at 35 ± 2°C. The biologically oxidized, acidic ferric sulfate solution (leaching solution) was circulated by peristaltic pumps. The flow rate was set at 20 mL per minute. 1:4, 1:2, 1:1.33 and 1:1 ore: leaching (g: mL) solution ratios were tested for maximum copper leaching.
Leaching of copper in large columns: The columns (dia 6" and length 96") were charged with 60 kg of 18-8# size ore and held at 35 ± 5°C. One hundred twenty liters of leaching solution was circulated by centrifugal pumps. The flow rate was set at 1 lit. per minute. The loss in volume due to evaporation of the leaching solution was maintained by adding 0.005 M sulfuric acid. The leaching solution was circulated till it became fully pregnant with copper.
The total concentration of iron of the oxidized and copper pregnant solution was determined by o-phenanthroline method (Herrera et al., 1989). The copper contents of the pregnant solution were determined by AAS 2380 Perkin Elmer, USA.
Fig. 1 shows that the conversion of Fe++ to Fe+++ started after 24 hrs. The rate of the oxidation was slow initially but after 96 hrs. it became quite fast and remains as such till 288 hrs. The oxidation of Fe++ was statistically stopped at 360 hrs. After 360 hrs ferric iron concentration reached to 2950±20 ppm. The initial slow rate of the oxidation is probably because of the lag phase of Thiobacillus growth. This phase lasts for 96 hrs. It is also clear from the Fig. 1 that Thiobacillus ferrooxidan oxidized Fe++ in log phase of growth more efficiently than in the lag phase of growth and remained constant in stationary phase. The oxidation of Fe++ slow down and became more or less constant in stationary phase of growth which may be because of the toxicity of high concentration of Fe+++ ion to Thiobacillus, which retarded the metabolism, involved in the oxidation of Fe++. Said and Jhonson (1987) and Pakinkar and Agate (1987) have also reported the tolerance of Thiobacillus sp. to metals.
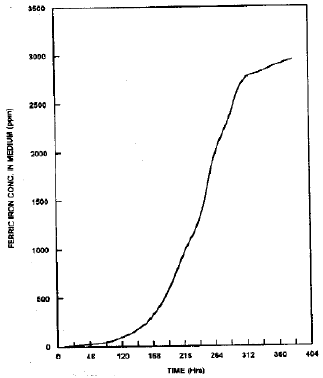 | |
| Fig. 1: | Oxidation pattern of ferrous iron by Thiobacillus ferrooxidan |
Copper leaching in columns: Chalcopyrite is the main source of copper in the Saindak ore material and the secondary copper mineral i.e., copper oxide is in negligible amounts. The effects of the leaching solution: ore is presented in Fig. 2 shows that the leaching of copper was inversely proportional to the quantity of the leaching solution used and ferric ion consumed. The ratio 1:1 was found best for the maximum leaching of copper. In this ratio the copper leached was 16 percent better from 1:1.33, 31.77 percent from 1:2, 38.94 percent from 1:4 ratio. The decrease in the leached copper in larger quantities of leached solution because of the greater deposition of Fe++ iron on the surface of ore, which hinder the release of copper from the ore.
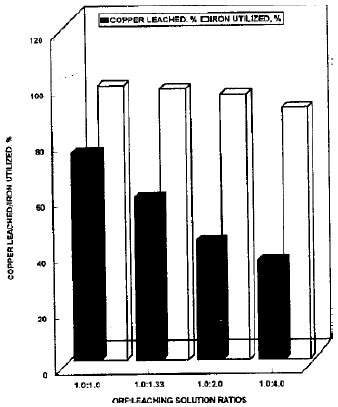 | |
| Fig. 2: | Efficiency of ferric iron for copper release in different ore: leaching solution ratios |
In small columns (dia 2.5", length 48" ), the increase in the concentration of copper in the leach solution displayed more or less linearity up to 96 hrs and same was true for the decrease in the iron contents. The copper concentration in the pregnant solution was 70.65 after 120 hrs of leaching and only 4.21 percent increase in copper contents was observed from 120 to 144 hrs. This shows that the copper leaching was stopped statistically after 120 hrs. A total of 74.86 percent of copper present in ore was leached in 144 hrs (Fig. 3).
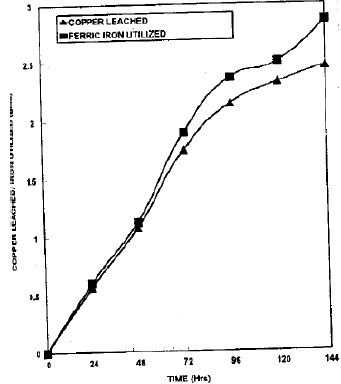 | |
| Fig. 3: | Copper leaching and iron utilization pattern in small columns |
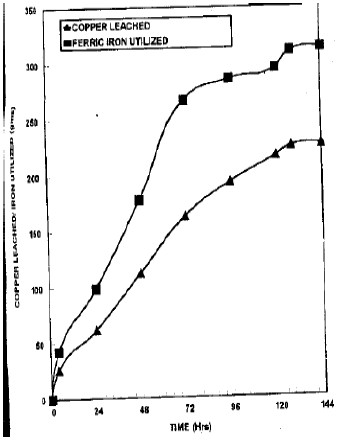 | |
| Fig. 4: | Copper leaching and iron utilization pattern in 60 kg columns |
Fig. 4, shows that the pattern of leaching of copper in large columns was more or the less the same as that of small columns. However the quantity of copper leached was increased from 70 to 88.39 percent with a difference of 8 hrs in leaching time.
The average rate of copper leaching was 2.23 percent per hr up to 72 hours. However after 72 hrs it was declined to 1.14 percent per hr but remain significant (p = 0.01) up to 128 hrs. The rate of leaching became non-significant after 128 hrs (rate = 0.08 percent per hour). In large columns although the copper leaching was increased from 74.86 to 85.92 percent but the efficiency of the Fe+++ iron in the leaching solution was decreased i.e., from 1.08 gm iron/gm copper to 1.49 gm iron/gm copper. 88.64 percent copper leaching from the chalcopyrite concentrate was obtained by Khinvasara and Agate (1987) in glass bioreactor under efficient aeration and high rates of oxygen and carbon dioxide mass transfer achieved under good agitation conditions. Krafft and Hallberg (1993) got 0-0.03 gm Cu /lit from the S-ore and 1.5-1.9 gm Cu/lit from K-ore. Ballester and Cooper (1987) achieved a leaching rate of 260 mg/lit/hr when they treat the chalcopyrite concentrate with the conditioning solution containing Cu++, Ag+ and ![]() prior to the inoculation with Thiobacillus ferrooxidan.
prior to the inoculation with Thiobacillus ferrooxidan.
The results of the present study gives a base for the pilot-plant and commercial bioleaching process for the efficient recovery of copper from Saindak ore as well as from chalcopyrite ores of copper weather high or low grade. The two stage process permits the separate enhancement of two processes involved in the bioleaching of minerals. On the one hand the biological oxidation stage is more effective and fast, at the same time inhibitory phenomenon produced as a result of the bacterial breaking caused by the abrasion of the solid particles on microorganisms is prevented. On the other hand the chemical attack of the ferric iron to sulfide ore can be performed at higher temperatures to get enhanced recovery rate of copper from the ore, to shorten the processing time.