ABSTRACT
Mortality of Meloidogyne javanica larvae was directly proportional to the concentration of culture filtrates of all tested fungi viz., Verticillium chlamydosporium„ Paecilomyces lilacinus and Talaromyces flavus and the duration of exposure. Maximum mortality of larvae was observed in higher concentration of P. lilacinus (82%) followed by V. chlamydosporium (66%) Similarly different dilutions of culture filtrates significantly inhibited hatching of M. javanica eggs. Larval emergence was however, inversely proportional to filtrate concentrations. Significant lower hatching was observed with P. lilacinus 17%) followed by V. chlamydosporium (15%) in 'S' concentrations.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.1999.161.163
URL: https://scialert.net/abstract/?doi=pjbs.1999.161.163
INTRODUCTION
Various biocontrol agents viz., Verticillium chlamydosporium and Paecilomyces lilacinus which are parasites of eggs of root knot and cyst nematodes (Jatala et al., 1979; Zaki and Maqbool, 1993) have also been found to effectively control root knot nematodes on crop plants (Zaki and Maqbool, 1991, 1996). Talaromyces flavus, a biocontrol agent of root infecting fungi (Fahima and Henis, 1990; Ehteshamul-Haque et al., 1994) has been reported to reduce root knot infestation on okra plants (Zaki and Maqbool, 1998). Fungi and phytoparasitic nematodes being a common inhabitant of crop rhizosphere showing synergistic, antagonistic or antibiotic relationship (Jorgenson, 1970). Since they occur together in the rhizosphere, the toxic metabolite naturally produced by microorganisms may be responsible for keeping low level of nematode population. Comparatively little work has been done about the effect of toxic fungal metabolites on nematodes. It was therefore, considered useful to study the effect of fungal culture filtrates of V. chlamydosporium, P. lilacinus and T. flavus on hatching and mortality of root knot nematode larvae.
MATERIALS AND METHODS
Pure cultures of V. chlamydosporium, P. lilacinus and T. flavus were obtained from the Culture Collection of the Department of Botany, University of Karachi. Culture filtrates were prepared by growing these fungi in Potato Dextrose Broth in 250 mls flasks, 100 mls in each, for 15 days at 28°C. Mycelia! mats were removed and the media were filtered through Whatman No. 1 filter paper. This solution was designated as standard (S). Further dilutions (25, 50 and 75%) were prepared by adding requisite amount of sterilized distilled water. Two mls of each solution and 0.1 ml of distilled water containing about 50-60 second stage larvae of M. javanica were poured in each cavity block. Nematodes kept in distilled water served as control. Each treatment was replicated thrice. Number of dead nematodes (unmoved) in each cavity block were counted after 24, 48 and 72 hours.
Freshly collected equal sized egg masses were transferred to each cavity block (one egg mass per cavity block) containing 4 mls of different dilutions of culture filtrates. Egg masses placed in distilled water served as control, There were three replicates for each treatment. Number of hatched larvae in each treatment was counted after 24, 48 and 72 hours. Means egg hatch and larval mortality were calculated and data was statistically analysed.
RESULTS AND DISCUSSION
The larval mortality percentage differed with species and concentrations of culture filtrates. Mortality of M. javanica larvae was directly proportional to the concentrations of filtrates and the duration of exposure (Table 1). Culture filtrates of P. lilacinus and V. chlamydosporium were more effective in causing death of larvae as compared to filtrate of T. flavus and control. Maximum mortality of larvae was observed in higher concentration of P. lilacinus (82%) followed by V. chlamydosporium (66%).
Similarly different dilutions of culture filtrates significantly inhibited hatching of M. javanica eggs. Larval emergence was, however, inversely proportional to filtrate concentrations. Significant lower hatching was observed with P. lilacinus (7%) followed by V. chlamydosporium (15%) in "S" concentrations (Table 2).
There are reports that culture filtrates of several soil borne fungi are known to exhibit nematicidal action beside inhibiting larval emergence of plant parasitic nematodes (Alam et al., 1973; Cayrol et al., 1989; Ali, 1990). Helminthosporium nodulosum, Trichoderma lignorum, Curvularia tuberculata, Penicillium corylophilum and A. niger killed the nematode and inhibited the egg hatch of M. incognita and Hoplolaimus indicus (Alam et al., 1973). Culture filtrates of Aspergillus spp., were found to have some nematicidal properties (Mankau, 1969; Khan et al., 1984). Culture filtrates of Fusarium oxysporum and F. solani exhibited high degree of nematicidal effect on M. incognita (Mani and Sethi, 1984). Arthrobotrys oligospora and Dactylaria brachopage inhibited hatching of M. incognita (Walia and Swarup, 1985).
| Table 1: | Effect of fungal culture filtrates on mortality percentage of second stage larvae of Meloidogyne javanica |
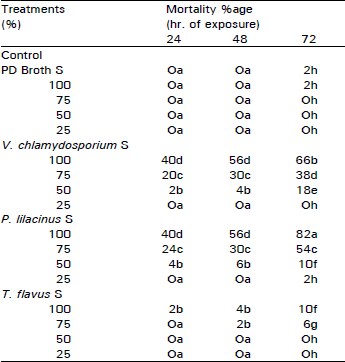 | |
Means followed by the same latters in each column are not significantly different at 5% level according to Duncan’s multiple range test | |
| Table 2: | Effect of fungal culture filtrates on hatching of Meloidogyne javanica eggs |
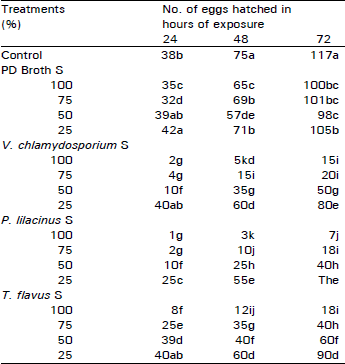 | |
| Means followed by the same letters in each column are not significantly different at 5% level according to Duncan's multiple range test | |
In the present studies the culture filtrates of all tested fung inhibited egg hatch and mortality of M. javanica to a varying degree. The difference in response to the nematode may due to differences in the nature of toxic metabolites due to differences in the nature of toxic metabolites Verticillium and Talaro myces are known to produce toxins and antibiotic like paecilotoxin, verticillin A, B and C, vermiculin, vermicillin, talaron and vermiotatin (Minato et al., 1973; Fuska et al., 1979; Mikami et al., 1989). 0ur results strongly indicate the production of toxins by P. Iilacinus and V. chlamydosporiurn in growth media whip have adverse effect on the hatching of eggs and mortality of second stage larvae of M. javanica. There is need to characterize the nematicidal principle present in the culture filtrates of these fungi.
REFERENCES
- Ali, A.H.H., 1990. Nematicidal action of fungal culture filtrates. Jap. J. Nematol., 20: 1-7.
CrossRefDirect Link - Cayrol, J.C., C. Djian and L. Pijarowski, 1989. Study of the nematicidal properties of the culture filtrate of the nematophagous fungus Paecilomyces lilacinus. Revue de Nematologie, 12: 331-336.
Direct Link - Ehteshamul-Haque, S., M.J. Zaki, M. Abid and A. Ghaffar, 1994. Use of biocontrol agents with Bradyrhizobium japonicum in the control of root-knot nematode in chickpea. Pak. J. Nematol., 12: 149-154.
Direct Link - Fahima, T. and Y. Henis, 1990. Interactions between Pathogen, Host and Biocontrol Agent: Multiplication of Trichoderma hamatum and Talaromyces flavus on Roots of Diseased and Healthy Hosts. In: Biological Control of Soil-Borne Plant Pathogens, Hornby, D. (Ed.). CAB International, Wallingford, Oxon, ISBN: 0851986374, pp: 165-180.
- Khan, T.A., M.F. Azam and S.I. Husain, 1984. Effect of fungal filtrates of Aspergillus niger and Rhizoctonia solani on penetra non and development of root-knot nematodes and the plant growth of tomato var. marglobe. Indian J. Nematol., 14: 106-109.
Direct Link - Mani, A. and C.L. Sethi, 1984. Effect of culture filtrates of Fusarium oxysporium f. sp. ciceri and Fusarium solani on hatching and juvenile mobility of Meloidogyne incognita. Nematropica, 14: 139-144.
Direct Link - Mikami, Y., K. Yazawa, K. Fukushima, T. Arai, S.I. Udagawa and R.A. Samson, 1989. Paecilotoxin production in clinical or terrestrial isolates of Paecilomyces lilacinus strains. Mycopathologia, 108: 195-199.
CrossRefDirect Link - Minato, H., M. Matsumoto and T. Katayama, 1973. Studies on the metabolites of Verticillium sp. structures of verticillins A, B and C. J. Chem. Soc. Perkin Trans., 1: 1819-1825.
CrossRefDirect Link - Zaki, M.J. and M.A. Maqbool, 1993. Occurrence of Verticillium chlamydosporium an egg parasite of root-knot nematodes Meloidogyne javanica and M. incognita in Pakistan. Pak. J. Nernatol., 11: 37-40.
Direct Link - Zaki, M.J. and M.A. Maqbool, 1998. Use of biocontrol fungi with carbofuran in the control of root knot nematodes in Okra. Pak. J. Biol. Sci., 1: 27-28.
CrossRefDirect Link








